The intricate world of chemistry, often perceived as abstract, forms the bedrock for countless technological advancements, including the sophisticated capabilities of modern drones. While seemingly disparate, understanding fundamental chemical concepts like “lone pairs” is crucial for pushing the boundaries of drone utility, especially in specialized fields such as remote sensing, environmental monitoring, and hazardous material detection. Lone pairs, non-bonding electron pairs in a molecule, are not merely theoretical constructs; they are critical determinants of a molecule’s shape, polarity, and reactivity, factors that directly influence how a chemical substance behaves and, consequently, how it can be identified, quantified, and interacted with by advanced drone payloads.

The Fundamental Building Blocks of Molecular Function for Remote Sensing
At its core, a lone pair represents a pair of valence electrons that are not shared with another atom in a covalent bond. These unshared electrons occupy specific regions of space around an atom, exerting significant influence over the molecule’s overall structure and electronic properties. For drone-based remote sensing, understanding these fundamental molecular attributes is paramount, as they dictate how chemicals interact with electromagnetic radiation or specific sensor materials, thereby enabling their detection from a distance.
Understanding Electron Configuration and Valence Shells
Every atom possesses a unique electron configuration, dictating how its electrons are distributed in shells and subshells. The outermost shell, known as the valence shell, contains the electrons primarily involved in chemical bonding. When atoms bond to form molecules, they often do so to achieve a stable octet (eight valence electrons) or duet (two valence electrons for hydrogen and helium). Electrons involved in these shared bonds are called bonding pairs. However, atoms like nitrogen, oxygen, and halogens often have valence electrons that are not part of a bond. These are the lone pairs. For instance, in a water molecule (H₂O), oxygen has six valence electrons. Two are shared with hydrogen atoms, forming two bonding pairs. The remaining four valence electrons exist as two lone pairs on the oxygen atom. This seemingly simple detail is profound, as these lone pairs are not inert; they are highly active in determining the molecule’s properties. In the context of drone-based chemical analysis, knowing the electron configuration helps predict which elements will likely carry lone pairs, guiding the selection and calibration of sensors designed to detect specific functional groups or elements.
The VSEPR Theory and Molecular Geometry: Shaping Sensor Interactions
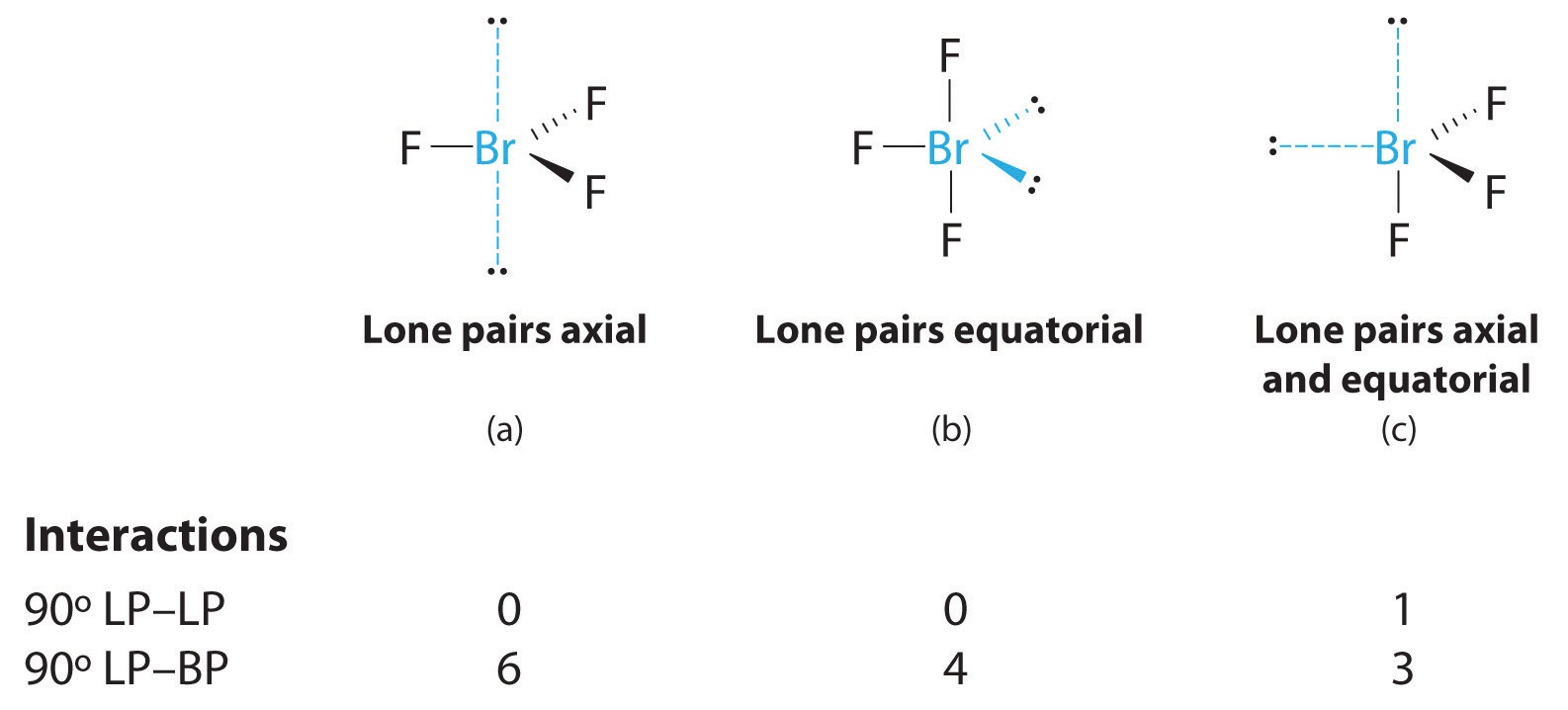
The Valence Shell Electron Pair Repulsion (VSEPR) theory provides a powerful framework for predicting the three-dimensional geometry of molecules, a property critically influenced by lone pairs. VSEPR theory posits that electron pairs (both bonding and lone pairs) around a central atom will arrange themselves as far apart as possible to minimize repulsion. Lone pairs, being localized solely on the central atom and not shared between two nuclei, exert a greater repulsive force than bonding pairs. This increased repulsion compresses the bond angles between bonding pairs, significantly altering the molecule’s shape.
