Cyclins are a group of proteins crucial for the regulation of the cell cycle. They act as regulatory subunits for cyclin-dependent kinases (CDKs), enzymes that drive the cell cycle forward. By binding to CDKs, cyclins activate them and direct them to specific substrates, thus controlling the transitions between different phases of the cell cycle, including DNA replication, chromosome condensation, and cell division. The precise temporal and spatial expression of cyclins is tightly regulated, ensuring that the cell cycle progresses in an orderly and accurate manner. Dysregulation of cyclin activity can lead to uncontrolled cell proliferation, a hallmark of cancer.
The Dynamic Dance of Cyclin Expression and CDK Activity
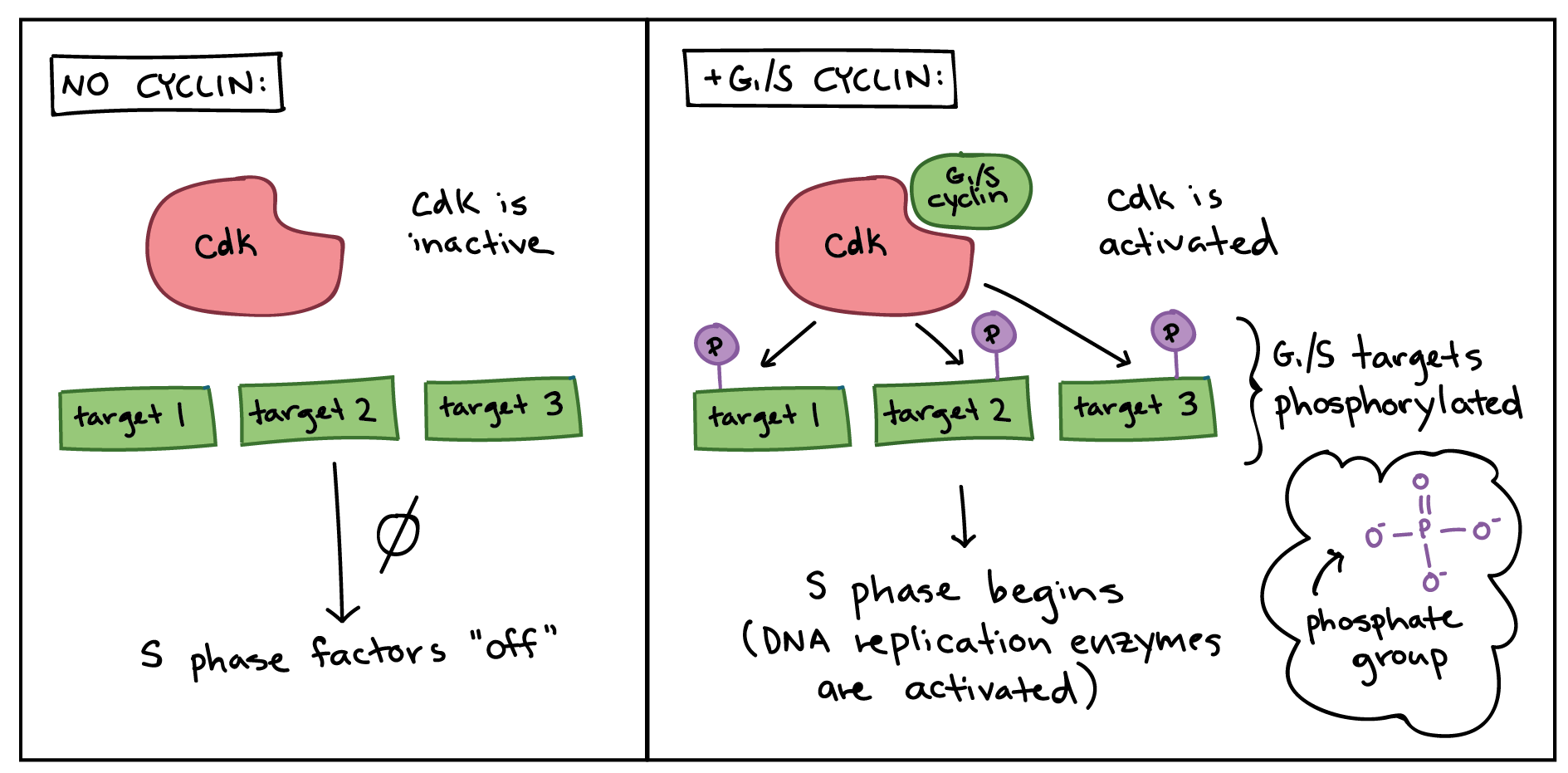
The cell cycle is a complex series of events that a cell undergoes to grow and divide. This intricate process is meticulously orchestrated by a molecular machinery, at the heart of which lie cyclins and their partners, the cyclin-dependent kinases (CDKs). Think of CDKs as the engines of the cell cycle, providing the necessary enzymatic power to drive key cellular processes. However, these engines are only activated and directed by cyclins, which act as the keys that turn them on and specify their targets.
