In the intricate world of cellular biology, the smooth and efficient functioning of life hinges on a vast array of chemical reactions. These reactions are the very essence of metabolism, powering everything from muscle contraction to DNA replication. However, many of these crucial biochemical transformations would proceed at an impractically slow pace, or not at all, without the assistance of specific non-protein molecules. These essential helpers are broadly classified as cofactors and coenzymes. Understanding their roles is fundamental to comprehending how enzymes, the protein catalysts of these reactions, achieve their remarkable efficiency and specificity.
The Crucial Role of Enzymes in Biological Reactions
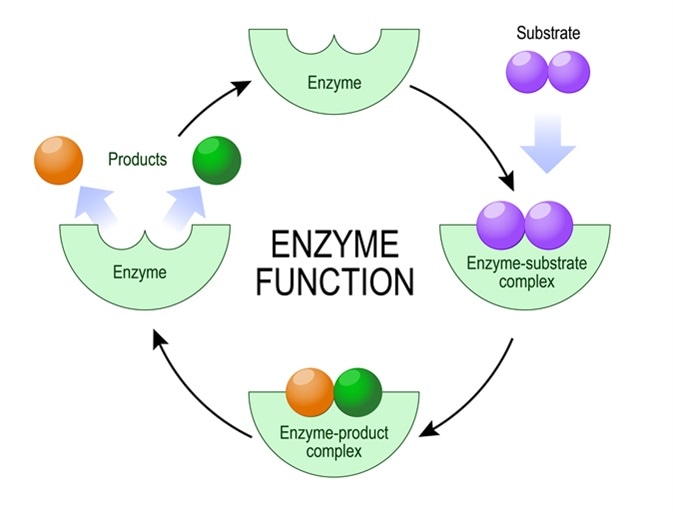
Enzymes are the workhorses of cellular biochemistry. These protein molecules possess a unique three-dimensional structure, including a specific region known as the active site. This active site is where the substrate, the molecule upon which the enzyme acts, binds. The binding of the substrate to the active site forms an enzyme-substrate complex, which facilitates the conversion of the substrate into product(s). Without enzymes, most biochemical reactions in living organisms would occur too slowly to sustain life. For instance, the breakdown of a single glucose molecule for energy would take an astronomically long time without the help of enzymes in cellular respiration.
The Need for Enzyme Efficiency and Specificity
Enzymes are not only catalysts but also highly specific. Each enzyme typically catalyzes only one or a very limited number of reactions. This specificity is crucial for maintaining order within the complex cellular environment, preventing unwanted side reactions and ensuring that metabolic pathways proceed in the correct sequence. The efficiency of enzymes is astounding; they can increase reaction rates by factors of millions or even billions compared to uncatalyzed reactions. This dramatic acceleration is achieved through various mechanisms, including lowering the activation energy of the reaction, orienting substrates correctly, and stabilizing the transition state.
Factors Influencing Enzyme Activity
While the protein structure of an enzyme is paramount, its catalytic prowess can be significantly influenced by its environment and the presence of other molecules. Factors such as temperature, pH, and substrate concentration all play a role in determining how effectively an enzyme functions. However, beyond these external conditions, a critical internal factor lies in the necessity of certain non-protein components for optimal activity. These components, cofactors and coenzymes, are not part of the enzyme’s polypeptide chain but are intimately involved in its catalytic process.
Defining Cofactors and Coenzymes: Essential Partners in Catalysis
At a fundamental level, cofactors and coenzymes are molecules that bind to enzymes and are essential for their catalytic activity. While often used interchangeably, there’s a subtle but important distinction between them based on their chemical nature. Both are vital for the proper functioning of many enzymes, acting as indispensable partners that enable these protein catalysts to perform their designated tasks.
