Unveiling the Power of Gel Electrophoresis in Protein Analysis
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis, commonly abbreviated as SDS-PAGE, stands as a cornerstone technique in molecular biology and biochemistry. Its primary function is to separate proteins based on their molecular weight. This seemingly simple separation has profound implications, enabling researchers to analyze protein composition, purity, and relative abundance in complex biological samples. From fundamental research to diagnostic applications, SDS-PAGE provides an indispensable window into the proteome.
The Fundamental Principle: Charge, Size, and Denaturation
At its core, SDS-PAGE leverages two key principles: electrophoresis and protein denaturation. Electrophoresis, in general, is a process that utilizes an electric field to move charged molecules through a medium. Proteins, being complex molecules, possess inherent charges due to their amino acid composition. However, to make SDS-PAGE a tool for size-based separation, a crucial modification is introduced: the use of sodium dodecyl sulfate (SDS).

The Role of SDS: Uniform Negative Charge and Denaturation
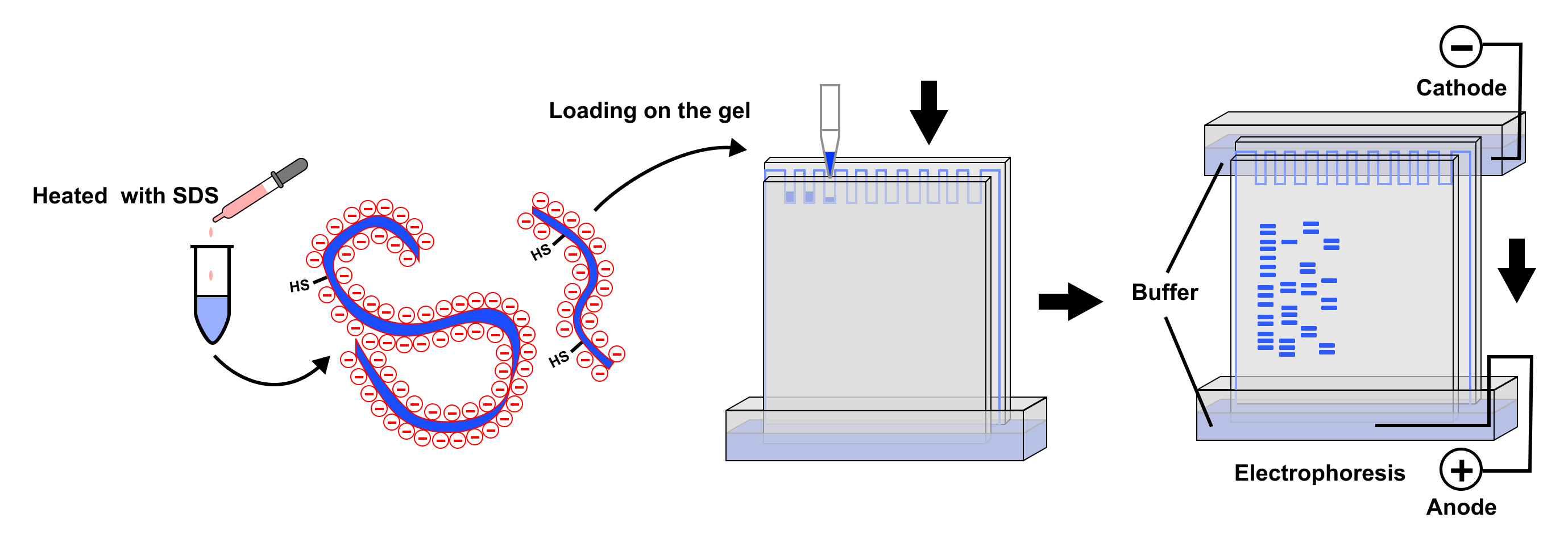
Sodium dodecyl sulfate is an anionic surfactant. When added to a protein sample, SDS molecules bind uniformly along the polypeptide chain, typically at a ratio of approximately one SDS molecule for every two amino acids. This extensive coating of SDS has two critical effects:
- Denaturation: SDS disrupts the non-covalent bonds that maintain a protein’s three-dimensional structure (secondary, tertiary, and quaternary structures). This unfolding process linearizes the protein, exposing its amino acid backbone.
- Uniform Negative Charge: By coating the protein chain, SDS imparts a strong net negative charge to each protein molecule. This negative charge is largely independent of the protein’s intrinsic amino acid composition and, importantly, is proportional to the protein’s mass.
This uniform charge-to-mass ratio is the linchpin of SDS-PAGE. Without SDS, protein separation would be a complex interplay of charge, size, and shape, making it difficult to isolate the effect of molecular weight alone.
The Polyacrylamide Gel: A Molecular Sieve
The “PAGE” in SDS-PAGE refers to the polyacrylamide gel, the matrix through which the denatured, negatively charged proteins migrate. This gel is formed by the polymerization of acrylamide monomers and a cross-linking agent, typically N,N’-methylenebisacrylamide (bis-acrylamide). The concentration of acrylamide in the gel is a critical parameter that dictates the pore size.
Manipulating Pore Size for Optimal Separation
- Higher Acrylamide Concentration: Results in smaller pore sizes. This is ideal for separating smaller proteins, as they can navigate through the gel more efficiently, and the smaller differences in migration can be resolved.
- Lower Acrylamide Concentration: Results in larger pore sizes. This is better suited for separating larger proteins, preventing them from getting “stuck” in the gel matrix and allowing them to migrate further.
