Understanding the fundamental differences between heat and temperature is crucial for anyone working with or even passively experiencing technologies that rely on thermal principles. While often used interchangeably in everyday language, these two concepts represent distinct physical phenomena. In the realm of advanced technology, particularly in fields like remote sensing, imaging, and even the operational parameters of sophisticated electronics, a precise grasp of thermal dynamics is not just academic; it’s essential for performance, safety, and innovation. This article will demystify heat and temperature, exploring their scientific definitions and their critical relevance across various technological domains.
The Microscopic Dance: Temperature as Average Kinetic Energy
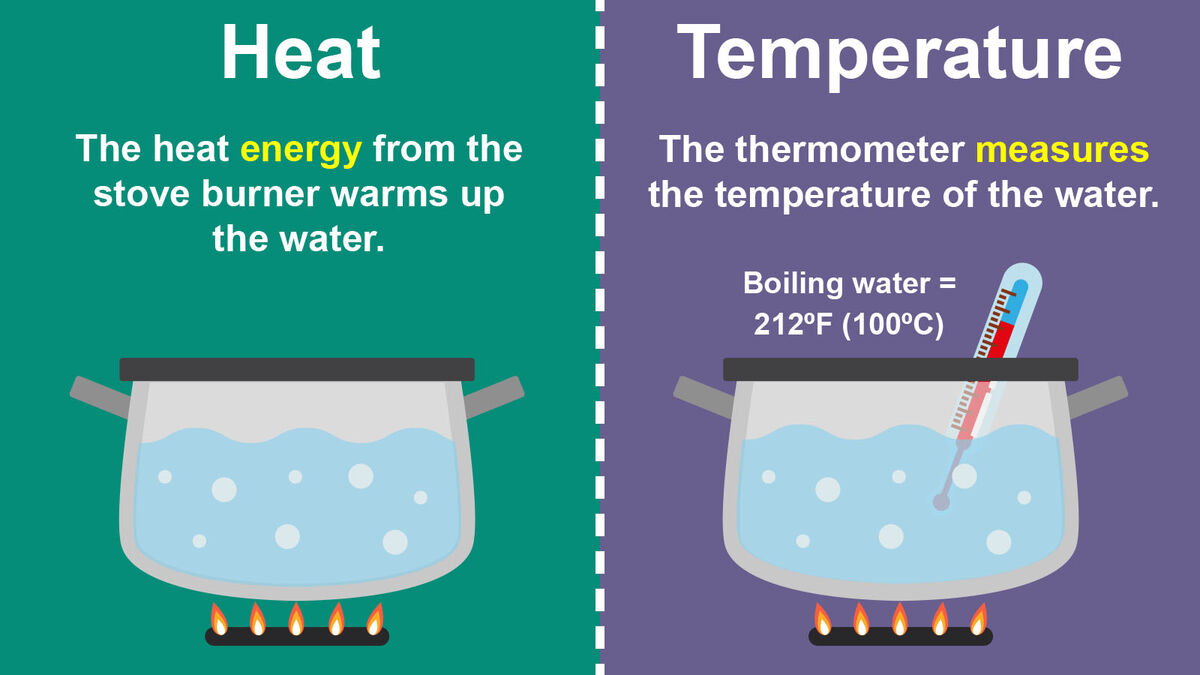
At its core, temperature is a measure of the average kinetic energy of the particles within a substance. Imagine a vast collection of tiny entities – atoms or molecules – constantly in motion. They vibrate, they rotate, and in gases and liquids, they translate (move from place to place). This ceaseless movement is what gives matter its internal energy. Temperature quantifies this average motion.
Kinetic Energy and Molecular Motion
The faster these particles move, the higher their average kinetic energy, and consequently, the higher the temperature. Conversely, when particles move slower, their kinetic energy decreases, and the temperature drops. This relationship is direct and linear, as described by the kinetic theory of gases. For example, when we heat a metal object, we are essentially adding energy to its atoms, causing them to vibrate more vigorously. This increased vibration translates directly to a higher temperature reading.