Cofactors: The Broader Category
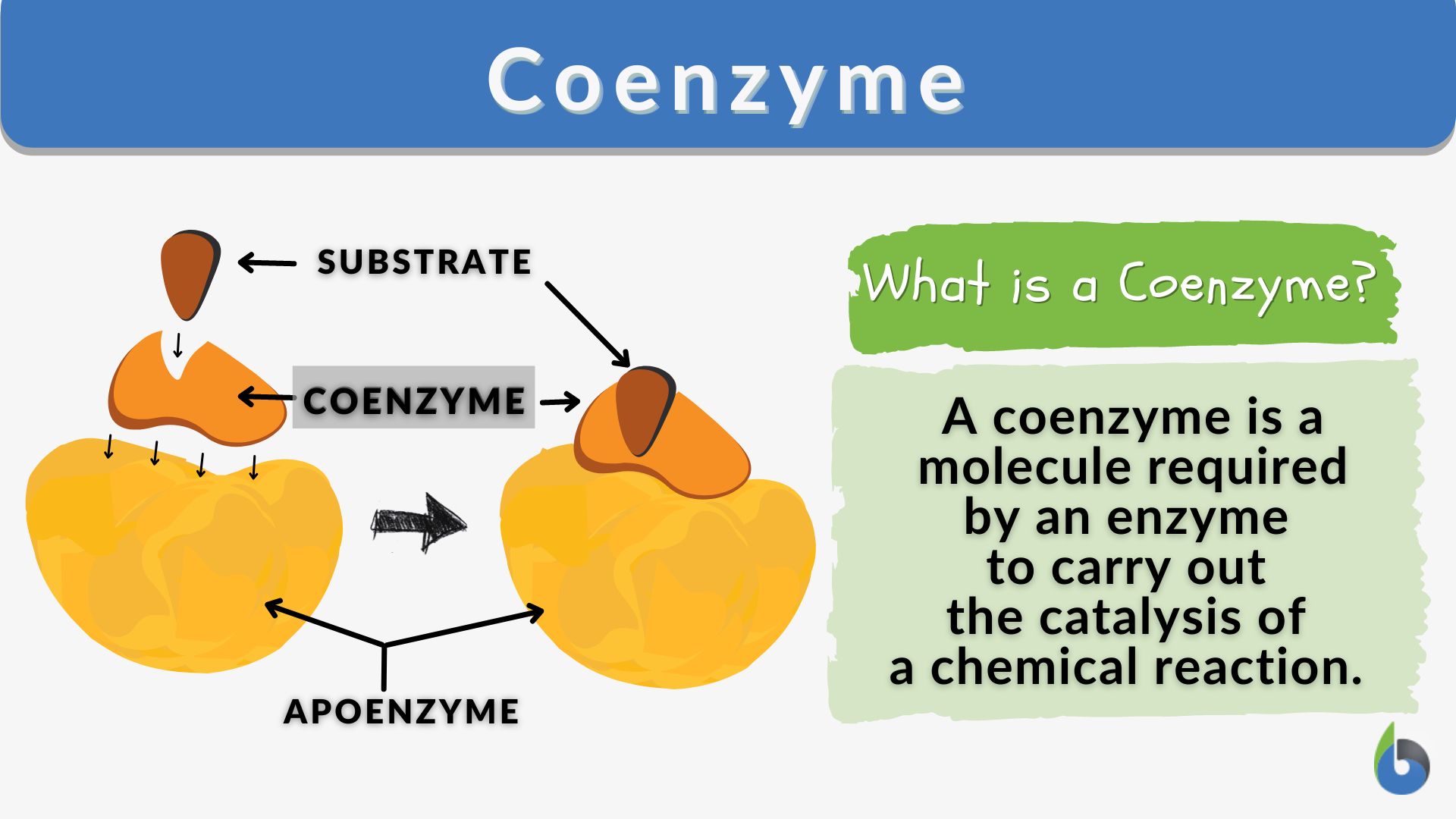
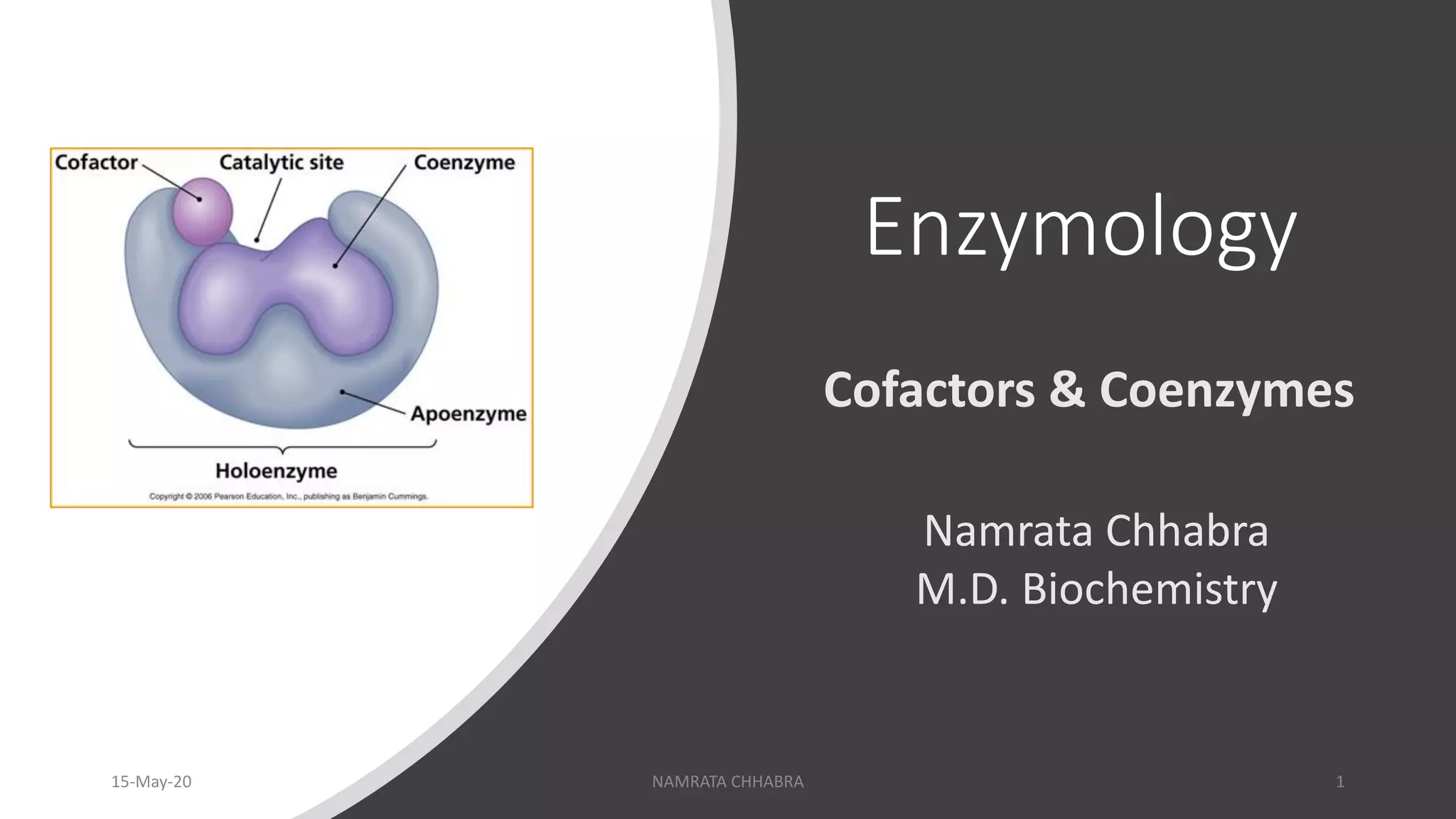
Cofactors represent the broader category of non-protein chemical compounds required for the biological activity of many enzymes. They can be inorganic ions or organic molecules. When an enzyme requires a cofactor to be active, it is referred to as an apoenzyme. The complete, active enzyme-cofactor complex is known as a holoenzyme. The cofactor essentially participates in the chemical reaction catalyzed by the enzyme, often by donating or accepting electrons, or by stabilizing a particular chemical intermediate.
Inorganic Cofactors: Metal Ions
Many enzymes require inorganic ions as cofactors. These metal ions, such as magnesium (Mg²⁺), zinc (Zn²⁺), iron (Fe²⁺/Fe³⁺), and copper (Cu²⁺), play diverse roles. For instance, Mg²⁺ is a common cofactor for enzymes involved in the transfer of phosphate groups, such as those in DNA polymerase and ATPases. It helps to stabilize the negatively charged phosphate groups of ATP, making it easier for the enzyme to bind and transfer them. Zinc ions are crucial for the activity of enzymes like carbonic anhydrase, which catalyzes the interconversion of carbon dioxide and water to bicarbonate and protons, and for alcohol dehydrogenase, which oxidizes alcohols. Iron is a vital component of cytochromes, proteins involved in electron transport chains, and of enzymes like catalase, which breaks down hydrogen peroxide.
