Esophageal atresia (EA) is a complex congenital condition that presents a significant challenge in the field of pediatric medicine. While seemingly a purely biological anomaly, the journey of understanding, diagnosing, and treating EA is increasingly being shaped by advancements in technology and innovative approaches. This article explores esophageal atresia, not just as a medical diagnosis, but through the prism of how technological innovation is revolutionizing our comprehension and care for affected infants and their families.
The Genesis of Esophageal Atresia: Unraveling Developmental Pathways
Esophageal atresia is characterized by a malformation where the esophagus, the tube connecting the mouth to the stomach, does not form completely. Instead, it ends in a blind-ended pouch. This condition rarely occurs in isolation and is often associated with other birth defects, most notably tracheoesophageal fistula (TEF), where an abnormal connection forms between the trachea (windpipe) and the esophagus. Understanding the intricate developmental pathways that lead to these anomalies is a crucial area where technological innovation is making inroads.
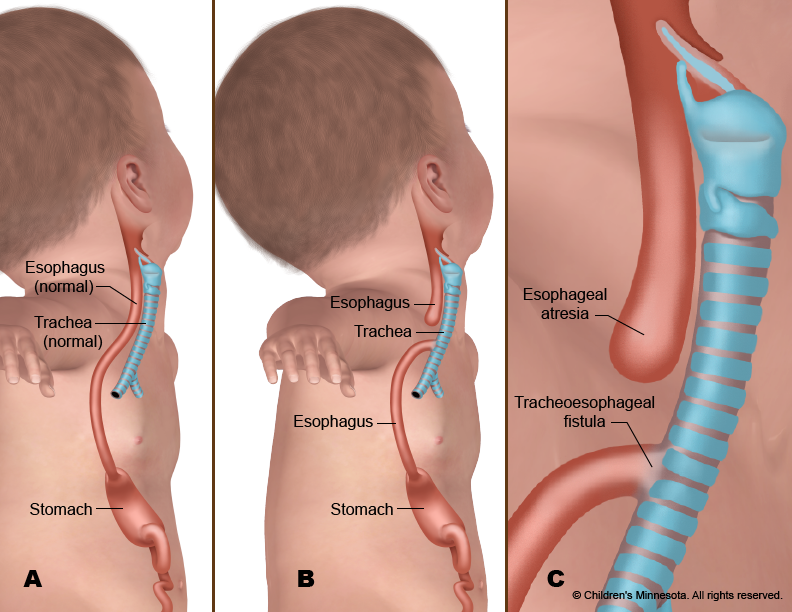
Epigenetic and Genetic Innovations in Developmental Biology
The origins of EA are rooted in complex developmental processes during the early stages of embryonic growth. Traditionally, understanding these origins relied on observational studies and inferential genetics. However, the advent of advanced genomic sequencing technologies has opened new avenues for research. Innovations in high-throughput sequencing allow researchers to analyze the entire genome of affected infants and their parents, identifying subtle genetic variations or mutations that may predispose individuals to EA.
Furthermore, the field of epigenetics, which studies heritable changes in gene expression that do not involve alterations to the underlying DNA sequence, is providing profound insights. Technologies like ChIP-sequencing (Chromatin Immunoprecipitation sequencing) and methyl-seq (DNA methylation sequencing) are enabling scientists to map epigenetic modifications that occur during esophageal development. By comparing these profiles in affected and unaffected embryos (often using animal models or induced pluripotent stem cells), researchers can pinpoint critical regulatory elements that, when disrupted, lead to EA. This innovation allows for a more granular understanding of the molecular choreography that goes awry during development, paving the way for potential future interventions.
Advanced Imaging Techniques for Embryonic Development
Visualizing the incredibly delicate processes of embryonic development in real-time has historically been a significant challenge. However, technological leaps in embryonic imaging are transforming this landscape. Confocal microscopy and light-sheet microscopy offer unprecedented resolution and speed, allowing researchers to observe the formation of the esophagus and trachea with remarkable clarity. These techniques, coupled with sophisticated computer-aided reconstruction and visualization software, enable the creation of three-dimensional models of developing embryos.
These innovative imaging tools are crucial for identifying the precise timing and nature of developmental disruptions that lead to EA. By observing how cells migrate, differentiate, and interact during the critical period of esophageal and tracheal formation, scientists can better understand the mechanical and molecular forces at play. This detailed visualization is not only fundamental for basic research but also has potential applications in developing prenatal diagnostic tools, allowing for earlier identification of potential issues.
Diagnostic Frontiers: Leveraging Technology for Early and Accurate Detection
The diagnosis of esophageal atresia, particularly in the neonatal period, requires prompt and accurate identification to initiate life-saving interventions. Technological advancements have significantly improved both prenatal and postnatal diagnostic capabilities.
Prenatal Screening and Fetal Ultrasound Innovations
While EA is not always detectable via standard prenatal ultrasounds, advancements in the field are improving detection rates. High-resolution 3D and 4D ultrasounds provide more detailed anatomical views of the fetus, allowing for better visualization of the esophageal tract. Sophisticated fetal echocardiography can also help identify associated cardiac anomalies, which are common in infants with EA.
