The Fundamental Forces at Play
Filtration, at its core, is the separation of solid particles from a fluid (liquid or gas) by passing the fluid through a porous medium that retains the solids. While seemingly straightforward, the driving force behind this separation is a nuanced interplay of physical principles. Understanding these fundamental forces is crucial for optimizing membrane performance across a vast spectrum of applications, from water purification and gas separation to pharmaceutical processing and medical devices. At the most basic level, filtration is propelled by a pressure difference or a concentration gradient, compelling the fluid and its dissolved or suspended components to move through the membrane’s intricate pore structure.
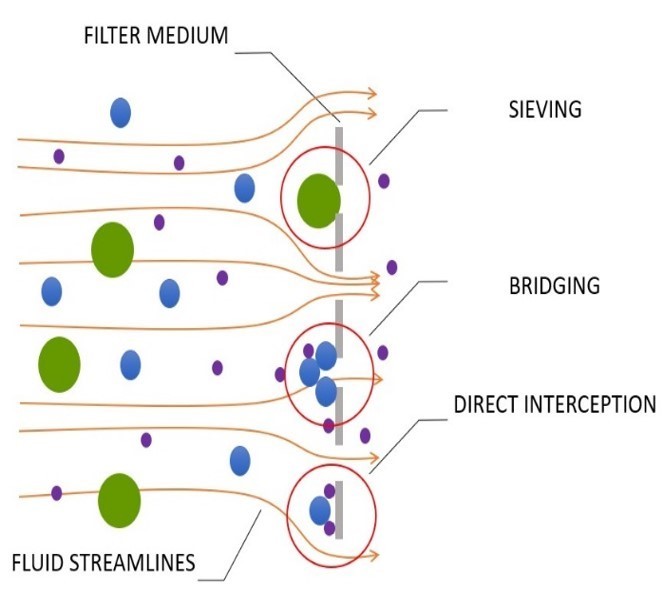
Pressure-Driven Filtration: The Dominant Engine
The most ubiquitous driving force in membrane filtration is pressure difference. This can manifest in several forms:
Transmembrane Pressure (TMP)
Transmembrane pressure is the difference in hydrostatic pressure across the membrane. In most filtration scenarios, a higher pressure is applied to the feed side (retentate) of the membrane than to the permeate side. This pressure gradient forces the fluid and smaller dissolved molecules or ions to pass through the membrane pores, while larger particles or molecules are retained. TMP is a critical parameter directly influencing flux, the rate at which permeate passes through the membrane per unit area. Increasing TMP generally leads to higher flux, but it also exacerbates fouling—the accumulation of rejected material on the membrane surface—which can significantly reduce performance and membrane lifespan.
Applied Pressure Scenarios
- Dead-End Filtration: In this configuration, the feed fluid flows perpendicular to the membrane surface. All fluid either passes through the membrane as permeate or is retained as retentate. As filtration progresses, the concentration of rejected species increases at the membrane surface, leading to cake formation and a rapid decline in flux.
- Cross-Flow Filtration (Tangential Flow Filtration): Here, the feed fluid flows parallel to the membrane surface, with a small portion passing through as permeate. The bulk of the fluid, along with the rejected particles, is recirculated. This tangential flow creates a shear force along the membrane surface that helps to sweep away accumulated material, minimizing cake layer formation and mitigating fouling. This allows for higher operating fluxes and longer operational cycles compared to dead-end filtration, especially for processing feeds with high solids content.
- Vacuum Filtration: In this case, a negative pressure (vacuum) is applied to the permeate side of the membrane, while the feed side is at atmospheric pressure. This creates a pressure difference that drives the filtration process. Vacuum filtration is often used when dealing with heat-sensitive materials or when high pressures are not feasible.
Factors Influencing Pressure-Driven Flux
The flux achieved in pressure-driven filtration is governed by Darcy’s Law for flow through porous media, which can be adapted for membranes:
$J = frac{K Delta P}{mu L}$
Where:
- $J$ is the flux (volume per unit area per unit time)
- $K$ is the permeability of the membrane (dependent on pore size and tortuosity)
- $Delta P$ is the transmembrane pressure
- $mu$ is the viscosity of the fluid
- $L$ is the effective thickness of the membrane
This equation highlights that higher pressure ($ Delta P $) and higher membrane permeability ($K$) lead to increased flux. Conversely, increased fluid viscosity ($ mu $) and membrane thickness ($L$) reduce flux. Fouling acts as an effective increase in membrane thickness and a reduction in effective pore size, thus decreasing flux.
Concentration-Driven Filtration: Exploiting Chemical Potential
While pressure is the workhorse, concentration gradients also play a significant role, particularly in certain advanced membrane processes. This driving force is rooted in the concept of chemical potential, which represents the potential energy of a substance due to its concentration and its environment. Substances naturally move from regions of high chemical potential (high concentration) to regions of low chemical potential (low concentration).

Osmosis and Reverse Osmosis (RO)
Osmosis is a special case of diffusion where a solvent (typically water) moves across a semipermeable membrane from a region of higher solvent concentration (lower solute concentration) to a region of lower solvent concentration (higher solute concentration). This movement is driven by the osmotic pressure of the more concentrated solution.
