In the advanced world of drone technology, where precision flight, extended endurance, and rapid response are paramount, the underlying power source—the battery—is often the unsung hero. While pilots focus on flight parameters and camera settings, the intricate chemical dance within a drone battery dictates its very capabilities. At the heart of this dance lies a fundamental chemical concept: the ionic compound. Understanding what an ionic compound is, and more importantly, how it functions within drone accessories like batteries, provides crucial insight into optimizing performance, extending lifespan, and appreciating the marvels of modern electrochemical energy storage.

The Fundamental Chemistry of Power: Ionic Compounds in Drone Batteries
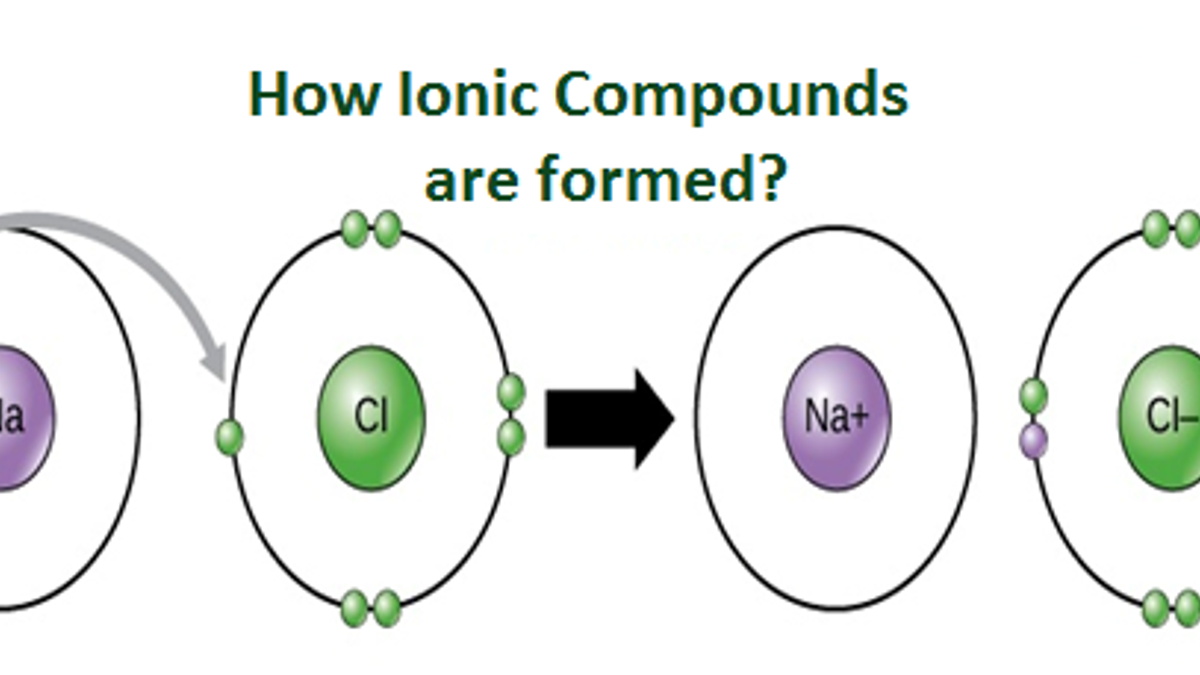
An ionic compound is a chemical compound formed by electrostatic attraction between positively charged ions (cations) and negatively charged ions (anions). These ions are formed when atoms either gain or lose electrons to achieve a stable electron configuration, typically resembling that of a noble gas. The transfer of electrons creates atoms with an electrical charge, and the strong attractive forces between these oppositely charged ions constitute an ionic bond. Unlike covalent compounds, where electrons are shared, ionic compounds are characterized by this complete transfer, leading to a rigid, crystalline lattice structure in their solid state.
In the context of drone batteries, particularly the ubiquitous lithium-ion (Li-ion) batteries, ionic compounds are not merely theoretical constructs; they are the active ingredients and essential facilitators of energy storage and release. The very movement of charge that powers a drone is fundamentally an ionic process.
