Cytochrome c is a vital protein that plays a crucial role in the fundamental process of cellular respiration, the mechanism by which living organisms convert nutrients into energy. It is a heme protein, meaning it contains a heme group, a porphyrin ring with an iron atom at its center. This iron atom is key to cytochrome c’s function, as it can readily accept and donate electrons, facilitating the transfer of energy within the cell. While its primary and most well-known role is in ATP production, cytochrome c also has a more complex and, in some contexts, sinister dual nature, being a critical player in programmed cell death, or apoptosis. Understanding cytochrome c is therefore essential for grasping the intricate biochemical machinery that sustains life and orchestrates cellular demise.

The Electron Transport Chain: Cytochrome c’s Energetic Role
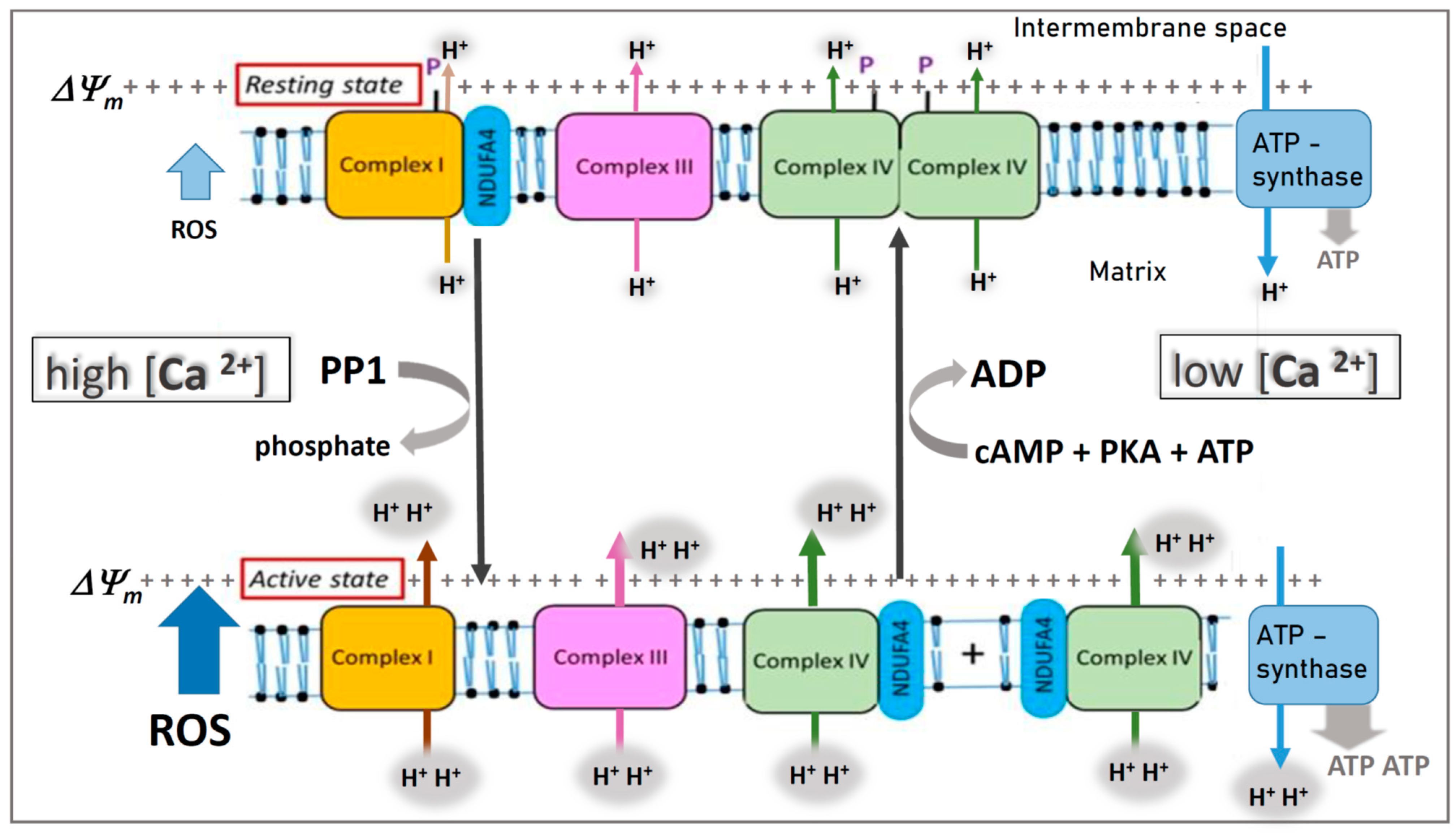
The primary theatre of action for cytochrome c is the inner mitochondrial membrane, where it acts as a mobile electron carrier within the electron transport chain (ETC). This multi-protein complex is the final stage of aerobic respiration, responsible for generating the vast majority of the cell’s adenosine triphosphate (ATP), the universal energy currency.