Organic Cofactors: Coenzymes
The term “coenzyme” specifically refers to organic molecules that act as cofactors. Unlike inorganic ions, coenzymes are typically derived from vitamins, which are essential organic compounds that humans and many other animals cannot synthesize themselves and must obtain from their diet. These coenzymes often function as transient carriers of specific chemical groups, electrons, or atoms. They bind to the enzyme’s active site, participate in the catalytic step, and are then released, often unchanged, to participate in further catalytic cycles.
Coenzymes: The Organic Helpers
Coenzymes are arguably the most well-known type of cofactor. Their origins in vitamins underscore their importance in human health and nutrition, as deficiencies in these vitamins can lead to a cascade of metabolic problems due to the impaired function of coenzyme-dependent enzymes. The chemical structure of coenzymes is often complex, and they are designed to efficiently shuttle specific chemical moieties between different molecules during metabolic processes.
Examples of Essential Coenzymes and Their Vitamin Precursors
-
NAD⁺ and NADP⁺ (Nicotinamide Adenine Dinucleotide and Nicotinamide Adenine Dinucleotide Phosphate): These crucial coenzymes are derived from niacin (Vitamin B₃). They act as electron carriers in numerous oxidation-reduction reactions, particularly in cellular respiration and photosynthesis. NAD⁺ is primarily involved in catabolic pathways (breaking down molecules), while NADP⁺ is more commonly found in anabolic pathways (building up molecules), especially in lipid synthesis and the pentose phosphate pathway. In their reduced forms, NADH and NADPH, they carry high-energy electrons.
-
FAD and FMN (Flavin Adenine Dinucleotide and Flavin Mononucleotide): These coenzymes are derived from riboflavin (Vitamin B₂). Similar to NAD⁺/NADP⁺, FAD and FMN are also involved in redox reactions, serving as electron carriers. They are particularly important in the citric acid cycle and the electron transport chain, as well as in the breakdown of fatty acids. FAD can accept two electrons and two protons, forming FADH₂.
-
Coenzyme A (CoA): Derived from pantothenic acid (Vitamin B₅), CoA is a remarkably versatile coenzyme. Its primary role is to carry acyl groups, most notably the acetyl group (CH₃CO-). This makes it central to the metabolism of carbohydrates, fats, and amino acids. The formation of acetyl-CoA is a critical link between glycolysis and the citric acid cycle, and it also plays a key role in fatty acid synthesis and breakdown.
-
Thiamine Pyrophosphate (TPP): This coenzyme is derived from thiamine (Vitamin B₁). TPP is essential for enzymes that catalyze the decarboxylation of alpha-keto acids, such as pyruvate and alpha-ketoglutarate. This means it helps to remove a carboxyl group (-COOH) from these molecules, releasing carbon dioxide. It is vital for carbohydrate metabolism, particularly in the conversion of pyruvate to acetyl-CoA.
-
Pyridoxal Phosphate (PLP): Derived from pyridoxine (Vitamin B₆), PLP is a crucial coenzyme for enzymes involved in amino acid metabolism. It participates in a wide variety of reactions, including transamination (transferring amino groups), decarboxylation (removing carboxyl groups), and racemization (interconverting L- and D-amino acids).
-
Biotin: This vitamin acts as a coenzyme for enzymes involved in carboxylation reactions, meaning it helps to add a carboxyl group (-COOH) to a molecule. Biotin-dependent enzymes are key in the synthesis of fatty acids and in gluconeogenesis (the synthesis of glucose from non-carbohydrate sources).
-
Tetrahydrofolate (THF): Derived from folic acid (Vitamin B₉), THF is a vital coenzyme for carrying and transferring one-carbon units (like methyl, formyl, and methylene groups). This is essential for the synthesis of purines and pyrimidines (building blocks of DNA and RNA) and for the interconversion of amino acids.
-
Cobalamin (Vitamin B₁₂): Vitamin B₁₂ is a complex coenzyme involved in a limited number of crucial human reactions, including the conversion of methylmalonyl-CoA to succinyl-CoA (important for the metabolism of certain amino acids and fatty acids) and the remethylation of homocysteine to methionine.
How Cofactors and Coenzymes Function in Enzyme Mechanisms
The binding and function of cofactors and coenzymes are diverse and depend on the specific enzyme and reaction. Their presence often dictates the catalytic mechanism, enabling transformations that would otherwise be impossible. The interaction between the enzyme, coenzyme, and substrate is a finely tuned dance that drives cellular metabolism.
