While the title “What is Xanax Schedule?” might initially suggest a topic outside of our usual purview, a deeper dive into the technological and societal implications of controlled substances, particularly as they relate to their distribution, monitoring, and the potential for illicit markets, brings us into the fascinating intersection of Tech & Innovation. Specifically, understanding drug scheduling and its impact can inform the development of sophisticated technological solutions for tracking, authentication, and even the prevention of misuse. This exploration delves into how advancements in data management, blockchain technology, and secure digital identities can play a crucial role in regulating and understanding the “schedule” of substances like Xanax.
Understanding Drug Scheduling and its Implications
Drug scheduling is a critical component of regulatory frameworks designed to control and monitor substances with potential for abuse or dependence. Xanax, a brand name for alprazolam, is a benzodiazepine medication widely prescribed for anxiety and panic disorders. Its classification within a specific schedule is not arbitrary; it’s based on a comprehensive assessment of its medical utility, potential for abuse, and the likelihood of causing physical or psychological dependence.
The Controlled Substances Act (CSA) and Drug Scheduling
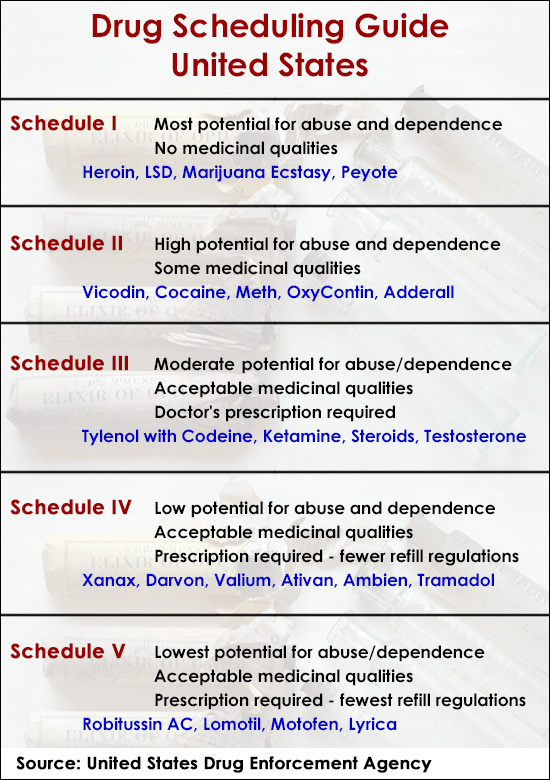
In the United States, the Controlled Substances Act (CSA) of 1970 is the federal drug policy that categorizes psychoactive drugs into five distinct schedules. This categorization is determined by the Drug Enforcement Administration (DEA) in conjunction with the Food and Drug Administration (FDA). The scheduling dictates the level of control placed on the manufacture, possession, and distribution of these substances.
Schedule I Drugs
Drugs with no currently accepted medical use and a high potential for abuse. Examples include heroin, LSD, and marijuana (though its scheduling is a subject of ongoing debate and legislative action).
Schedule II Drugs
Drugs with a high potential for abuse, which may lead to severe psychological or physical dependence. These drugs have a currently accepted medical use. Examples include cocaine, methamphetamine, and strong opioid painkillers like oxycodone and fentanyl.
Schedule III Drugs
Drugs with a moderate to low potential for physical and psychological dependence. These drugs have a currently accepted medical use. Examples include ketamine, anabolic steroids, and certain prescription drugs containing less than 15 milligrams of hydrocodone per dosage unit.
Schedule IV Drugs
Drugs with a low potential for abuse and a low risk of physical and psychological dependence. These drugs have a currently accepted medical use. Examples include Xanax (alprazolam), Valium (diazepam), and Ambien (zolpidem).
Schedule V Drugs
Drugs with the lowest potential for abuse among the scheduled substances. They contain limited quantities of certain stimulants or depressants and have a currently accepted medical use. Examples include cough preparations containing codeine.
Xanax’s Classification: Schedule IV
Xanax, or alprazolam, falls under Schedule IV of the Controlled Substances Act. This classification signifies that while it possesses a recognized medical utility and is used to treat legitimate health conditions, it still carries a risk of abuse and can lead to psychological or physical dependence with prolonged use. This “schedule” designation has profound implications for how Xanax is prescribed, dispensed, and regulated. It necessitates stricter prescription requirements, limits on refills, and increased scrutiny of dispensing practices compared to non-scheduled medications.
Technological Innovations in Drug Monitoring and Control
The “schedule” of a drug like Xanax, while a legal and medical classification, presents significant challenges for effective monitoring and control. The potential for diversion, illicit manufacturing, and misuse necessitates robust technological solutions. Modern advancements in various fields offer promising avenues for addressing these challenges, moving beyond traditional paper-based tracking and manual oversight.
Blockchain Technology for Supply Chain Integrity
The inherent vulnerabilities in traditional pharmaceutical supply chains, from manufacturing to patient dispensing, create opportunities for counterfeit drugs and diversion. Blockchain technology offers a decentralized, immutable ledger that can revolutionize supply chain management for controlled substances.