Reverse Osmosis (RO) harnesses this principle but with a twist. To force solvent from a more concentrated solution to a less concentrated one (or to achieve purification), an applied pressure exceeding the osmotic pressure is necessary. The driving force in RO is effectively the net pressure difference that overcomes the osmotic pressure. The permeate is essentially purified solvent, leaving behind the concentrated solutes. This is a prime example where both pressure and concentration (osmotic pressure) are intrinsically linked.
Diffusion Dialysis
In diffusion dialysis, a concentration gradient is the sole driving force. This process is used for separating components based on their differing diffusion rates across a membrane. Typically, it’s employed for acid recovery from metal salt solutions. The acid, being unbound and small, diffuses rapidly across the membrane from a concentrated feed to a dilute diluent, while the metal ions, being larger and often complexed, diffuse much slower or not at all. The driving force is the difference in the chemical potential of the acid on either side of the membrane.
Pervaporation
Pervaporation is a membrane separation process used for separating liquid mixtures, particularly azeotropes and heat-sensitive compounds, by utilizing a vacuum or sweep gas on the permeate side. The driving force is a combination of the partial pressure of the components in the liquid feed and the lower partial pressure on the permeate side. Components that preferentially permeate the membrane will evaporate into the vapor phase on the permeate side, driven by their reduced chemical potential in the vacuum.
Other Contributing Forces
Beyond bulk pressure and concentration differences, other forces can influence filtration, especially at the micro and nanoscale:
Electrically Driven Filtration
- Electroosmotic Flow (EOF): When a voltage is applied across a charged membrane or through charged pores in the presence of an electrolyte solution, the ions in the solution are attracted to the charged surfaces, dragging the bulk fluid along with them. This electroosmotic flow can contribute to or even drive filtration in specific applications, such as electrokinetic chromatography or some microfluidic devices.
- Electrophoresis: Charged particles suspended in a fluid can be moved by an electric field. If a membrane is present, electrophoresis can contribute to the separation by moving charged particles towards or away from the membrane, influencing their passage.
Thermal Diffusion (Soret Effect)
While less common as a primary driving force in typical filtration, temperature gradients can induce a mass flux due to differences in the thermal diffusion coefficients of components. This phenomenon, known as the Soret effect, can lead to minor separation effects in some membrane processes, particularly when significant temperature differences exist across the membrane.
Adsorption and Desorption
In some cases, the interaction between the components of the feed and the membrane material itself can indirectly influence filtration. If a component preferentially adsorbs onto the membrane surface, it can increase the effective concentration of that component near the membrane, potentially altering the driving forces for other components. Conversely, desorption can clear the membrane surface.
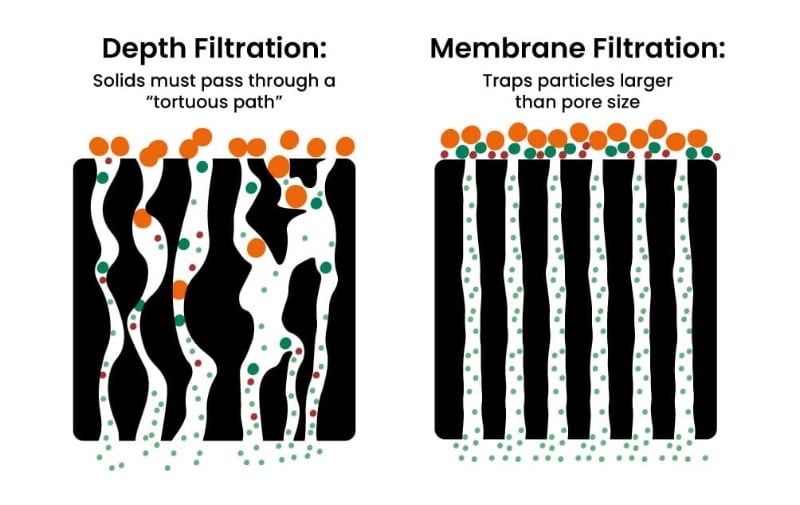
Synergistic Effects and Process Optimization
In many real-world membrane filtration systems, multiple driving forces can be at play, often synergistically. For instance, in cross-flow microfiltration of a biological suspension, the applied transmembrane pressure is the primary driver, but the shear forces from the tangential flow (related to fluid dynamics) are critical for managing fouling, indirectly influencing the effective driving force by maintaining membrane permeability. Similarly, in RO, the balance between applied pressure and osmotic pressure dictates the net driving force for water permeation.
Understanding the dominant driving forces, and the subtle influences of secondary forces, is paramount for:
- Selecting the appropriate membrane material and pore size: Different forces are best exploited by membranes with specific surface charges, pore structures, and chemical affinities.
- Optimizing operating parameters: Adjusting pressure, flow rates, temperature, and electrical potential can maximize separation efficiency and minimize energy consumption.
- Designing effective fouling mitigation strategies: Knowing which forces contribute to fouling allows for targeted countermeasures.
- Predicting and controlling permeate quality and flux: The driving forces directly dictate the separation performance and the rate at which purified fluid is obtained.
In conclusion, while pressure difference is the most commonly recognized and utilized force driving filtration through a membrane, concentration gradients and even electrical forces play vital roles in specialized applications. A comprehensive grasp of these fundamental principles is the cornerstone of successful and efficient membrane technology development and deployment across diverse scientific and industrial landscapes.
