The term “gas” is incredibly broad, encompassing a vast array of substances that share the fundamental characteristic of existing in a gaseous state – a state of matter where particles are widely separated and move randomly. This inherent fluidity and expansiveness allow gases to fill any container they occupy, unlike solids or liquids. However, when we delve deeper, the “types of gas” can be categorized and understood through various lenses, each offering a unique perspective on their properties, applications, and significance. For this exploration, we will focus on the fundamental chemical and physical distinctions that define different gases, setting the stage for understanding their diverse roles in technology and industry.
The Fundamental Nature of Gases: Molecular Behavior and States of Matter
To grasp the different types of gases, we must first understand the foundational principles governing their existence. Gases are defined by the arrangement and motion of their constituent particles – atoms or molecules. Unlike the tightly packed and ordered structures of solids, or the more fluid but still relatively close arrangement of liquids, gas particles are in constant, rapid, and largely independent motion.
Kinetic Molecular Theory: The Engine of Gaseous Behavior
The behavior of gases is best explained by the Kinetic Molecular Theory. This theory posits several key assumptions:
- Particles in Constant Motion: Gas particles are in perpetual, random motion, colliding with each other and the walls of their container.
- Negligible Intermolecular Forces: The forces of attraction or repulsion between gas particles are considered negligible, especially at low pressures and high temperatures. This means particles essentially behave independently.
- Vast Interparticle Distances: The volume occupied by the gas particles themselves is insignificant compared to the total volume of the container. There are large empty spaces between particles.
- Elastic Collisions: Collisions between gas particles and between particles and container walls are perfectly elastic. This means no kinetic energy is lost during a collision; it is simply transferred.
- Average Kinetic Energy is Proportional to Temperature: The average kinetic energy of gas particles is directly proportional to the absolute temperature of the gas. As temperature increases, particles move faster, leading to more frequent and forceful collisions.
These postulates are crucial for understanding why gases exhibit properties like compressibility, expansibility, and diffusion, and how they differ from other states of matter. The kinetic energy of the particles is the driving force behind these behaviors, and temperature is the direct measure of this average kinetic energy.
States of Matter: The Gas Phase in Context
The gaseous state is one of the fundamental states of matter, alongside solid, liquid, and plasma. Understanding the transitions between these states helps define what a gas truly is.
- Solid to Gas (Sublimation): Some substances can transition directly from a solid to a gaseous state without passing through a liquid phase (e.g., dry ice, which is solid carbon dioxide, sublimates into gaseous carbon dioxide).
- Liquid to Gas (Vaporization/Boiling): When a liquid absorbs enough energy, its particles gain sufficient kinetic energy to overcome the intermolecular forces holding them together, escaping into the gaseous phase. This can occur gradually through evaporation or more rapidly through boiling at a specific temperature.
- Gas to Liquid (Condensation): The reverse of vaporization, where gas particles lose kinetic energy and come closer together, forming a liquid.
- Gas to Solid (Deposition): The reverse of sublimation, where a gas directly transforms into a solid (e.g., frost forming on a cold surface).
The state of a substance is dependent on temperature and pressure. Under specific conditions, a substance will exist as a gas. The very nature of a gas – its ability to expand indefinitely and fill its container – is a direct consequence of the high kinetic energy and minimal intermolecular forces of its constituent particles.
Classification of Gases: Chemical Composition and Properties
While all gases share the fundamental characteristics of their state of matter, they are differentiated by their chemical composition and resulting physical and chemical properties. This leads to a rich diversity of gases with distinct applications.