Secure and Transparent Tracking
Each step of a drug’s journey – from its synthesis and packaging to its shipment and final dispensing – can be recorded as a transaction on a blockchain. This creates a transparent and tamper-proof record, making it incredibly difficult to introduce counterfeit products or divert legitimate ones without detection. For Schedule IV drugs like Xanax, this could significantly reduce the risk of diversion into the illicit market.
Smart Contracts for Automated Compliance
Smart contracts, self-executing contracts with the terms of the agreement directly written into code, can be deployed on blockchain platforms. These can automate compliance with regulatory requirements. For instance, a smart contract could be programmed to release a batch of Xanax for dispensing only after specific verification steps are met, such as authorized prescription validation and pharmacist confirmation.
Enhanced Traceability and Recall Management
In the event of a recall or a need to investigate a specific batch, blockchain provides immediate and granular traceability. Authorities and manufacturers can quickly identify the exact origin and distribution path of a particular drug, streamlining recall processes and improving public safety.
Advanced Data Analytics and AI for Anomaly Detection
The sheer volume of data generated by pharmaceutical transactions, prescriptions, and dispensing patterns presents an opportunity for sophisticated analysis. Artificial intelligence (AI) and advanced data analytics can be leveraged to identify anomalous patterns that might indicate diversion or misuse.
Predictive Analytics for Abuse Potential
By analyzing historical data on prescription patterns, patient demographics, and geographical trends, AI algorithms can predict areas or patient groups with a higher risk of Xanax abuse. This allows for proactive intervention and targeted monitoring.
Real-time Monitoring of Dispensing Patterns
AI can monitor real-time dispensing data across pharmacies and healthcare providers. Deviations from established norms, such as unusually high prescribing rates for a particular physician or excessive refills for certain patients, can trigger alerts for further investigation. This is particularly relevant for Schedule IV drugs where dependence can develop over time.
Identifying Illicit Market Indicators
AI can also be trained to identify patterns indicative of the illicit market, such as unusual price fluctuations, online sales of prescription drugs without proper authorization, or the emergence of counterfeit packaging.
Secure Digital Identity and Prescription Management
The integrity of the prescription process is paramount for controlled substances. Secure digital identity solutions and advanced prescription management systems can bolster this integrity.
Biometric Authentication for Prescribers and Patients
Implementing biometric authentication (e.g., fingerprint or facial recognition) for healthcare professionals issuing prescriptions and for patients picking them up adds a significant layer of security. This prevents unauthorized prescription writing and ensures that the medication is dispensed to the intended recipient.
Encrypted and Verified Digital Prescriptions
Moving towards fully encrypted and digitally signed prescriptions can eliminate the risk of forged paper prescriptions. These digital prescriptions can be securely transmitted directly to pharmacies, with verification mechanisms in place to confirm their authenticity and prevent tampering.
Patient Prescription Monitoring Programs (PMPs) Enhancement
Existing Prescription Drug Monitoring Programs (PDMPs) are valuable tools. Integrating them with advanced data analytics and AI can enhance their effectiveness. Real-time data entry and cross-state data sharing, facilitated by robust technological infrastructure, are crucial for preventing “doctor shopping” and prescription fraud for medications like Xanax.
The Evolving Landscape of Controlled Substance Regulation
The “schedule” of Xanax is a dynamic designation, subject to review and potential reclassification based on evolving scientific understanding and societal impact. Technological advancements are not just tools for enforcement but also integral to this evolving landscape.
Research and Development in Alternative Treatments
While Xanax is an effective medication, research continues into alternative treatments for anxiety and panic disorders that may have lower dependence potential. Innovations in areas like digital therapeutics, behavioral therapies delivered through apps, and non-addictive pharmacological agents are constantly emerging. The “schedule” of a drug can influence the economic incentives for developing such alternatives.
International Cooperation and Data Harmonization
The illicit drug trade is a global issue. Technological solutions for tracking and monitoring controlled substances must also consider international cooperation. Harmonizing data standards and developing interoperable systems across different countries is essential for combating cross-border diversion and illicit trafficking. Blockchain and cloud-based data solutions can facilitate this global interoperability.
Ethical Considerations and Privacy
As technology becomes more integrated into drug monitoring, ethical considerations, particularly regarding patient privacy, become paramount. Implementing robust data anonymization techniques, secure data storage, and clear guidelines on data access and usage are critical. The goal is to enhance public safety and regulatory compliance without infringing on individual rights. The development of privacy-preserving technologies within AI and blockchain is a key area of innovation.

Conclusion
The “schedule” of Xanax, as a Schedule IV controlled substance, underscores the delicate balance between therapeutic benefit and the potential for harm. While the classification itself is a regulatory construct, its practical implications necessitate a continuous evolution of control mechanisms. The realm of Tech & Innovation is no longer an ancillary support but a central pillar in addressing these challenges. From the immutable transparency offered by blockchain to the predictive power of AI and the enhanced security of digital identities, technology is providing unprecedented tools to monitor, regulate, and ultimately safeguard the distribution and use of controlled substances. As our understanding of these substances and the technologies to manage them grows, the future of drug scheduling and control will undoubtedly be shaped by further innovation, aiming for a safer and more secure pharmaceutical landscape for all.
