The fields of chemistry and technology are intertwined in ways that often go unnoticed by the casual observer. While chemistry might seem like a purely theoretical science confined to laboratories, its principles are fundamental to countless technological advancements that shape our modern world. From the development of advanced materials used in cutting-edge electronics to the sophisticated chemical processes powering renewable energy sources, chemistry provides the foundational understanding necessary for innovation. In this context, grasping core chemical concepts like extensive and intensive properties is not just an academic exercise, but a crucial stepping stone for anyone looking to understand or contribute to the technological frontier.

Understanding the Foundation: Defining Extensive and Intensive Properties
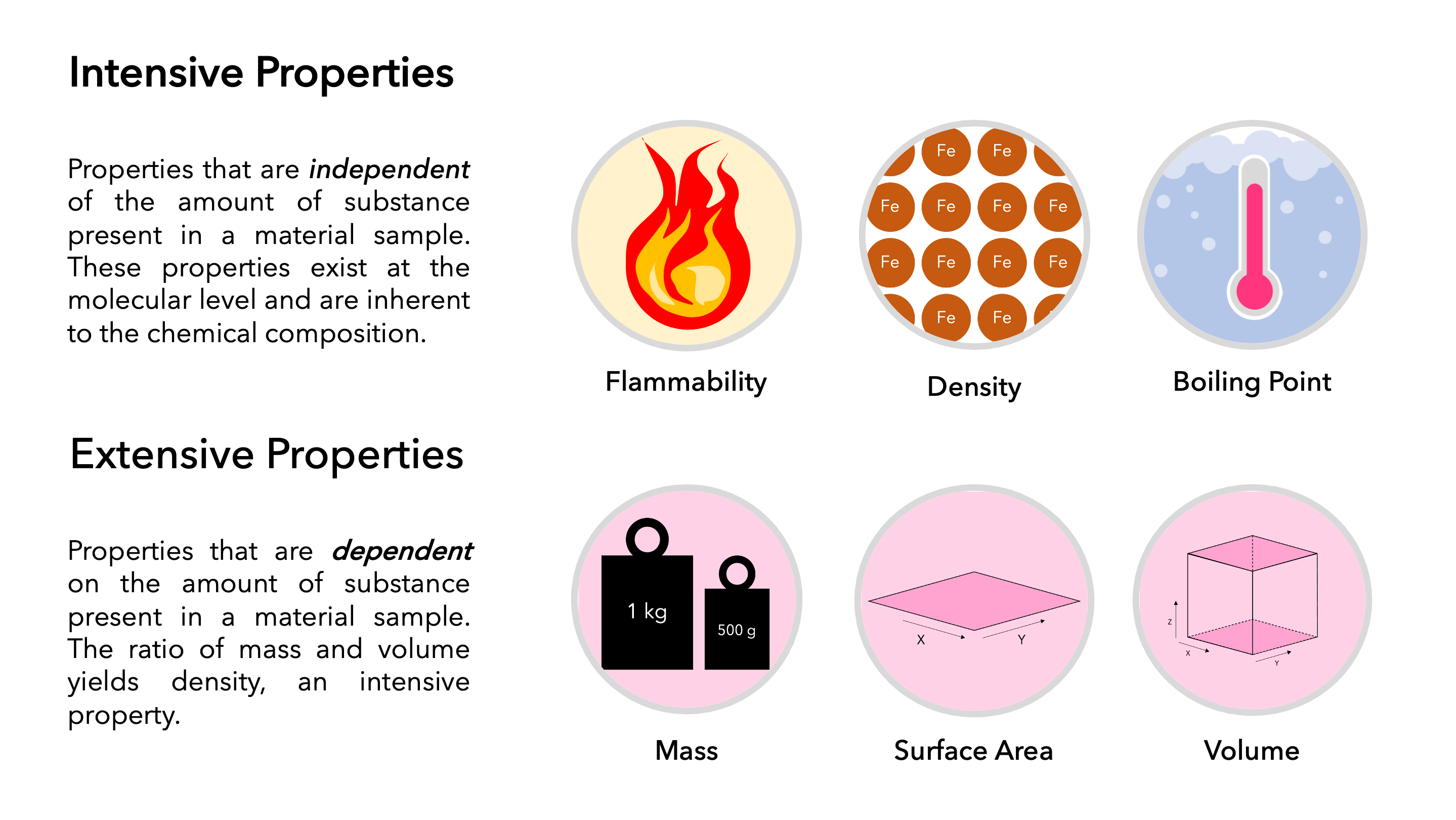
At the heart of chemical measurement and analysis lie the concepts of extensive and intensive properties. These two categories of physical characteristics of matter are distinguished by their dependence on the amount of substance present. Understanding this distinction is paramount for accurate scientific observation, experimental design, and the interpretation of data. It forms a bedrock principle that influences everything from basic laboratory procedures to the complex engineering of advanced technologies.
Extensive Properties: The Dependency on Substance Amount
An extensive property is a characteristic of a system that is directly proportional to the size or amount of matter within the system. In simpler terms, if you double the amount of a substance, you also double its extensive properties. These properties are additive; if you combine two systems, the extensive property of the combined system is the sum of the extensive properties of the individual systems.