Absolute Zero and the Kelvin Scale
The concept of absolute zero is a theoretical temperature at which all molecular motion ceases. This fundamental limit is designated as 0 Kelvin (0 K) on the Kelvin scale, which is the standard scientific unit for temperature. The Celsius and Fahrenheit scales are also commonly used, but the Kelvin scale, being an absolute scale, avoids negative values and directly relates to the total absence of thermal energy.
- Kelvin (K): The SI unit, where 0 K is absolute zero. Water freezes at 273.15 K and boils at 373.15 K.
- Celsius (°C): Commonly used, with 0°C as the freezing point of water and 100°C as its boiling point at standard atmospheric pressure.
- Fahrenheit (°F): Primarily used in the United States, with 32°F as the freezing point of water and 212°F as its boiling point.
The conversion formulas are:
$K = °C + 273.15$
$°C = (K – 273.15)$
$°C = (°F – 32) times 5/9$
$°F = (°C times 9/5) + 32$
Understanding these scales is vital for calibrating sensors, interpreting data from thermal imaging, and ensuring that electronic components operate within their specified thermal envelopes.
The Flow of Energy: Heat as Energy Transfer
While temperature describes the state of a system’s internal energy in terms of average kinetic energy, heat is the transfer of that thermal energy from a region of higher temperature to a region of lower temperature. Heat is energy in transit, a process, not a property of a substance itself. A substance possesses internal energy, which is related to its temperature, but it does not possess heat.
Mechanisms of Heat Transfer
Heat can be transferred through three primary mechanisms: conduction, convection, and radiation.
- Conduction: The transfer of heat through direct contact between particles. In solids, this occurs as vibrating atoms collide with their neighbors, passing on kinetic energy. In metals, free electrons also play a significant role in conducting heat. This is why a metal spoon placed in hot soup becomes hot.
- Convection: The transfer of heat through the movement of fluids (liquids or gases). When a fluid is heated, it becomes less dense and rises, while cooler, denser fluid sinks, creating a circulatory current that distributes heat. This is how a radiator heats a room or how weather patterns form.
- Radiation: The transfer of heat through electromagnetic waves. Unlike conduction and convection, radiation does not require a medium and can travel through a vacuum. The most common example is the sun’s energy reaching Earth. All objects with a temperature above absolute zero emit thermal radiation.
Quantifying Heat: Joules and Calories
The standard unit of energy in the International System of Units (SI) is the Joule (J). Heat transfer is measured in Joules. Other units, like the calorie (cal) and the British Thermal Unit (BTU), are also used. A calorie is defined as the amount of heat required to raise the temperature of one gram of water by one degree Celsius.
The amount of heat transferred is dependent on three factors:
- The temperature difference: A larger temperature difference leads to a faster rate of heat transfer.
- The thermal conductivity of the materials involved: Materials with high thermal conductivity transfer heat more readily.
- The surface area of contact: A larger surface area facilitates greater heat transfer.
The Interplay in Technological Applications
The distinction between heat and temperature is paramount in various technological fields. Consider how these concepts are applied:
Thermal Imaging and Remote Sensing
Thermal imaging cameras, often found on drones for applications like industrial inspection, search and rescue, and environmental monitoring, detect infrared radiation emitted by objects. The intensity of this radiation is directly related to the object’s temperature. However, the camera is not measuring “heat” directly; it’s measuring the radiation that results from the object’s internal thermal energy. Interpreting these images requires understanding that a warmer object emits more infrared radiation, indicating a higher temperature and a greater tendency to transfer heat to its surroundings.
For instance, when inspecting power lines with a thermal camera, a hot spot (higher temperature) might indicate an impending failure due to increased electrical resistance, which generates more heat. Similarly, in a search and rescue operation, a person’s body heat will stand out against a cooler background, allowing for their detection via thermal signatures. The effectiveness of these systems relies on precise temperature readings and the ability to infer heat loss or gain based on these measurements.