For example, a methane molecule (CH₄) with four bonding pairs and no lone pairs around carbon adopts a perfect tetrahedral geometry with bond angles of 109.5°. Ammonia (NH₃), however, has three bonding pairs and one lone pair around nitrogen. The lone pair’s stronger repulsion pushes the bonding pairs closer, resulting in a trigonal pyramidal shape with bond angles reduced to approximately 107°. Water (H₂O) has two bonding pairs and two lone pairs on oxygen, leading to an even greater compression of the H-O-H bond angle to roughly 104.5°, giving it a bent or V-shape.
Why is molecular geometry crucial for drone technology? The shape of a molecule dictates its polarity, its ability to interact with other molecules, and how it absorbs or scatters light. A drone-mounted spectrometer, for instance, relies on a molecule’s unique vibrational or rotational modes—which are directly tied to its geometry—to identify it. Different shapes lead to different dipole moments and distinct spectral fingerprints. For advanced chemical sensing, a deep understanding of VSEPR theory allows engineers to predict molecular behavior, design more selective sensing elements, and interpret spectroscopic data with higher accuracy, transforming raw data from drone sensors into actionable intelligence about environmental conditions or chemical threats.
Driving Chemical Reactivity and Identification in Drone-Based Analytics
Beyond influencing shape, lone pairs are central to a molecule’s reactivity, playing a pivotal role in how molecules participate in chemical reactions. Their presence makes certain atoms electron-rich, driving specific types of interactions that are key to chemical detection and analysis performed by autonomous or remotely operated drone systems.
Lone Pairs as Nucleophiles: Guiding Chemical Reactions for Detection
Lone pairs often act as nucleophiles—electron-pair donors—in chemical reactions. Because lone pairs are concentrated regions of negative charge, they are attracted to electron-deficient sites (electrophiles) on other molecules. This nucleophilic character is fundamental to many reactions, including acid-base chemistry and various organic reactions. For instance, the lone pair on the nitrogen atom in ammonia makes it a base, readily accepting a proton. Similarly, oxygen’s lone pairs in water allow it to act as a solvent and participate in hydrolysis reactions.
In the realm of drone-based chemical analytics, this nucleophilic nature can be harnessed for selective detection. Specialized drone payloads might include micro-reactors or sensor surfaces coated with specific electrophilic compounds designed to selectively react with target molecules possessing lone pairs. By detecting the products of these reactions, or the changes they induce on a sensor’s electrical or optical properties, drones can identify and quantify specific substances in the environment. This principle is vital for developing sensors that can distinguish between similar compounds in complex mixtures, enhancing the specificity of remote chemical analysis.
Spectroscopic Signatures: Detecting Lone Pair Interactions with Drone-Mounted Sensors
Spectroscopy, the study of the interaction between matter and electromagnetic radiation, is a cornerstone of remote sensing. Lone pairs significantly influence a molecule’s spectroscopic signature, providing unique fingerprints that drone-mounted sensors can detect. For example, lone pairs contribute to a molecule’s dipole moment, which dictates its ability to absorb or emit microwave and infrared radiation. Rotational and vibrational spectroscopy, often employed by drones for atmospheric gas analysis, relies heavily on these properties.
Furthermore, lone pairs are often involved in electronic transitions that are detectable by UV-Vis spectroscopy. Molecules with lone pairs often have non-bonding (n) electrons that can be excited to higher energy anti-bonding orbitals. These n→π* transitions are characteristic of many organic molecules containing heteroatoms (like oxygen, nitrogen, sulfur) with lone pairs. A drone equipped with a compact UV-Vis spectrometer can leverage these unique absorption patterns to identify specific pollutants, biological agents, or industrial chemicals by analyzing the light transmitted through or reflected from a target area. Understanding how lone pairs contribute to these spectral features is indispensable for designing and calibrating these sophisticated drone sensing platforms.
