Proliferative Vitreoretinopathy (PVR) is a severe and often sight-threatening complication that can arise after retinal detachment surgery. It represents a complex biological process where cellular proliferation and contraction occur within the vitreous gel and on the surface of the retina, leading to the formation of scar-like tissue. This abnormal tissue can exert traction on the retina, causing it to wrinkle, fold, and eventually detach again, often in a more extensive and challenging manner than the initial detachment. Understanding the pathogenesis, clinical manifestations, and management strategies for PVR is crucial for ophthalmologists and retinal specialists aiming to preserve vision in affected patients.
Understanding the Pathogenesis of PVR
The development of PVR is a multifactorial process, broadly categorized into several stages, each characterized by distinct cellular and molecular events.
Cellular and Molecular Mechanisms
At its core, PVR involves the activation and migration of various cell types, primarily retinal pigment epithelial (RPE) cells, glial cells (astrocytes and Müller cells), and fibrocytes. Following a retinal break or surgical intervention, these cells are exposed to the vitreous cavity.
- Inflammation and Cytokine Release: The initial insult, whether it’s a retinal tear or the surgical trauma, triggers an inflammatory response. This involves the release of pro-inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interleukins (IL-1, IL-6), and transforming growth factor-beta (TGF-β). These mediators play a pivotal role in attracting and activating RPE and glial cells.
- Epithelial-Mesenchymal Transition (EMT): RPE cells, which normally form a single layer behind the retina, can undergo EMT. This is a process where stationary epithelial cells transform into migratory, fibroblast-like cells. During EMT, RPE cells lose their characteristic cell-cell junctions, gain motility, and begin to proliferate and produce extracellular matrix (ECM) components.
- Fibroblast Activation and ECM Deposition: Activated RPE cells and glial cells differentiate into myofibroblasts, which are characterized by their contractile properties and their ability to synthesize large amounts of ECM proteins, including collagen, fibronectin, and glycosaminoglycans. This deposition of ECM leads to the formation of membranes on the posterior surface of the retina and within the vitreous.
- Contractile Forces: The myofibroblast-like cells within these membranes exert significant contractile forces. This chronic traction on the retina can pull it away from its underlying supportive layers, leading to folds, striae, and ultimately, redetachment. The contraction of these membranes is a hallmark of PVR and is responsible for its progressive nature.
- Vascular Endothelial Growth Factor (VEGF): VEGF is also implicated in PVR. It promotes neovascularization and increased vascular permeability, potentially contributing to inflammation and further ECM deposition.
Pre-disposing Factors and Risk Factors
Certain factors increase an individual’s susceptibility to developing PVR:
- Initial Retinal Detachment Characteristics: Large retinal detachments, detachments involving the macula (macular retinal detachment), and detachments with multiple retinal breaks are associated with a higher risk of PVR.
- Posterior Vitreous Detachment (PVD): While PVD is a common cause of retinal tears, a complex or incomplete PVD can lead to increased vitreous traction on the retinal tear, predisposing to PVR.
- Previous Ocular Surgery: Multiple previous retinal detachment surgeries, cataract surgery, or vitreous surgery can increase the risk of PVR due to repeated trauma and inflammatory insults.
- Inflammatory Conditions: Ocular inflammation, such as uveitis, can predispose to PVR by creating a pro-inflammatory environment conducive to cellular proliferation.
- Trauma: Ocular trauma, especially penetrating injuries, can lead to vitreous hemorrhage and inflammation, increasing the risk of PVR.
- Age: While PVR can occur at any age, it is more common in older individuals, likely due to age-related changes in the vitreous and increased prevalence of other ocular conditions.
- Genetic Predisposition: Emerging research suggests a potential genetic component, with certain genetic variations possibly influencing an individual’s response to retinal injury and their likelihood of developing PVR.
Clinical Manifestations and Staging of PVR
The clinical presentation of PVR can vary widely, ranging from subtle visual disturbances to profound vision loss. The severity and progression are often described using a staging system.
Early Signs and Symptoms
In its early stages, PVR might not present with overt symptoms, or the symptoms may be attributed to the initial retinal detachment or the surgical procedure itself. However, as the proliferative process progresses, patients may experience:
- Increased Floaters: A sudden increase in the number or size of floaters can indicate bleeding or inflammatory activity within the vitreous.
- Flashes of Light (Photopsia): These can be a sign of ongoing retinal stimulation or traction.