Electrochemistry Explained: A Brief Overview
Electrochemistry is the branch of chemistry that deals with the relationship between electricity and chemical reactions. In a battery, chemical energy is converted into electrical energy through redox (reduction-oxidation) reactions, where electrons are transferred from one substance to another. Crucially, for electrons to flow through an external circuit, there must be a corresponding movement of ions within the battery’s internal structure to maintain charge neutrality.
During discharge, lithium ions (Li+) migrate from the anode (negative electrode) through an electrolyte to the cathode (positive electrode), while electrons flow simultaneously through the external circuit to power the drone’s motors, sensors, and flight controller. During charging, this process is reversed, with lithium ions moving back to the anode. The electrolyte, a critical component of any battery, is often an ionic compound or a solution containing ionic compounds, specifically designed to allow rapid ion transport while preventing electron flow.
Why Ionic Bonds Matter for Energy Storage
The strength and nature of ionic bonds are directly relevant to the performance and safety of drone batteries. The ability of certain elements, particularly lithium, to readily form stable ions (Li+) by losing an electron makes them ideal for energy storage. These ions can then be reversibly intercalated (inserted) into and deintercalated (extracted) from the electrode materials, creating a stable and repeatable cycle of charge and discharge.
Furthermore, the solid-state structures of the electrode materials themselves, such as lithium cobalt oxide (LiCoO2) or lithium iron phosphate (LiFePO4) at the cathode, are often complex ionic compounds. Their crystal lattices provide specific sites for the reversible storage of lithium ions. The arrangement and stability of these ionic structures dictate the battery’s energy density, power output, and cycle life. A deeper understanding of these ionic interactions allows for the development of more efficient and durable battery chemistries, directly translating into longer flight times and safer drone operation.
Lithium-Ion Batteries: The Core of Modern Drone Performance
Lithium-ion batteries have become the gold standard for powering drones due to their high energy density, low self-discharge rate, and relatively long cycle life. These characteristics are intrinsically linked to the behavior of lithium ions and the ionic compounds within their structure.
The Role of Electrolytes: Facilitating Ion Movement
The electrolyte is arguably the most critical component for ionic compound functionality within a Li-ion battery. It’s the medium through which lithium ions shuttle between the anode and cathode. While the term “electrolyte” might sound generic, in Li-ion batteries, it typically consists of a lithium salt (an ionic compound, e.g., LiPF6, lithium hexafluorophosphate) dissolved in an organic solvent mixture.
This lithium salt dissociates into Li+ cations and complex anions (e.g., PF6-), providing the necessary mobile ions. The organic solvents provide a suitable environment for these ions to move freely without reacting with the electrodes. The quality of the electrolyte—its conductivity, stability across a wide temperature range, and compatibility with electrode materials—directly impacts the battery’s power delivery, efficiency, and safety. A poorly formulated electrolyte can lead to sluggish ion movement, reduced power output, or even dangerous side reactions.
Anode and Cathode Materials: Ionic Structures at Play
The electrodes of a Li-ion battery are sophisticated materials, often complex ionic compounds or structures that readily accommodate ions.

-
Cathode (Positive Electrode): Common cathode materials like Lithium Cobalt Oxide (LiCoO2), Lithium Manganese Oxide (LiMn2O4), Lithium Nickel Manganese Cobalt Oxide (NMC), and Lithium Iron Phosphate (LiFePO4) are all examples of layered or spinel ionic compounds. They are structured to have “intercalation sites” where lithium ions can be reversibly stored. The specific arrangement of metal cations (Co, Mn, Ni, Fe) and oxide anions (O2-) creates an ionic framework that stabilizes the lithium ions during charging and discharging. The choice of cathode material profoundly influences the battery’s voltage, capacity, and thermal stability—all critical factors for drone performance and safety.
-
Anode (Negative Electrode): Traditionally, graphite is used for the anode. While graphite itself is a covalent network, lithium ions intercalate into its layered structure. During charging, lithium ions migrate from the cathode and insert themselves between the graphene layers of the graphite anode, forming a lithium-intercalated graphite compound. This process is highly reversible, allowing for thousands of charge/discharge cycles. The ability of graphite to accommodate lithium ions in this way is a testament to the finely tuned electrochemical processes enabled by these ionic interactions.
Beyond Lithium-Ion: Exploring Future Battery Chemistries
While Li-ion batteries dominate the drone market today, research continues into next-generation battery technologies. Many of these advancements still heavily rely on or aim to improve upon the principles of ionic compound behavior.