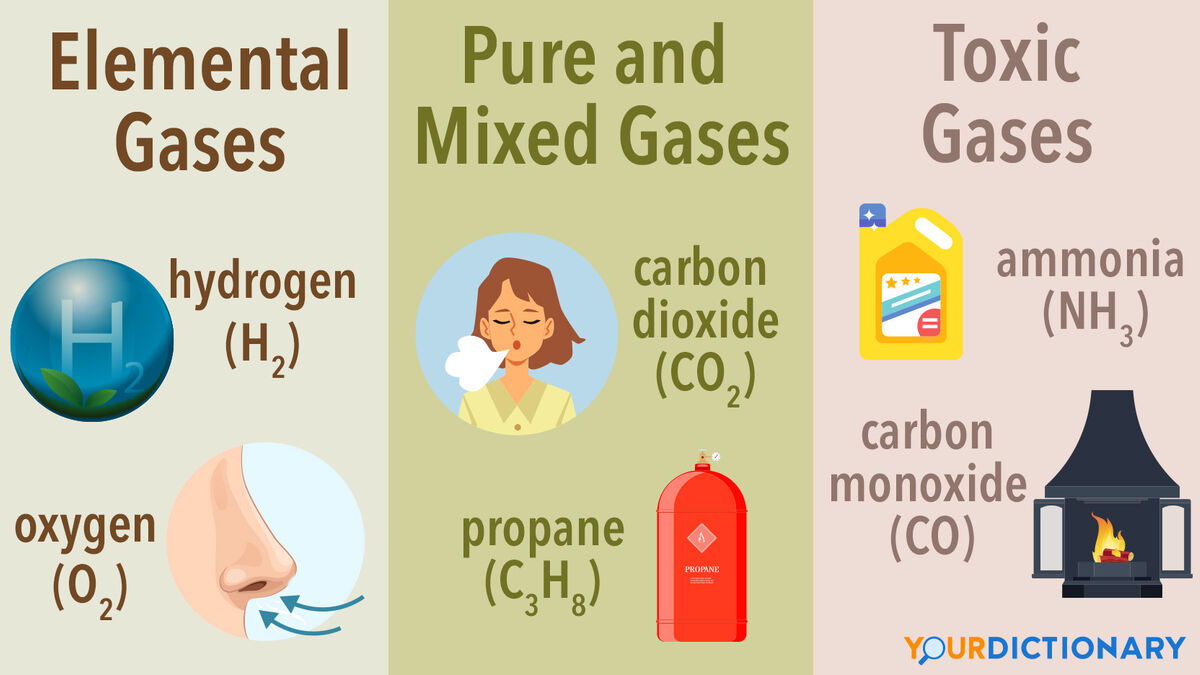
Elemental Gases: The Building Blocks
Elemental gases are gases that consist of only one type of atom. These are often found as diatomic molecules (two atoms of the same element bonded together) in their natural state.
- Hydrogen (H₂): The lightest and most abundant element in the universe. It is a colorless, odorless, and highly flammable gas. Hydrogen is a key component in fuels, industrial processes, and is being explored as a clean energy source.
- Nitrogen (N₂): The most abundant gas in Earth’s atmosphere (approximately 78%). It is a colorless, odorless, and relatively inert gas. Nitrogen is crucial for life, used in fertilizers, food preservation, and as an inert atmosphere in industrial applications.
- Oxygen (O₂): Essential for respiration in most living organisms. It is a colorless, odorless gas that supports combustion. Oxygen is used in medical applications, welding, and various industrial processes.
- Halogens (F₂, Cl₂, Br₂, I₂): Fluorine, chlorine, bromine, and iodine exist as diatomic molecules in the gaseous state (though bromine is a liquid at room temperature, its vapor is gaseous). These are highly reactive and toxic gases, used in disinfectants, refrigerants, and chemical synthesis.
- Noble Gases (He, Ne, Ar, Kr, Xe, Rn): These are monatomic gases (existing as single atoms) due to their stable electron configurations. They are known for their inertness, making them useful in applications like lighting (neon signs, argon in welding), cryogenics (helium), and radiation detection.
Compound Gases: Combinations of Elements
Compound gases are formed when two or more different elements combine chemically to form molecules that exist in the gaseous state under standard conditions.
- Carbon Dioxide (CO₂): A colorless, odorless gas produced by respiration, combustion, and fermentation. It is a greenhouse gas, essential for photosynthesis, and used in carbonated beverages and fire extinguishers.
- Methane (CH₄): The simplest hydrocarbon and a major component of natural gas. It is a colorless, odorless, flammable gas used as a fuel and in the production of other chemicals.
- Ammonia (NH₃): A colorless gas with a pungent odor. It is produced synthetically and is widely used in fertilizers, cleaning products, and as a refrigerant.
- Sulfur Dioxide (SO₂): A colorless gas with a sharp, suffocating odor. It is produced by the burning of sulfur-containing fuels and is a major air pollutant, though it has some industrial uses.
- Nitric Oxide (NO) and Nitrogen Dioxide (NO₂): These are related gases that are components of air pollution and are involved in atmospheric chemistry.
- Ozone (O₃): A molecule composed of three oxygen atoms. In the stratosphere, it forms a protective layer against UV radiation. In the troposphere, it is a harmful pollutant.
The distinction between elemental and compound gases is fundamental, highlighting how atomic structure and bonding dictate the properties and reactivity of a gaseous substance.
Functional Classifications: Gases by Application and Properties

Beyond their chemical composition, gases are often categorized by their functional properties and how they are utilized in various fields. This perspective is particularly relevant when discussing their impact on technology, industry, and everyday life.
Inert Gases: The Stable Companions
Inert gases, primarily the noble gases and nitrogen, are characterized by their low reactivity. This stability makes them invaluable for creating controlled environments and preventing unwanted chemical reactions.