- Visual Distortion: Straight lines may appear wavy or bent (metamorphopsia), a classic sign of retinal distortion due to underlying traction.
- Decreased Visual Acuity: As the macula becomes involved or significantly distorted, central vision loss occurs.
- Shadow or Veil in Vision: A dark shadow or curtain moving across the field of vision signifies further retinal detachment.
The Buckle-Siegel Classification System
The Buckle-Siegel classification is a widely used system to grade the severity of PVR. It categorizes PVR based on the presence and extent of preretinal membranes and retinal distortions.
- Stage A (Early PVR): This stage is characterized by subtle changes. It may include RPE proliferation in the posterior vitreous, pigment granules on the anterior hyaloid face, or minimal vitreous haze. There are no significant retinal distortions or detachments.
- Stage B (Moderate PVR): This stage involves the development of more distinct preretinal membranes. These membranes are typically located on the posterior surface of the retina and may begin to exert mild traction. Retinal folds or striae may be present, and the detachment may have spread.
- Stage C (Severe PVR): This is the most advanced stage. It is defined by the presence of significant preretinal membranes that cause substantial traction. This leads to marked retinal distortion, including puckering, bridging folds, and circumpapillary contraction. The retina may appear significantly pulled towards the vitreous. The Buckle-Siegel classification further subdivides Stage C into:
- C1: Mild contraction, retinal folds, less than one quadrant.
- C2: Moderate contraction, significant retinal folds, bridging folds involving more than one quadrant.
- C3: Severe contraction, significant puckering and tenting of the retina, often with significant visual compromise.
- Stage D (Total Retinal Detachment): In this stage, the entire retina is detached, and there are extensive contractile membranes that have essentially encased the retina.
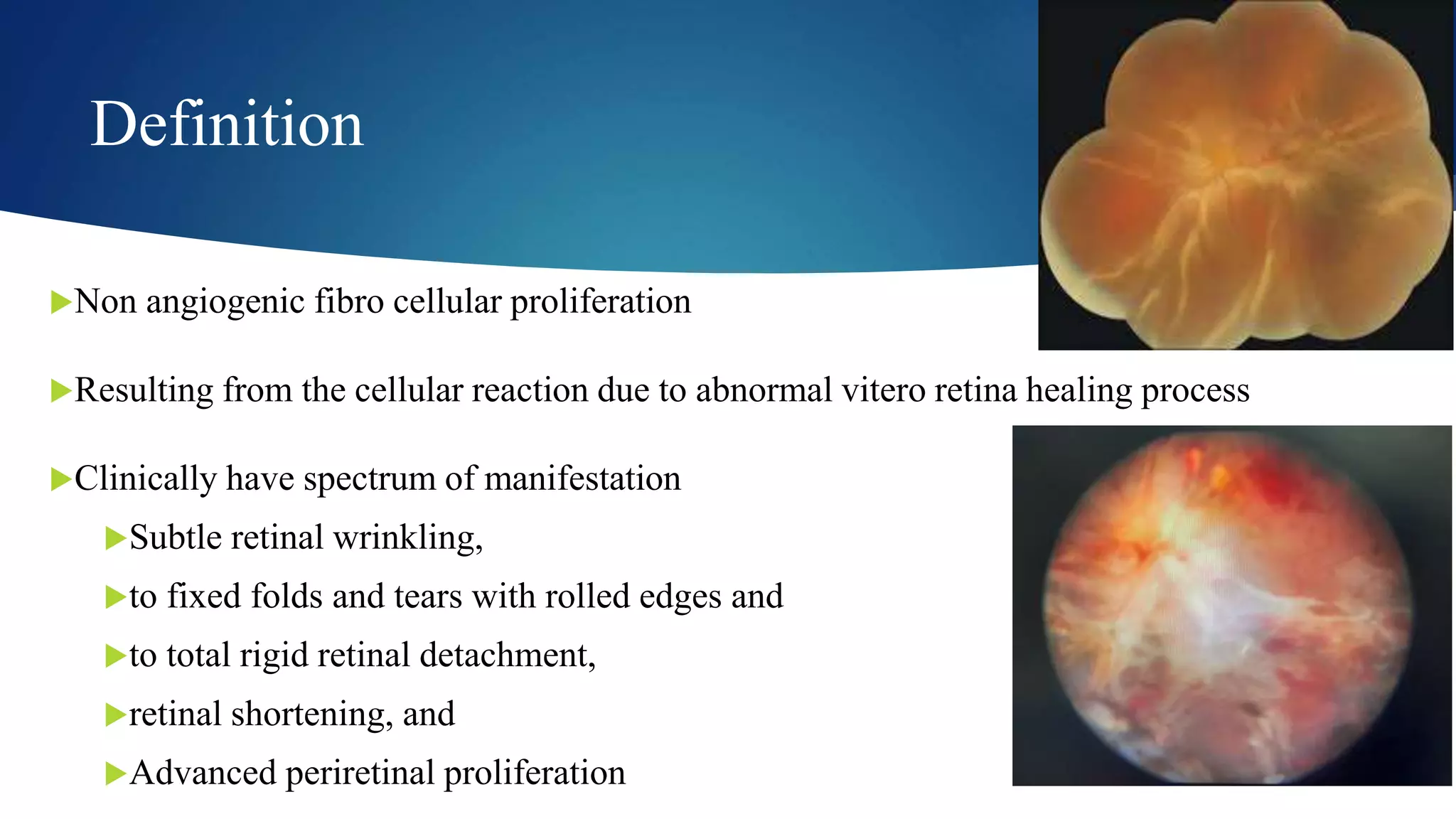
It’s important to note that PVR is a dynamic process, and a patient’s condition can progress through these stages if left untreated or if initial treatments are not fully successful.
Diagnostic Approaches for PVR
Diagnosing PVR requires a thorough ophthalmic examination, often supplemented by advanced imaging techniques.
Ophthalmic Examination
A comprehensive dilated fundus examination is the cornerstone of PVR diagnosis. The ophthalmologist will assess:
- Vitreous Cavity: Looking for vitreous haze, pigment clumps (tobacco dust), and membranes within the vitreous.
- Retinal Surface: Identifying retinal folds, striae, tractional bands, and the extent and pattern of retinal detachment.