Solid-State Batteries: A New Frontier for Ionic Conduction
Solid-state batteries are a prime example of future technology seeking to leverage and enhance ionic compound properties. Instead of a liquid or gel electrolyte, these batteries use a solid electrolyte. These solid electrolytes are typically advanced inorganic ionic compounds (e.g., lithium garnets, sulfides, or oxides) engineered to exhibit high ionic conductivity—meaning lithium ions can move through them almost as freely as in a liquid electrolyte, but without the flammability risks associated with organic solvents.
The promise of solid-state batteries for drones includes higher energy density (potentially leading to significantly longer flight times), improved safety (no flammable liquid electrolyte), and a wider operating temperature range. The challenge lies in synthesizing solid electrolytes with sufficient ionic conductivity and ensuring good interfacial contact between the solid electrolyte and the solid electrodes, all while maintaining mechanical stability. The development hinges entirely on optimizing the ionic structure and properties of these novel solid compounds.
Other Emerging Technologies and Ionic Principles
Other emerging battery chemistries, such as lithium-sulfur (Li-S) and lithium-air (Li-air) batteries, also involve complex ionic reactions and the formation of various ionic compounds during their operation. Li-S batteries, for instance, form different lithium polysulfide ionic compounds as intermediates during discharge. Understanding and controlling the behavior of these ionic species is key to overcoming challenges like shuttle effects and poor cycle life. Even in these advanced concepts, the principles governing ionic compound formation, stability, and mobility remain central to achieving practical, high-performance power sources for future drone applications.
Optimizing Battery Life and Performance Through Ionic Understanding
For drone operators, a practical understanding of battery chemistry, specifically the role of ionic compounds, can translate into better care and extended performance of expensive drone battery accessories.
Factors Affecting Ion Mobility and Battery Degradation
Several factors can impede the efficient movement of ions and degrade the ionic compounds within a battery:
- Temperature Extremes: Both very high and very low temperatures can adversely affect the electrolyte’s ionic conductivity. Cold temperatures slow down ion movement, reducing power output and capacity. High temperatures can accelerate unwanted side reactions, leading to electrolyte decomposition and electrode degradation.
- Overcharging/Over-discharging: Pushing a Li-ion battery beyond its recommended voltage limits (overcharging) or draining it too low (over-discharging) can cause irreversible structural damage to the electrode materials’ ionic lattices and degrade the electrolyte. This leads to reduced capacity and internal resistance increases.
- High Charge/Discharge Rates: Repeatedly subjecting batteries to extremely high charge or discharge currents can induce mechanical stress on the electrode materials, leading to particle cracking and loss of active ionic material. It can also accelerate the formation of unwanted solid electrolyte interphase (SEI) layers, which impede ion movement.
Best Practices for Maintaining Drone Batteries
Armed with this knowledge, drone pilots can implement best practices:
- Store at Recommended Charge: Most Li-ion batteries perform best when stored at around 30-50% charge, not fully charged or fully depleted. This minimizes stress on the ionic structures and slows down degradation.
- Avoid Temperature Extremes: Operate and store drone batteries within their specified temperature ranges. Use insulated battery bags in cold weather and avoid leaving batteries in direct sunlight or hot vehicles.
- Use Compatible Chargers: Always use chargers designed for your specific battery type and chemistry. Proper charging protocols ensure that the ionic compounds are not overstressed.
- Monitor Battery Health: Regularly check internal resistance (IR) readings. An increasing IR indicates degradation in the internal ionic pathways and electrode materials.
- Balance Cells: For multi-cell LiPo batteries, ensure cells are balanced. Imbalances can lead to some cells being over-discharged or overcharged, accelerating localized ionic degradation.
In conclusion, while the average drone enthusiast may not delve into the molecular structure of lithium salts, the reliable flight of their UAV is a direct consequence of the precise and controlled behavior of ionic compounds. From the electrolytes facilitating ion transport to the electrode materials housing these charge carriers, ionic chemistry underpins every aspect of drone battery performance. A deeper appreciation for these fundamental chemical principles empowers users to better manage their drone accessories and pushes innovators to develop ever more advanced power solutions for the skies of tomorrow.