Furthermore, ongoing research into fetal DNA analysis in maternal blood (non-invasive prenatal testing or NIPT) holds promise for detecting chromosomal abnormalities and genetic predispositions associated with certain birth defects. While EA itself might not be directly screened for via NIPT, identifying syndromes that frequently co-occur with EA can prompt closer fetal anomaly scanning, potentially leading to earlier suspicion and diagnosis. The innovation here lies in the non-invasive nature and the potential for broad genetic screening.
Postnatal Diagnostic Tools and Minimally Invasive Approaches
Once a baby is born, the immediate postnatal period is critical for diagnosis. While clinical signs and the inability to pass a feeding tube are often indicative, radiographic imaging remains the cornerstone of postnatal diagnosis.
Plain X-rays are fundamental, often revealing the blind-ended pouch of the esophagus and the presence of air in the stomach, which suggests a fistula. However, for more nuanced cases or to better delineate the anatomy of the fistula, contrast radiography is employed. This involves introducing a contrast agent into the esophagus to visualize its path and any abnormal connections. Innovations in low-osmolarity, water-soluble contrast agents have been developed to minimize potential harm to the delicate neonatal tissues.
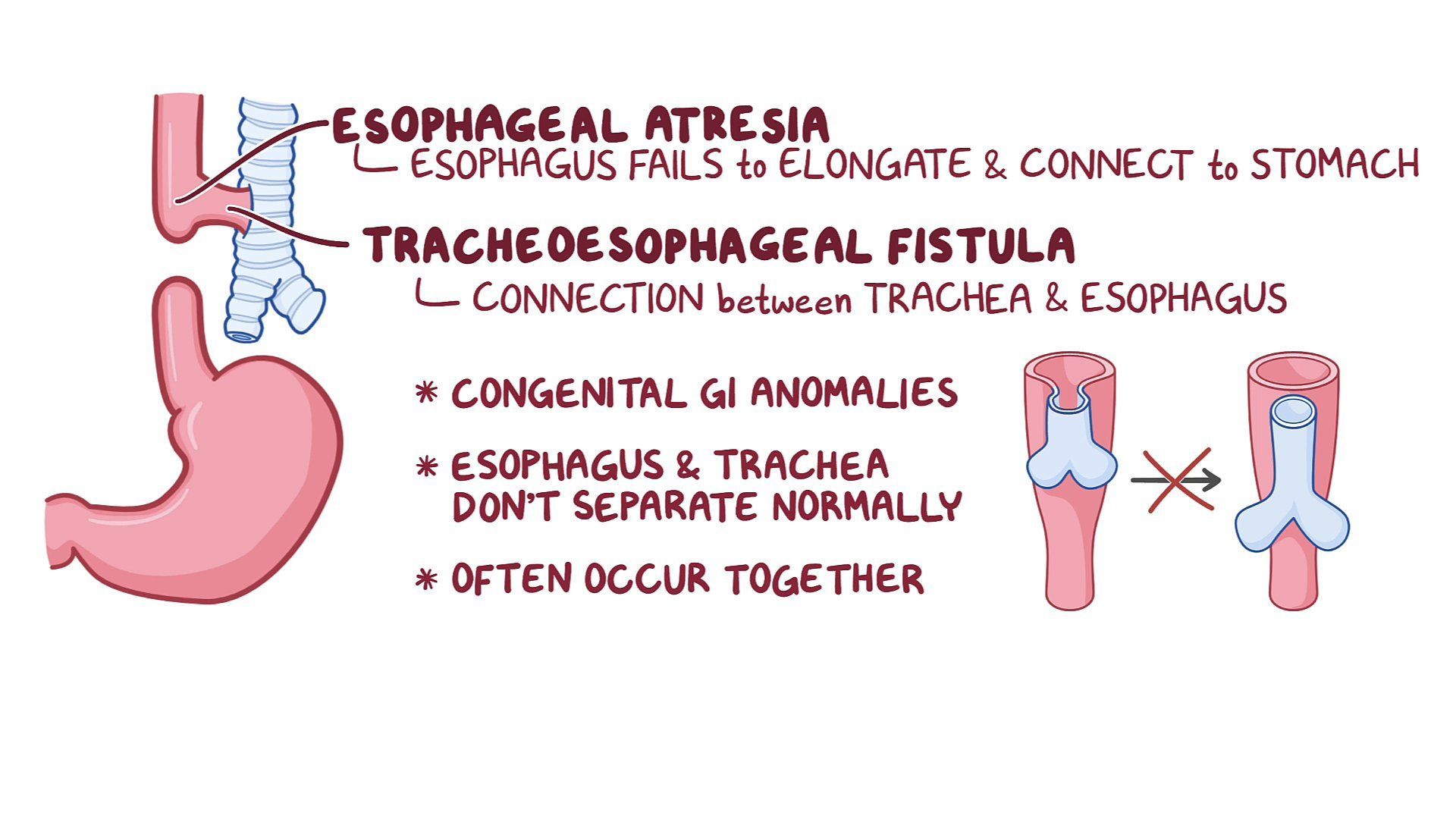
Beyond traditional radiography, the integration of minimally invasive diagnostic procedures is a significant technological advancement. In some cases, flexible bronchoscopy can be used to visualize the tracheobronchial tree and potentially identify the fistula opening from within the airway. Similarly, esophagoscopy, performed with very small, flexible endoscopes, allows for direct visualization of the esophageal pouch and confirmation of atresia. The innovation in this area lies in the miniaturization of endoscopic equipment, making these procedures safer and more feasible for fragile newborns.