- Retinal Pigment Epithelium (RPE): Observing for RPE proliferation and pigment migration.
- Presence of Retinal Breaks: Locating any new or old retinal breaks that may have contributed to the detachment.
Ancillary Diagnostic Tools
- Ultrasound Biomicroscopy (UBM) and B-scan Ultrasonography: These imaging modalities are valuable when the view of the retina is obscured by dense vitreous hemorrhage or opacities. They can help visualize preretinal membranes and assess the extent of retinal detachment.
- Optical Coherence Tomography (OCT): OCT provides high-resolution cross-sectional images of the retina. It is crucial for differentiating between rhegmatogenous retinal detachment, tractional retinal detachment, and exudative retinal detachment. OCT can clearly delineate the presence and thickness of preretinal membranes, their attachment to the retina, and the degree of retinal distortion. It is particularly useful in identifying subtle membranes or early signs of PVR that might be missed on clinical examination alone.
- Fundus Photography and Fluorescein Angiography (FA): While less critical for initial PVR diagnosis, fundus photography documents the extent of PVR. FA can sometimes reveal leakage from neovascularization or highlight areas of RPE compromise, although its role in PVR management is limited.
Management and Treatment Strategies for PVR
Managing PVR is one of the most challenging aspects of retinal surgery, often requiring complex and multi-step approaches. The goal is to relieve the traction exerted by the membranes and reattach the retina.
Surgical Interventions
The primary treatment for PVR is surgical. The choice of surgical technique depends on the severity and stage of PVR.
- Vitrectomy: This is the most common surgical procedure. It involves the removal of the vitreous gel, which contains the proliferative membranes.
- Membrane Peeling: During vitrectomy, surgeons meticulously attempt to peel or dissect the preretinal membranes from the surface of the retina. This is often a delicate and challenging part of the surgery, as the membranes can be firmly adherent to the retina, and aggressive peeling can lead to iatrogenic retinal tears.
- Fluid-Air Exchange and Gas Tamponade: After membrane removal, the retina is flattened, and an intraocular gas bubble or silicone oil is often injected to maintain retinal apposition and allow healing.
- Scleral Buckling: In certain cases, particularly when combined with vitrectomy, a scleral buckle may be used. This is an external surgical procedure that compresses the sclera, creating an indentation that supports the retina from the outside and can help flatten retinal folds caused by PVR.
- Intraocular Lens (IOL) Management: If an IOL is present, it may need to be removed or repositioned if it interferes with surgical access or visualization.