Structure and Function of Cytochrome c in the ETC
Cytochrome c itself is a small, soluble protein, typically around 12-13 kilodaltons in size. Its structure is characterized by a conserved core with a heme prosthetic group covalently linked to the protein scaffold. This heme group, specifically the iron atom within it, is the site of electron transfer. In its reduced state, the iron is Fe²⁺, and in its oxidized state, it is Fe³⁺.
The ETC is comprised of several large protein complexes (Complexes I-IV) embedded in the inner mitochondrial membrane, along with mobile carriers like ubiquinone (Coenzyme Q) and cytochrome c. The flow of electrons through these complexes is a stepwise process, with each step releasing a small amount of energy. This energy is harnessed to pump protons (H⁺ ions) from the mitochondrial matrix to the intermembrane space, creating an electrochemical gradient.
Cytochrome c’s specific role is to shuttle electrons from Complex III (also known as the cytochrome bc₁ complex or Complex III) to Complex IV (cytochrome c oxidase). It is located in the intermembrane space and associates transiently with both complexes. From Complex III, cytochrome c receives electrons that have been passed along from ubiquinone. It then diffuses across the intermembrane space to Complex IV, where it donates these electrons to the waiting copper centers.
The Proton Gradient and ATP Synthesis
The accumulation of protons in the intermembrane space creates a substantial proton motive force. This force drives protons back into the mitochondrial matrix through a specialized enzyme complex called ATP synthase. As protons flow through ATP synthase, its rotational mechanism catalyzes the phosphorylation of adenosine diphosphate (ADP) to ATP, effectively converting the potential energy stored in the proton gradient into chemical energy. Cytochrome c’s efficient and continuous electron transfer is thus directly linked to the cell’s capacity to produce ATP and sustain its metabolic activities. Without the effective functioning of cytochrome c, the ETC would grind to a halt, ATP production would plummet, and cellular functions would be severely compromised.
Apoptosis: Cytochrome c’s Role in Programmed Cell Death
While its role in energy production is paramount, cytochrome c exhibits a dramatic Jekyll and Hyde transformation when it comes to programmed cell death, or apoptosis. Under normal, healthy conditions, cytochrome c is firmly localized within the mitochondrial intermembrane space, diligently performing its duties in the ETC. However, under specific stress signals, cytochrome c can be released into the cytosol, initiating a cascade of events that leads to the cell’s controlled self-destruction.
Release from Mitochondria: A Critical Checkpoint

The release of cytochrome c from mitochondria into the cytosol is a pivotal and often irreversible step in the intrinsic pathway of apoptosis. This pathway is activated by internal cellular damage or stress, such as DNA damage, oxidative stress, or the absence of survival signals. These insults can trigger the assembly of a multi-protein complex within the mitochondrial outer membrane called the “apoptosome.”
The formation of the apoptosome is initiated by the release of cytochrome c. Once in the cytosol, cytochrome c binds to a protein called Apaf-1 (apoptotic protease activating factor-1). This binding event causes a conformational change in Apaf-1, allowing it to oligomerize and recruit other Apaf-1 molecules, forming a wheel-like structure. This assembled structure is the apoptosome.