Cyclins: The Oscillating Regulators of Cell Division
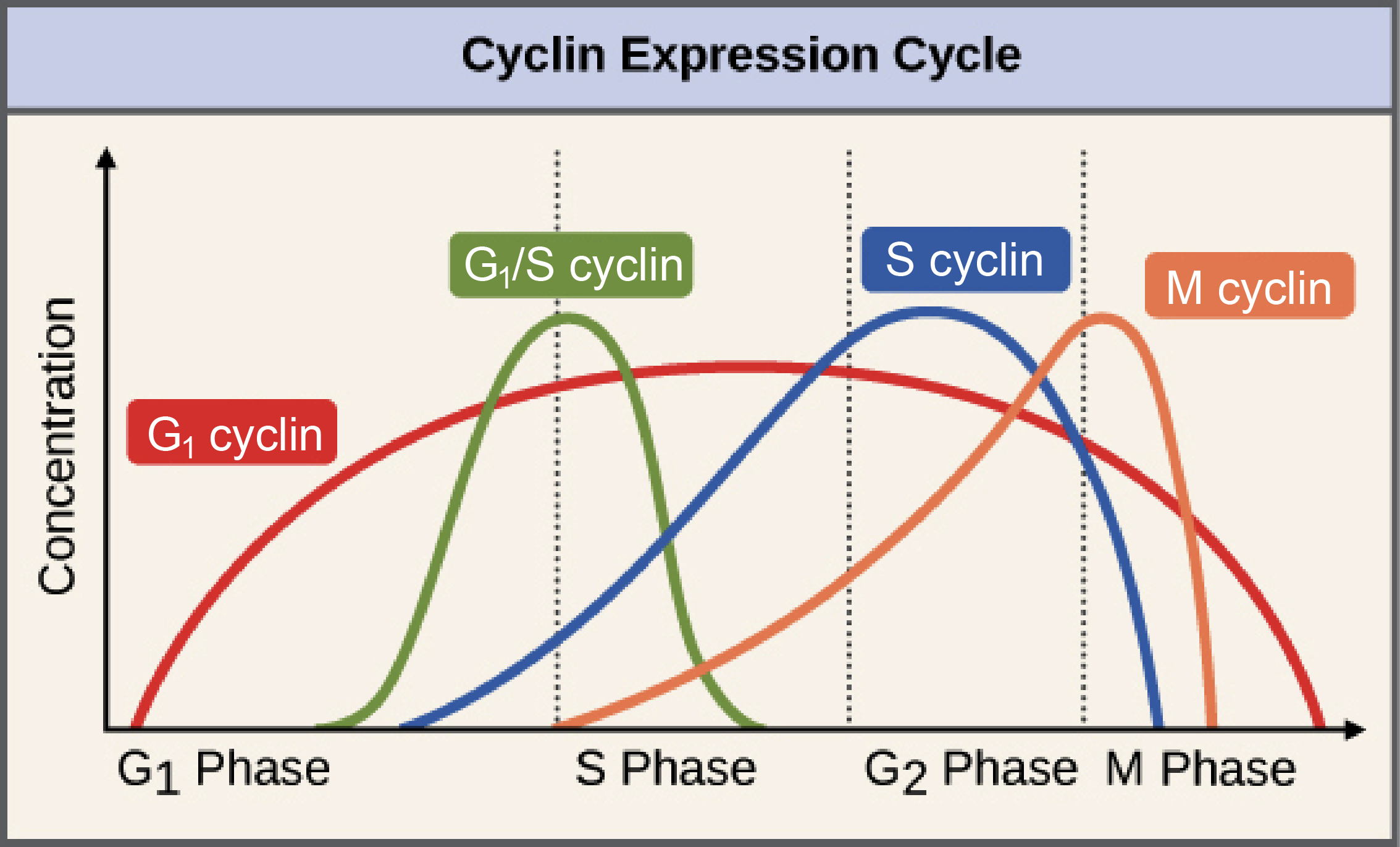
The name “cyclin” itself hints at their fundamental characteristic: their concentration fluctuates cyclically throughout the cell cycle. Unlike most other proteins within the cell, cyclins are synthesized and degraded in a highly regulated manner. This dynamic expression pattern is not arbitrary; it’s a critical mechanism for ensuring that specific CDK complexes are active only at the appropriate times and in the correct cellular compartments.