- Retinal Cryopexy or Laser Photocoagulation: These methods may be used to seal retinal breaks or to create chorioretinal adhesions around areas of PVR to prevent further progression.
Pharmacological and Adjunctive Therapies
While surgery is the mainstay, adjunctive therapies are being investigated and used to reduce the incidence and severity of PVR.
- Intraocular Corticosteroids: Steroids can reduce inflammation and inhibit fibroblast proliferation. They may be injected intraocularly during or after surgery.
- Anti-VEGF Agents: While primarily used for neovascularization, anti-VEGF agents might have a role in reducing inflammation and potentially limiting the proliferative process in some cases of PVR.
- Tissue Plasminogen Activator (tPA) and Heparin: In select cases of early PVR or vitreous hemorrhage associated with PVR, intraocular injections of tPA and heparin have been used to help liquefy vitreous membranes and blood, facilitating their removal during vitrectomy.
- Pharmacological Vitreolysis: Research is ongoing into agents that can chemically dissolve vitreous membranes.
- Postoperative Management: Close monitoring and management of intraocular pressure, patient positioning, and adherence to activity restrictions are critical for successful outcomes after surgery.
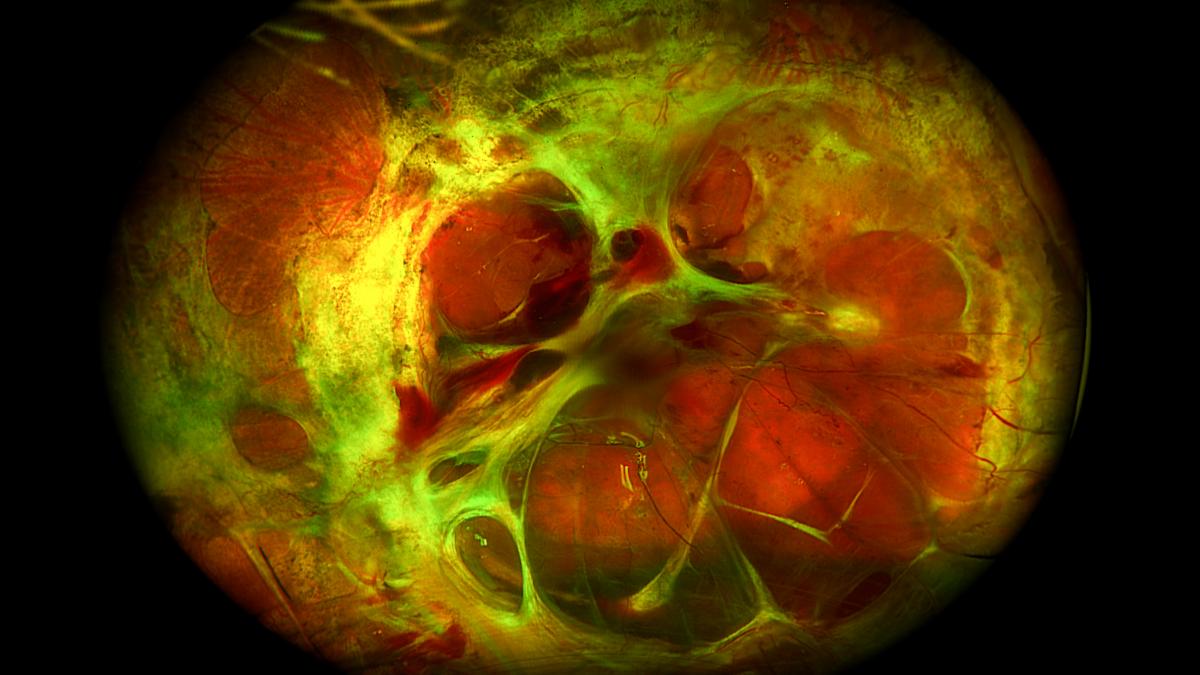
Prognosis and Recurrence
The prognosis for patients with PVR is variable and depends heavily on the extent of PVR at the time of initial treatment, the success of surgical intervention, and the presence of other ocular comorbidities. Despite aggressive surgical management, PVR has a significant recurrence rate. Multiple surgeries may be required in complex cases. The visual outcome can range from significant vision improvement to severe, irreversible vision loss, including blindness. Early detection, timely intervention, and advanced surgical techniques are paramount in optimizing outcomes for individuals affected by this challenging retinal condition.