- Shielding Gases in Welding: Argon and helium are used as shielding gases in arc welding processes. They displace air (which contains oxygen and nitrogen that can oxidize or embrittle the molten metal) to protect the weld from contamination, ensuring a strong and clean bond.
- Inert Atmospheres in Manufacturing: Nitrogen is widely used to create an inert atmosphere in the production of electronics, food packaging, and pharmaceuticals. This prevents oxidation, spoilage, and degradation of sensitive materials.
- Cryogenics: Liquid helium and liquid nitrogen, due to their extremely low boiling points, are used as refrigerants for cooling superconducting magnets in MRI machines, particle accelerators, and scientific research.
- Lighting and Displays: Neon gas produces a distinctive red glow when an electric current passes through it, forming the basis of neon signs. Other noble gases are used in fluorescent lights and specialized displays.
Reactive Gases: The Transformative Agents
Reactive gases are those that readily participate in chemical reactions. While this reactivity can pose hazards, it is also the basis for many essential industrial processes and energy generation.
- Combustion Gases: Oxygen is crucial for combustion, the process that releases energy from fuels. Understanding the behavior of gases like carbon monoxide (CO) and carbon dioxide (CO₂) produced during combustion is vital for engine design, emissions control, and energy efficiency.
- Industrial Synthesis: Gases like hydrogen are used in the Haber-Bosch process for ammonia production, a cornerstone of modern agriculture. Chlorine gas is used in the production of plastics and disinfectants.
- Fuel Gases: Methane (natural gas), propane, and butane are commonly used as fuel gases for heating, cooking, and power generation. Their controlled combustion releases thermal energy.
- Plasma Applications: While plasma is considered a distinct state of matter, it is often generated by introducing specific gases into an electric field. Gases like argon and nitrogen are commonly used in plasma torches for cutting and welding, and in plasma etching for semiconductor manufacturing.
Specialty Gases: Tailored for Precision
Specialty gases are high-purity gases or precisely blended gas mixtures designed for specific, often critical, applications. Their purity and composition are meticulously controlled.
- Calibration Gases: These gas mixtures are used to calibrate analytical instruments, ensuring their accuracy and reliability in fields like environmental monitoring, industrial safety, and medical diagnostics.
- Semiconductor Manufacturing: Ultra-high purity gases and complex gas mixtures are essential for etching, deposition, and doping processes in the fabrication of integrated circuits.
- Medical Gases: Pure oxygen, nitrous oxide (for anesthesia), and medical air are examples of specialty gases used in healthcare, requiring stringent quality control.
- Scientific Research: A wide array of pure gases and custom mixtures are used in laboratories for spectroscopy, chromatography, and other analytical techniques.
The functional classification of gases highlights their diverse roles, from enabling fundamental life processes to powering advanced technological innovations. Understanding these roles requires an appreciation for both their chemical nature and their specific physical properties under various conditions.
The Future of Gases: Innovation and Emerging Applications
The study and application of gases continue to evolve, driven by advancements in materials science, energy technology, and environmental concerns. New types of gases and novel applications are constantly being explored.
Hydrogen as a Clean Energy Carrier
Hydrogen gas is increasingly recognized as a promising clean energy carrier. Its potential to be produced from renewable sources and its ability to generate electricity through fuel cells with only water as a byproduct make it a key focus for decarbonization efforts. Research is ongoing in efficient hydrogen production, storage, and transportation.
Advanced Materials and Gas Interactions
The interaction of gases with advanced materials is opening new frontiers. This includes:
- Gas Sensors: Development of highly sensitive and selective gas sensors for detecting pollutants, hazardous substances, and even biomarkers for medical diagnostics.
- Gas Storage: Research into new materials and methods for safely and efficiently storing gases like hydrogen under high pressure or at lower temperatures.
- Gas Separation and Purification: Innovative membrane technologies and adsorption processes for separating and purifying gases, crucial for industrial efficiency and environmental remediation.

Sustainable Industrial Processes
There is a growing emphasis on developing more sustainable industrial processes that minimize gas emissions and maximize resource efficiency. This involves:
- Carbon Capture and Utilization (CCU): Technologies to capture carbon dioxide from industrial sources and convert it into valuable products, such as fuels, chemicals, or building materials.
- Green Chemistry: Designing chemical processes that use less hazardous gases and generate fewer harmful byproducts.
The ongoing exploration of “types of gas” is not merely an academic exercise; it is fundamental to our ability to innovate and address some of the most pressing challenges facing society, from energy sustainability to environmental protection and advanced manufacturing. As our understanding deepens, the role of gases in shaping our future will undoubtedly continue to expand.