G1 Cyclins: Initiating the Journey
At the beginning of the cell cycle, during the G1 phase, cells prepare for DNA replication. This phase is driven by a class of cyclins often referred to as G1 cyclins, such as Cyclin D. Cyclin D levels begin to rise as the cell receives external growth signals, like growth factors. This increase in Cyclin D allows it to bind to and activate specific CDKs, notably CDK4 and CDK6. The resulting CDK4/6-Cyclin D complexes play a pivotal role in phosphorylating key target proteins, such as the retinoblastoma protein (Rb). Phosphorylation of Rb releases it from its inhibitory hold on transcription factors like E2F. Once released, E2F can then activate the transcription of genes essential for DNA synthesis and progression into the S phase of the cell cycle. This initiation phase is crucial for ensuring the cell commits to division.
S-Phase Cyclins: The Engine of DNA Replication
Once the cell has committed to division and passed the critical restriction point in G1, it enters the S phase, where DNA replication occurs. This phase is driven by S-phase cyclins, most notably Cyclin E and Cyclin A. Cyclin E, in complex with CDK2, is important for initiating DNA replication. It further phosphorylates Rb, ensuring sustained E2F activity, and also targets other proteins involved in DNA replication initiation. As the cell progresses further into S phase, Cyclin A levels rise, forming complexes with CDK2 and sometimes CDK1. This complex is crucial for the progression of DNA replication forks and the maintenance of DNA synthesis. The coordinated action of Cyclin E and Cyclin A ensures that the entire genome is faithfully duplicated before the cell can proceed to the next stages.
M-Phase Cyclins: Orchestrating Mitosis
The final stages of the cell cycle involve mitosis (M phase), the process of nuclear division and cell division. This dramatic event is meticulously orchestrated by M-phase cyclins, primarily Cyclin B, which forms complexes with CDK1. The Cyclin B-CDK1 complex, often referred to as the “mitotic CDK” or “MPF” (Maturation Promoting Factor), is the master regulator of entry into mitosis. Upon activation, MPF phosphorylates a vast array of substrates, leading to the dramatic morphological changes characteristic of mitosis, including chromosome condensation, nuclear envelope breakdown, and the formation of the mitotic spindle. Once mitosis is complete, the cell enters cytokinesis. For this process to occur smoothly, the activity of MPF must be rapidly and abruptly shut down.
Cyclin Degradation: The Switch to Cell Cycle Progression
A critical aspect of cyclin function is their controlled degradation. This process is primarily mediated by the ubiquitin-proteasome system. Specific ubiquitin ligases, such as the Anaphase-Promoting Complex/Cyclosome (APC/C), target cyclins for destruction at specific points in the cell cycle. For example, the degradation of Cyclin B by the APC/C is essential for the exit from mitosis. When Cyclin B levels drop, MPF activity is abolished, allowing the cell to decondense its chromosomes, reform the nuclear envelope, and prepare for cytokinesis. Similarly, the degradation of S-phase cyclins is crucial for preventing re-replication of DNA within a single cell cycle. This timely destruction ensures that the cell progresses through the cycle and doesn’t get stuck in a particular phase.