Electronics and Thermal Management
Modern electronic devices, from smartphones to high-performance computing systems, generate significant amounts of heat during operation. This heat is a byproduct of electrical resistance as electrons flow through circuits. If not managed effectively, this heat can lead to component failure, reduced performance, and even fire hazards.
Thermal management strategies are employed to dissipate this heat, preventing the internal temperature from exceeding safe operating limits. This involves using heat sinks (materials with high thermal conductivity to draw heat away), fans (to promote convective cooling), and thermal paste (to improve heat transfer between components and heat sinks). The goal is to maintain the internal components at a temperature that allows for optimal and safe operation. Understanding the difference between the heat generated (energy transfer) and the resulting temperature of the components is crucial for designing effective cooling solutions.
Aerospace and Flight Technology
In aerospace, thermal management is critical for the reliable operation of various systems. Engines, avionics, and even the airframe itself are subject to extreme temperature variations.
- Engine Performance: The combustion process in jet engines generates immense heat. Maintaining optimal operating temperatures is essential for thrust, efficiency, and longevity. Conversely, extreme cold at high altitudes can affect fuel viscosity and the performance of hydraulic systems.
- Avionics: Electronic components in aircraft must operate within specific temperature ranges. Heat generated by onboard computers and communication systems needs to be dissipated efficiently to prevent malfunctions.
- Aerodynamic Heating: At high speeds, friction with the air can cause significant heating of an aircraft’s surfaces, particularly its leading edges. This phenomenon, known as aerodynamic heating, must be accounted for in aircraft design and material selection.
The temperature of an aircraft’s components directly impacts their structural integrity and operational capabilities. Understanding heat transfer mechanisms is vital for designing systems that can withstand and manage these thermal stresses, ensuring safe and efficient flight.
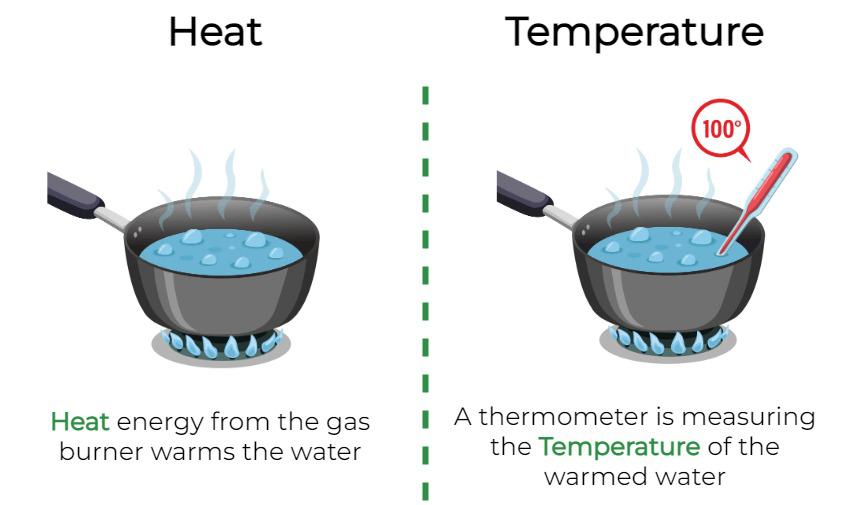
Conclusion: A Unified Understanding
While temperature is a measure of the average kinetic energy of particles within a system, and heat is the transfer of thermal energy between systems due to a temperature difference, these concepts are inextricably linked. One describes a state, while the other describes a process. In the ever-evolving landscape of technology, from the sensitive optics of advanced cameras to the complex thermal dynamics of flight systems, a nuanced understanding of both heat and temperature is not merely beneficial; it is fundamental to innovation, efficiency, and safety. Recognizing their distinct roles allows engineers, scientists, and enthusiasts alike to design, analyze, and interact with the world of thermal phenomena more effectively.