Therapeutic Innovations: Precision Surgery and Future Possibilities
The primary treatment for esophageal atresia is surgical correction, aiming to connect the two ends of the esophagus and repair any fistula. Technological innovations are transforming both the surgical procedures themselves and the broader therapeutic landscape.
Minimally Invasive Surgical Techniques and Robotic Assistance
The evolution of surgical techniques has moved towards minimally invasive approaches to reduce trauma, shorten recovery times, and improve cosmetic outcomes. Thoracoscopic surgery, utilizing small incisions and specialized instruments guided by a camera, has become a viable option for repairing EA in certain cases. This approach leverages advancements in surgical optics and instrument design.
The ultimate frontier in surgical innovation is the potential application of robotic-assisted surgery. While still in its nascent stages for complex neonatal procedures like EA repair, robotic platforms offer enhanced dexterity, precision, and visualization. Imagine tiny robotic arms guided by a skilled surgeon, allowing for incredibly fine manipulations within the confined space of a newborn’s chest. This technological integration holds the promise of even more refined surgical outcomes.
Bioengineering and Regenerative Medicine for Esophageal Reconstruction
A significant long-term challenge for some infants with EA, particularly those with long-gap atresia where the distance between the esophageal ends is too great for primary connection, is the need for esophageal reconstruction. Traditional methods often involve using segments of the bowel, which can lead to complications.
This is where cutting-edge bioengineering and regenerative medicine are poised to revolutionize treatment. Innovations in tissue engineering are exploring the creation of artificial esophageal conduits using scaffolds seeded with the patient’s own cells. These scaffolds, often made from biocompatible polymers, provide a structure for cells to grow and differentiate, ultimately forming functional esophageal tissue.
3D bioprinting is an exciting technology within this realm, allowing for the precise deposition of cells and biomaterials to create complex tissue structures layer by layer. The vision is to one day print a personalized esophageal graft that perfectly matches the patient’s needs, eliminating the risks associated with using intestinal segments. Furthermore, research into stem cell therapy and growth factor delivery aims to stimulate the body’s own regenerative capabilities to promote the growth and repair of esophageal tissue. These innovations represent a paradigm shift from purely reconstructive surgery to true regenerative therapies, offering hope for a more complete and natural restoration of esophageal function.
The Role of Data Science and AI in Managing Esophageal Atresia
Beyond direct diagnosis and treatment, data science and artificial intelligence (AI) are playing an increasingly important, albeit often behind-the-scenes, role in improving the care of infants with esophageal atresia.
Predictive Analytics for Early Intervention and Outcome Prediction
The vast amounts of clinical data generated from the diagnosis, treatment, and follow-up of infants with EA can be a goldmine for innovation. Machine learning algorithms can be trained on this data to identify patterns and predict potential complications. For instance, AI models could potentially predict which infants are at higher risk for specific post-operative issues like anastomotic leaks or strictures, allowing for proactive monitoring and early intervention.
Furthermore, AI can be used to optimize treatment pathways. By analyzing the outcomes of different surgical approaches and post-operative care protocols across large patient populations, AI can help identify the most effective strategies for specific patient profiles. This data-driven approach moves healthcare towards more personalized medicine, where treatment is tailored to the individual’s unique characteristics and predicted response.

Innovations in Remote Monitoring and Telehealth
The long-term follow-up of children with EA often requires ongoing monitoring for issues such as feeding difficulties, respiratory problems, and growth. Telehealth platforms and wearable sensor technology are emerging as innovative tools to facilitate this.
Imagine parents being able to remotely monitor their child’s vital signs, feeding patterns, or even specific physiological markers through connected devices. This data can be transmitted securely to healthcare providers, allowing for early detection of subtle changes that might otherwise go unnoticed. AI algorithms can then analyze this continuous stream of data, flagging any anomalies for immediate attention. This innovation extends the reach of specialized care beyond the hospital walls, improving convenience for families and potentially leading to better long-term health outcomes for children born with esophageal atresia.
In conclusion, while esophageal atresia is a challenging congenital condition, the continuous wave of technological innovation is profoundly impacting every aspect of its management. From unraveling its developmental origins with advanced genetic and imaging tools, to enhancing diagnostic accuracy with sophisticated medical imaging and minimally invasive procedures, and finally to revolutionizing therapeutic possibilities with bioengineering, regenerative medicine, and the burgeoning power of AI and data science, the future of care for infants with EA is increasingly bright. These advancements underscore the transformative power of technology and innovation in overcoming complex medical hurdles and improving the lives of those affected.