Cyclin-Dependent Kinases (CDKs): The Catalytic Engines of the Cell Cycle
While cyclins provide the regulatory specificity, it is the cyclin-dependent kinases (CDKs) that perform the actual enzymatic work of driving the cell cycle forward. These are a family of serine/threonine protein kinases that, in their inactive state, are unable to phosphorylate their target proteins. Their activity is entirely dependent on their association with cyclins.
CDK Activation and Specificity
The binding of a specific cyclin to a CDK is the first crucial step in activating the kinase. However, this association is often not sufficient for full catalytic activity. Further modifications, such as phosphorylation at specific sites, can either enhance or inhibit CDK activity. Once activated, a CDK complex, consisting of a CDK subunit and its bound cyclin, can then bind to and phosphorylate specific target proteins. The particular set of proteins a CDK phosphorylates is determined by the specific cyclin it is associated with and the cellular context. This ensures that the correct cellular events are triggered at the right time.
Key CDK Partners and Their Roles
Different cyclins associate with different CDKs to drive distinct cell cycle events. For instance, as mentioned, Cyclin D partners with CDK4 and CDK6 to promote entry into S phase. Cyclin E and Cyclin A both partner with CDK2, with Cyclin E initiating DNA replication and Cyclin A driving its progression. The most prominent CDK in the M phase is CDK1, which partners with Cyclin B to orchestrate mitosis. The specificity of these pairings is paramount for the precise control of the cell cycle.
The Unfolding Consequences of Cyclin Dysregulation
The meticulous regulation of cyclin activity is vital for maintaining genomic integrity and preventing uncontrolled cell proliferation. When this regulation breaks down, the consequences can be severe, leading to a variety of diseases, most notably cancer.
Cancer and the Cyclin Network
In many types of cancer, the cell cycle control mechanisms are corrupted. This often involves alterations in the expression levels or activity of cyclins and CDKs. For example, overexpression of Cyclin D is a common event in various cancers, leading to excessive stimulation of CDK4/6 and premature entry into S phase. Similarly, mutations in CDK inhibitors, proteins that normally act as brakes on the cell cycle, can also lead to uncontrolled proliferation. The intricate feedback loops and checkpoints that normally prevent aberrant cell division are bypassed, allowing cells with damaged DNA to divide, accumulating further mutations and progressing towards malignancy. Understanding how cyclins function is therefore critical for developing targeted cancer therapies.
Therapeutic Strategies Targeting Cyclins
Given the central role of cyclins in cell proliferation and their frequent dysregulation in cancer, they have become attractive targets for therapeutic intervention. A significant advancement in cancer treatment has been the development of CDK inhibitors. These drugs work by blocking the activity of specific CDKs, thereby halting the progression of cancer cells through the cell cycle. For example, palbociclib, ribociclib, and abemaciclib are approved CDK4/6 inhibitors used in combination with hormonal therapy for certain types of breast cancer. These drugs effectively prevent the activation of transcription factors necessary for cell cycle progression, leading to cell cycle arrest and ultimately, tumor regression. Future research continues to explore novel therapeutic strategies that precisely target the cyclin-CDK machinery to combat various cancers and other proliferative diseases.
Beyond Cell Division: Emerging Roles of Cyclins
While their primary and most well-understood role is in cell cycle control, emerging research suggests that cyclins may also participate in other cellular processes. These novel functions highlight the multifaceted nature of these regulatory proteins and open new avenues for scientific inquiry.
Transcriptional Regulation and Beyond
Evidence is accumulating that certain cyclins and their associated CDK partners can influence gene expression independently of their role in cell cycle progression. For instance, some CDK complexes have been implicated in regulating the activity of transcription factors, thereby modulating the expression of genes involved in various cellular pathways, including differentiation and development. Furthermore, cyclins have been observed to play roles in processes such as DNA repair and RNA processing, suggesting a broader involvement in cellular homeostasis than previously appreciated. These findings underscore the complexity of cyclin biology and suggest that their influence extends far beyond the traditional boundaries of cell cycle regulation. The ongoing exploration of these alternative functions promises to further unravel the intricate roles of cyclins in cellular life.