For instance, consider mass. If you have 10 grams of water, its mass is 10 grams. If you take another 10 grams of water and combine them, you now have 20 grams of water, and the total mass is the sum of the initial masses. Similarly, volume is an extensive property. A liter of a substance occupies a specific amount of space. If you have two liters of the same substance, it occupies twice that space.
Other common extensive properties in chemistry include:
- Weight: This is directly related to mass and the gravitational pull. More mass means more weight.
- Energy (Internal Energy, Enthalpy, Entropy): These thermodynamic properties are related to the total energy contained within a system, which is dependent on the amount of substance. For example, the total heat content of a large body of water will be greater than that of a small cup of the same water.
- Number of Moles: This is a direct measure of the amount of substance and is inherently extensive.
The significance of extensive properties lies in their ability to quantify the “bulk” of a substance. In many technological applications, controlling or measuring the total quantity of a material is critical for process efficiency, product yield, and safety. For example, in the manufacturing of semiconductors, the precise control of the amount of precursor chemicals (measured by mass or volume) is vital for creating wafers with the desired properties. In the production of pharmaceuticals, the dosage of an active ingredient is an extensive property that directly impacts its efficacy and safety.
Intensive Properties: The Independence from Substance Amount
In contrast to extensive properties, intensive properties are those that do not depend on the amount of matter present in a system. Whether you have a small sample or a large quantity of a substance, its intensive properties remain the same. These properties are constant regardless of the size of the system and are therefore useful for identifying substances and understanding their fundamental characteristics.
A classic example of an intensive property is temperature. If you have a cup of boiling water and a bathtub full of boiling water, both are at 100°C (at standard atmospheric pressure). The amount of water doesn’t change its boiling point. Similarly, density is an intensive property. Density is defined as mass per unit volume (mass/volume). If you have a large block of iron, its density is the same as a small nail made of iron. While both mass and volume are extensive, their ratio (density) is intensive.
Other important intensive properties include:
- Melting Point and Boiling Point: These are specific temperatures at which a substance changes state and are characteristic of the substance itself, not the quantity.
- Color: The color of a substance is an intrinsic property that doesn’t change with the amount. A small speck of gold has the same golden hue as a large gold bar.
- Hardness: This refers to a material’s resistance to scratching or indentation and is an intrinsic characteristic.
- Concentration: While the total amount of solute and solvent are extensive properties, their ratio (concentration) is intensive. For example, a 1 molar solution of sodium chloride has the same concentration whether you have a liter or a milliliter of it.
- Viscosity: This measures a fluid’s resistance to flow and is independent of the volume of the fluid.
- Electrical Conductivity: The ability of a material to conduct electricity is an intrinsic property.
The utility of intensive properties in technology is immense. They serve as crucial identifiers and quality control metrics. For instance, the melting point of a metal alloy is a critical factor in its application in high-temperature environments. The viscosity of a lubricant determines its effectiveness in reducing friction. The color of a pigment is essential for aesthetic applications. In the realm of advanced materials, properties like electrical conductivity and thermal conductivity are intensive and dictate their suitability for specific electronic components or heat dissipation systems.
The Interplay and Application in Technological Advancement
The distinction between extensive and intensive properties is not merely academic; it has profound practical implications in various technological fields. Understanding how these properties behave allows engineers and scientists to predict the performance of materials, design efficient processes, and develop novel solutions.
Leveraging Intensive Properties for Material Characterization and Selection
Intensive properties are often the primary tools for characterizing and identifying substances. In material science and engineering, this is fundamental. When developing new alloys for aerospace applications, for instance, engineers will meticulously examine their intensive properties such as tensile strength, melting point, thermal expansion coefficient, and electrical conductivity. These properties are independent of the size of the sample tested and provide a reliable basis for comparison with existing materials and theoretical models.
Consider the development of advanced battery technologies. The specific energy density (an intensive property related to energy storage per unit mass or volume) is a key performance indicator. Similarly, the ionic conductivity of an electrolyte is an intensive property that dictates the rate at which ions can move, directly impacting charging and discharging speeds. When selecting materials for these applications, engineers rely on tables of intensive properties to pinpoint candidates that meet stringent performance requirements.
Furthermore, intensive properties are crucial for quality control in manufacturing. For example, in the production of high-purity silicon for microelectronics, parameters like resistivity (an intensive electrical property) are constantly monitored. Deviations from the expected resistivity indicate impurities or structural defects, allowing for immediate process adjustments. This ensures that the vast number of transistors fabricated on a silicon wafer will all function as intended.
Manipulating Extensive Properties for Scalable Processes and Product Design
While intensive properties define the intrinsic nature of a material, extensive properties are critical for scaling up production and designing products of specific sizes and capacities. The ability to control and predict how extensive properties change with quantity is essential for industrial processes.
In chemical manufacturing, reaction rates and yields are often influenced by the scale of the operation. While the reaction kinetics themselves might be governed by intensive properties like temperature and pressure, the overall throughput and efficiency of a large-scale chemical reactor depend on extensive properties like the total volume of reactants, the total surface area for heat exchange, and the total energy input. Chemical engineers design reactors based on these extensive parameters to ensure consistent product quality and efficient resource utilization.