The polyacrylamide gel acts as a molecular sieve. As the electric field is applied, the negatively charged protein molecules are pulled towards the positive electrode (anode). However, their movement is impeded by the gel matrix. Smaller proteins can navigate the pores more easily and thus migrate faster and further through the gel. Larger proteins, encountering more resistance from the gel’s intricate network of pores, move more slowly and cover less distance.
The Electrophoresis Process: Migration and Separation

The SDS-PAGE apparatus consists of a gel cast between two glass plates, submerged in a buffer solution. This buffer typically contains SDS and buffering salts to maintain a stable pH and ionic strength, which are crucial for efficient electrophoresis. Electrodes are placed in the buffer, and an electric current is applied.
Step-by-Step Migration:
- Sample Loading: The protein sample, pre-treated with SDS and a reducing agent (like DTT or $beta$-mercaptoethanol, which breaks disulfide bonds and further aids denaturation), is carefully loaded into wells at the top of the gel. A molecular weight marker (a mixture of proteins of known sizes) is also loaded in a separate well for comparison.
- Power Supply Activation: A constant voltage or current is applied across the gel. The electric field drives the negatively charged SDS-coated proteins towards the anode at the bottom of the gel.
- Migration and Separation: As proteins migrate, they encounter the polyacrylamide gel matrix. Smaller proteins move unimpeded through the pores, while larger proteins are retarded. This differential migration leads to the formation of distinct protein bands, separated according to their molecular weights.
- Running to Completion: The electrophoresis is typically run until the loading dye (a colored molecule that migrates slightly faster than most proteins) reaches the bottom of the gel. This ensures that all proteins have migrated sufficiently for clear separation.
Visualization: Making the Invisible Visible
Once the electrophoresis is complete, the separated protein bands are invisible to the naked eye. Therefore, a staining step is necessary to visualize them.
Common Staining Methods:
- Coomassie Brilliant Blue: This is the most widely used stain for total protein. Coomassie Blue binds to proteins, and the stained bands appear as distinct blue lines against a clear background. It’s a sensitive stain, allowing detection of proteins down to nanogram quantities.
- Silver Staining: Significantly more sensitive than Coomassie Blue, silver staining can detect proteins in picogram quantities. It involves a series of chemical reactions that precipitate silver ions onto the protein bands, resulting in very dark, visible lines. While highly sensitive, it is more complex and time-consuming than Coomassie staining.
- Fluorescent Stains: These stains emit fluorescence when excited by specific wavelengths of light, offering high sensitivity and a wider dynamic range. Examples include SYPRO Ruby and Deep Purple.
- Western Blotting: While not strictly a visualization technique for SDS-PAGE itself, Western blotting is a common downstream application. After SDS-PAGE separation, proteins are transferred to a membrane (e.g., nitrocellulose or PVDF). This membrane can then be probed with specific antibodies to detect and identify particular proteins of interest. This technique combines the size separation power of SDS-PAGE with the specificity of antibody recognition.
Applications of SDS-PAGE: A Versatile Tool
The ability of SDS-PAGE to resolve proteins by size makes it invaluable across a multitude of scientific disciplines.
Key Applications:
- Assessing Protein Purity: Researchers can determine if a purified protein sample contains contaminating proteins by observing the presence of additional bands.
- Estimating Molecular Weight: By comparing the migration distance of an unknown protein to that of a molecular weight marker, its approximate molecular weight can be estimated. This is a fundamental piece of information in protein characterization.
- Analyzing Protein Expression Levels: While SDS-PAGE provides a qualitative or semi-quantitative measure of protein abundance (depending on staining intensity), it can reveal changes in protein expression levels under different experimental conditions. For precise quantification, techniques like Western blotting or mass spectrometry are often employed.
- Quality Control in Protein Production: In biotechnology and pharmaceutical industries, SDS-PAGE is used to verify the integrity and purity of recombinant proteins produced for therapeutic or research purposes.
- Diagnosing Diseases: SDS-PAGE, particularly in conjunction with Western blotting, has been used in the diagnosis of certain diseases by detecting specific protein markers or changes in protein profiles. For instance, it played a historical role in the diagnosis of HIV.
- Studying Protein Complexes (Limited): While SDS-PAGE is primarily a denaturing technique, under certain conditions, it can be used to study the subunits of protein complexes or to assess the stability of protein interactions, although non-denaturing gel electrophoresis methods are generally preferred for preserving native protein structures.
Limitations and Considerations
Despite its widespread utility, SDS-PAGE has certain limitations that researchers must consider:
- Denaturing Conditions: The use of SDS and reducing agents irreversibly denatures proteins. Therefore, SDS-PAGE cannot be used to study the native function or three-dimensional structure of proteins.
- Limited Resolution for Similar Sized Proteins: Proteins with very similar molecular weights may not be resolved into distinct bands, especially in higher molecular weight ranges where migration differences are less pronounced.
- Influence of Post-Translational Modifications: While SDS binding largely standardizes the charge-to-mass ratio, some extensive post-translational modifications (PTMs) can slightly alter a protein’s migration pattern.
- Time-Consuming: The entire process, from sample preparation to visualization, can take several hours to over a day, depending on the staining method.
Conclusion: An Enduring Standard in Protein Science
SDS-PAGE remains an indispensable tool in the molecular biologist’s arsenal. Its straightforward principle, robust methodology, and versatility in separating proteins by molecular weight have solidified its position as a fundamental technique. From deciphering cellular mechanisms to developing novel diagnostics, the ability to visualize and analyze the protein landscape through SDS-PAGE continues to drive scientific discovery and innovation. While more advanced techniques have emerged, SDS-PAGE endures as a reliable, accessible, and informative method for protein analysis, consistently providing crucial insights into the complex world of proteins.