Enhancing Environmental Monitoring and Hazard Detection with Drone Technology
The theoretical understanding of lone pairs directly translates into practical applications for drone-enabled environmental monitoring and hazard detection. By integrating chemical knowledge into sensor design and data interpretation, drones can provide unprecedented capabilities for safeguarding public health and ecosystems.
Precision in Chemical Identification for Air and Water Quality Monitoring
Drones are increasingly deployed for real-time air and water quality monitoring, offering a flexible and cost-effective alternative to traditional stationary sensors or manned sampling missions. The precision of chemical identification in these applications hinges on the ability of drone-mounted sensors to selectively detect target compounds amidst a myriad of others. Lone pairs play a crucial role here. For instance, many common atmospheric pollutants like ammonia (NH₃), hydrogen sulfide (H₂S), and sulfur dioxide (SO₂) contain lone pairs that make them highly reactive and polar. These properties influence their interaction with specific adsorbent materials in gas sensors or their distinct absorption profiles in optical sensors.
By understanding the lone pair geometry and reactivity of these pollutants, engineers can design sensor arrays that leverage differential detection methods. For example, a drone could deploy a sensor that changes electrical resistance upon contact with a nucleophilic lone-pair containing molecule, while simultaneously employing a spectroscopic sensor to confirm the specific vibrational fingerprint. This multi-modal approach, informed by fundamental chemical principles, enhances the reliability and accuracy of environmental data collected by UAVs, enabling more effective pollution control and environmental management.
Rapid Response to Chemical Spills and Hazardous Gas Leaks
In emergency scenarios involving chemical spills or hazardous gas leaks, rapid and accurate identification of the released substances is critical for informing containment strategies and ensuring responder safety. Drones, with their ability to access dangerous or inaccessible areas without risking human life, are ideal platforms for such missions. Understanding lone pairs allows for the development of highly specialized drone payloads for these tasks.
For instance, many volatile organic compounds (VOCs) that are health hazards or contribute to atmospheric pollution possess lone pairs, influencing their vapor pressure, solubility, and reactivity. A drone equipped with a miniaturized mass spectrometer or a tunable diode laser absorption spectrometer (TDLAS) can detect and identify these compounds based on their unique mass-to-charge ratios or absorption spectra, respectively. Knowledge of how lone pairs affect bond strengths and molecular fragmentation patterns (relevant for mass spectrometry) or vibrational energy levels (relevant for TDLAS) guides the analysis of these complex spectral data. This chemical insight enables drone operators to swiftly identify unknown chemicals during a crisis, allowing for immediate and informed decision-making to mitigate hazards.
Innovation in Drone Payload Design: Leveraging Chemical Insights
The future of drone technology is intrinsically linked to its ability to perform increasingly complex and precise tasks. This evolution is driven by innovation in payload design, where a deep understanding of chemical principles, especially concerning lone pairs, is indispensable.
Optimizing Sensor Selectivity and Sensitivity for Specific Compounds
Developing chemical sensors for drone applications involves a constant pursuit of higher selectivity (the ability to detect a specific analyte without interference from others) and sensitivity (the ability to detect very low concentrations of an analyte). Lone pairs are a key consideration in this optimization process. For example, in the design of chemiresistive or electrochemical sensors, the sensing material’s surface properties are engineered to maximize specific interactions. If the target analyte contains a prominent lone pair, the sensor surface might be designed with complementary electrophilic sites to facilitate strong, selective binding. Conversely, if the target itself is electrophilic, the sensor material might present lone pair-rich sites.
This intricate dance of molecular recognition, guided by lone pair interactions, allows for the creation of smart sensors that can differentiate between closely related molecules. Imagine a drone system capable of not just detecting “a gas leak” but specifically identifying “methane,” “ethane,” or “propane” from their subtle differences in lone pair geometry or electron distribution, leading to much more targeted responses.
Future Trajectories: Autonomous Chemical Analysis and Manipulation
Looking ahead, the integration of AI and advanced chemical principles in drone technology promises revolutionary capabilities. Beyond mere detection, future drones might be able to perform autonomous chemical analysis, even limited manipulation or synthesis in situ. Imagine a drone not only identifying a specific contaminant but also autonomously deploying a targeted neutralizing agent, or collecting a precise chemical sample for laboratory analysis based on its understanding of the contaminant’s lone pair-driven reactivity.
Research into autonomous chemical laboratories on drones (lab-on-a-UAV) leverages knowledge of lone pairs for microfluidic control, selective reagent delivery, and real-time reaction monitoring. Furthermore, advances in drone-enabled remote chemical imaging, which visualizes the distribution of specific chemical species over an area, are heavily reliant on spectroscopic techniques that interpret signals influenced by lone pairs. As we delve deeper into the molecular world, the insights gained from understanding lone pairs will continue to unlock new frontiers for drone technology, transforming them into indispensable tools for scientific exploration, environmental protection, and industrial innovation.