Tight Binding and Prosthetic Groups
Some cofactors and coenzymes bind very tightly to their respective enzymes. If the cofactor or coenzyme is permanently bound to the enzyme, it is referred to as a prosthetic group. These prosthetic groups are an integral part of the enzyme’s structure and are present throughout its catalytic cycle. For example, heme, an iron-containing porphyrin ring, is a prosthetic group in enzymes like cytochromes and catalase. FAD and FMN can also act as prosthetic groups in some flavoproteins.
Loosely Bound Coenzymes and Cosubstrates
Other coenzymes bind more loosely to the enzyme and are often referred to as cosubstrates. These coenzymes participate in the reaction as if they were a substrate, becoming chemically altered during the process and then released from the enzyme. NAD⁺, for example, binds to alcohol dehydrogenase, accepts a hydride ion (H⁻) from ethanol, and is released as NADH. Subsequently, another molecule of NAD⁺ can bind to the enzyme. This transient binding allows a single molecule of coenzyme to participate in numerous catalytic cycles with different enzyme molecules.
Catalytic Mechanisms Mediated by Cofactors and Coenzymes
The specific chemical roles of cofactors and coenzymes are incredibly varied. Metal ions can act as Lewis acids, polarizing bonds and stabilizing negative charges. They can also facilitate redox reactions by changing oxidation states. Coenzymes, with their diverse functional groups, are adept at carrying and transferring atoms or chemical groups. For instance, the reactive aldehyde group in TPP can participate in the formation of carbanions, which are key intermediates in decarboxylation reactions. The amide nitrogen in NAD⁺ and NADP⁺ is responsible for accepting hydride ions, enabling the transfer of electrons. CoA’s sulfur atom is highly nucleophilic and readily forms thioester bonds, making it an excellent carrier of acyl groups.
The Significance of Cofactors and Coenzymes in Health and Disease
The indispensable nature of cofactors and coenzymes is starkly illustrated when their availability is compromised or when their function is impaired. Deficiencies in essential vitamins, the precursors to many coenzymes, lead to specific deficiency diseases, highlighting the critical role these molecules play in maintaining physiological health.
Vitamin Deficiencies and Associated Diseases
As mentioned earlier, many coenzymes are derived from vitamins. A lack of these vitamins in the diet can lead to serious health consequences. Scurvy, caused by a deficiency in Vitamin C (ascorbic acid), impairs collagen synthesis because Vitamin C is a cofactor for enzymes involved in this process. Beriberi results from a lack of thiamine (Vitamin B₁), impacting carbohydrate metabolism and leading to neurological and cardiovascular problems. Pellagra, a deficiency in niacin (Vitamin B₃), is characterized by skin rashes, diarrhea, and dementia, all linked to the impaired function of NAD⁺ and NADP⁺ dependent enzymes. Pernicious anemia is caused by a deficiency in Vitamin B₁₂, affecting DNA synthesis and red blood cell production.
Genetic Disorders Affecting Cofactor Metabolism
Beyond dietary deficiencies, genetic disorders can also disrupt the intricate pathways involved in the synthesis, transport, or utilization of cofactors and coenzymes. For example, certain rare genetic conditions can lead to enzyme deficiencies that prevent the proper activation or binding of specific coenzymes, leading to severe metabolic imbalances and health issues. These disorders underscore the highly regulated and essential nature of these biochemical partnerships.
Therapeutic Implications and Dietary Recommendations
Understanding the roles of cofactors and coenzymes has profound implications for medicine and nutrition. Supplementation with specific vitamins or minerals can correct deficiencies and alleviate associated diseases. Furthermore, knowledge of cofactor requirements can inform strategies for drug development, as some drugs work by inhibiting enzymes that rely on specific cofactors. Dietary recommendations are often guided by the need to ensure adequate intake of essential vitamins and minerals that serve as precursors for these vital coenzymes.
In conclusion, cofactors and coenzymes are not merely passive bystanders in biochemical reactions. They are active, essential partners that enable enzymes to perform their catalytic duties with remarkable speed and precision. From inorganic metal ions to complex vitamin-derived organic molecules, these helpers are fundamental to the very processes of life. Their intricate interplay with enzymes forms the bedrock of cellular metabolism, and their proper functioning is critical for maintaining health and preventing disease.