Activating the Caspase Cascade
The primary function of the apoptosome is to recruit and activate initiator caspases, specifically caspase-9. Caspases are a family of cysteine proteases that are essential executioners of apoptosis. They are synthesized as inactive zymogens and are activated through proteolytic cleavage. Within the apoptosome, multiple molecules of pro-caspase-9 are brought into close proximity, facilitating their auto-activation through self-cleavage.
Once activated, caspase-9 acts as an initiator caspase. It then proceeds to cleave and activate effector caspases, such as caspase-3, caspase-6, and caspase-7. These effector caspases are the workhorses of apoptosis, systematically dismantling cellular components. They cleave a wide array of cellular substrates, including structural proteins, DNA repair enzymes, and regulatory proteins, leading to the characteristic morphological changes associated with apoptosis, such as cell shrinkage, membrane blebbing, and the formation of apoptotic bodies. The controlled breakdown of cellular machinery ensures that the dying cell is tidily packaged and removed by phagocytic cells, preventing the release of cellular contents and subsequent inflammation.
Clinical Significance and Research Implications
The dual nature of cytochrome c, its indispensability for life and its critical role in cell death, makes it a molecule of immense clinical and research significance. Dysregulation of cytochrome c’s function or release is implicated in a variety of diseases, and its study offers potential therapeutic avenues.
Cytochrome c in Disease Pathogenesis
- Cancer: While cancer is characterized by uncontrolled cell proliferation, paradoxically, the induction of apoptosis is a key strategy in cancer therapy. Many chemotherapeutic drugs and radiation therapies work by inducing DNA damage, which can lead to the release of cytochrome c and subsequent apoptosis of cancer cells. Resistance to apoptosis, often involving defects in cytochrome c release or caspase activation, is a major mechanism by which cancer cells evade treatment. Understanding these pathways is crucial for developing more effective anti-cancer strategies.
- Neurodegenerative Diseases: Conditions like Alzheimer’s, Parkinson’s, and Huntington’s disease are characterized by the progressive loss of neurons. While the exact mechanisms are complex, excessive apoptosis is a contributing factor to this neuronal loss. Aberrant signaling pathways that lead to premature or excessive release of cytochrome c from neuronal mitochondria are being investigated as potential drivers of these diseases.
- Ischemic Heart Disease: During a heart attack, a lack of oxygen (ischemia) triggers a cascade of events that can lead to cell death. The release of cytochrome c from cardiac mitochondria contributes to the damage sustained by heart muscle cells. Targeting cytochrome c release or its downstream effects is a focus of research aimed at reducing the extent of damage following ischemic events.
- Inflammatory Diseases: While apoptosis is a controlled process, dysregulation can also impact inflammation. In some contexts, excessive or uncontrolled release of cytochrome c can contribute to inflammatory responses.
Therapeutic Potential and Research Applications
The critical role of cytochrome c in both life and death has made it a focal point for therapeutic development and basic research:
- Apoptosis Induction: For diseases where excessive cell survival is problematic (e.g., cancer), strategies aim to enhance cytochrome c release and downstream caspase activation. This could involve developing small molecules that mimic or enhance the signals that trigger apoptosis or by inhibiting proteins that suppress it.
- Apoptosis Inhibition: Conversely, in conditions where excessive cell death is detrimental (e.g., neurodegenerative diseases, stroke), researchers are exploring ways to prevent or reduce cytochrome c release. This might involve stabilizing mitochondrial membranes or developing inhibitors of the apoptosome or caspases.
- Biomarker: The presence of cytochrome c in the cytosol can serve as a biomarker for apoptosis. Its detection in the bloodstream or other bodily fluids could indicate cellular damage or disease progression.
- Mitochondrial Health: Cytochrome c’s central role in mitochondrial function makes it an indicator of overall mitochondrial health. Research into its structure, function, and regulation provides insights into broader mitochondrial biology, which is fundamental to many cellular processes.
In conclusion, cytochrome c, a seemingly simple heme protein, is a cornerstone of cellular life and a critical regulator of cell fate. Its elegant dance within the electron transport chain fuels our existence, while its potential release signals the controlled dismantling of the cell. The ongoing exploration of this remarkable molecule continues to unveil its complexities, promising new insights into disease mechanisms and novel therapeutic interventions.