In the design of consumer products, extensive properties play a direct role. The battery life of a smartphone is directly related to the total energy capacity (an extensive property) of its battery cells. The capacity of a storage container is an extensive property (volume). Even in seemingly simple products, the interplay of these properties is vital. For example, the weight of a portable electronic device (an extensive property) needs to be balanced with its desired functionality and battery life, which also involves extensive properties.
The field of nanotechnology exemplifies how understanding and controlling both types of properties is crucial. At the nanoscale, materials can exhibit drastically different properties compared to their bulk counterparts. While the fundamental intensive properties of the constituent atoms remain the same, their arrangement and the sheer number of atoms (an extensive property, albeit at a very small scale) can lead to unique emergent behaviors that are leveraged in advanced technologies.
Case Studies: Chemistry in Action in Tech and Innovation
The principles of extensive and intensive properties are not abstract concepts; they are actively applied in the development and refinement of technologies that are transforming our world.
Smart Materials and Sensors: Leveraging Intensive Properties for Detection
The development of smart materials and sensors is a prime example of how intensive properties are harnessed for technological innovation. These materials are designed to respond to specific stimuli, and their sensing capabilities often rely on changes in their intrinsic, intensive properties.
For instance, thermochromic materials change color (an intensive property) in response to temperature changes. This phenomenon is utilized in a wide range of applications, from mood rings and novelty items to more sophisticated uses like temperature indicators on packaging for sensitive goods or diagnostic tools in medical settings. The color change is a direct manifestation of the material’s chemical structure and its response to thermal energy, irrespective of the size of the material sample.
Chemoresistive sensors, widely used in gas detection and environmental monitoring, rely on the change in electrical resistance (an intensive property) of a sensing material when it interacts with specific chemical species. For example, metal oxide semiconductor sensors detect gases by a change in their conductivity upon exposure to the target analyte. The sensitivity and selectivity of these sensors are determined by the material’s intrinsic chemical and physical properties, not by the physical volume of the sensor itself.
In the field of biosensors, the immobilization of biological recognition elements (like enzymes or antibodies) onto a transducer surface allows for the detection of specific biomolecules. The signal generated by the transducer, often an electrical, optical, or electrochemical output, is a consequence of the interaction between the analyte and the recognition element, leading to a change in an intensive property of the transducer or the generation of a measurable signal proportional to the analyte concentration.
Sustainable Technologies: Optimizing Processes through Chemical Understanding
The drive towards sustainable technologies is heavily reliant on a deep understanding of chemical principles, including extensive and intensive properties.
In the development of catalysts for industrial processes, intensive properties are paramount. A catalyst’s efficiency, selectivity, and lifespan are all intensive properties. For example, the surface area to volume ratio (though involving both extensive and intensive concepts) of a porous catalyst is carefully engineered to maximize the number of active sites for reaction without compromising the overall structural integrity or introducing excessive mass. Understanding the kinetics of the catalyzed reaction, which is governed by intensive parameters like activation energy, allows for optimization of reaction conditions for maximum yield and minimal energy consumption.
The field of energy storage, particularly in battery technology and supercapacitors, relies heavily on manipulating both types of properties. The capacity of a battery (an extensive property) determines how much energy it can store, but its performance under various conditions is dictated by intensive properties such as the conductivity of the electrolyte, the electrochemical potential of the electrode materials, and the charge density of the active species. Engineers strive to maximize energy density (intensive) while ensuring safe and efficient operation across a range of scales and operating conditions, thus controlling extensive properties like battery pack size and weight.
Water purification and desalination technologies also benefit from this understanding. Reverse osmosis membranes, for instance, operate based on the intensive properties of the membrane material that allow water molecules to pass through while rejecting dissolved salts and other impurities. The efficiency of a large-scale desalination plant depends on the total surface area of these membranes (an extensive property) and the pressure applied, but the fundamental separation mechanism is governed by the intrinsic, intensive properties of the membrane itself.

Conclusion: The Enduring Relevance in a Technological Era
The concepts of extensive and intensive properties, seemingly basic chemical principles, are anything but trivial in the context of modern technology and innovation. They form the bedrock upon which we build our understanding of materials, design our processes, and engineer our future. From the microscopic world of semiconductors and nanoscale devices to the macroscopic scale of industrial manufacturing and sustainable energy systems, these classifications provide an essential framework for analysis, prediction, and control.
As technology continues to advance at an unprecedented pace, driven by breakthroughs in material science, artificial intelligence, and process engineering, the fundamental understanding of chemistry remains a critical enabler. The ability to distinguish between properties that scale with the amount of substance and those that remain constant provides the clarity needed to tackle complex challenges. Whether designing more efficient solar cells, developing novel drug delivery systems, or creating advanced computational hardware, the insightful application of these chemical concepts will continue to be a hallmark of true innovation. Therefore, a firm grasp of extensive and intensive properties is not just a requirement for chemists, but a fundamental asset for anyone aspiring to contribute to the technological advancements of tomorrow.
