Ozempic, a brand name for semaglutide, represents a significant advancement in the treatment of type 2 diabetes and, more recently, in weight management. Beyond its clinical applications, the development and efficacy of Ozempic are deeply rooted in sophisticated advancements within the realm of pharmaceutical technology and biological innovation. This article delves into the technological underpinnings of Ozempic, exploring its mechanism of action, the innovation behind its development, and its broader impact within the evolving landscape of medical technology.
The Molecular Innovation: Understanding GLP-1 Receptor Agonists
At its core, Ozempic’s revolutionary impact stems from its intelligent design as a glucagon-like peptide-1 (GLP-1) receptor agonist. This class of drugs mimics the action of a natural hormone produced in the gut, which plays a crucial role in regulating blood sugar and appetite. The innovation lies not only in identifying this biological pathway but in the meticulous engineering of a molecule that can effectively and sustainably interact with it.
Mimicking Natural Hormones: The Science of GLP-1
Glucagon-like peptide-1 (GLP-1) is an incretin hormone secreted by enteroendocrine L-cells in the small intestine in response to nutrient intake. Its physiological functions are multifaceted and directly relevant to metabolic health. GLP-1 plays a critical role in glucose homeostasis by:
- Stimulating insulin secretion: GLP-1 enhances the release of insulin from pancreatic beta-cells in a glucose-dependent manner. This means it prompts the pancreas to release more insulin when blood glucose levels are high, and less when they are low, thereby reducing the risk of hypoglycemia.
- Suppressing glucagon secretion: Glucagon is a hormone that raises blood glucose levels by promoting the release of stored glucose from the liver. GLP-1 inhibits glucagon secretion, further contributing to lower blood sugar levels.
- Slowing gastric emptying: By delaying the rate at which food leaves the stomach, GLP-1 helps to reduce post-meal blood glucose spikes and promotes feelings of fullness.
- Promoting satiety and reducing appetite: GLP-1 acts on the brain to signal satiety, leading to a reduction in food intake and, consequently, aiding in weight management.
The challenge for pharmaceutical innovation was to create a molecule that could leverage these beneficial effects of GLP-1 but overcome its inherent limitations. Natural GLP-1 has a very short half-life in the body, being rapidly degraded by an enzyme called dipeptidyl peptidase-4 (DPP-4) and cleared by the kidneys. This short duration of action would necessitate frequent injections, making it impractical for long-term therapeutic use.
Engineering Longevity: The Semaglutide Advantage
Semaglutide, the active ingredient in Ozempic, is a modified GLP-1 analog. The technological ingenuity lies in its structural modifications that confer a significantly longer duration of action. Key modifications include:
- Amino acid substitutions: Specific amino acids in the GLP-1 sequence are replaced with others that confer resistance to DPP-4 degradation. This is a common strategy in developing DPP-4 resistant peptides.
- Acylation with a fatty acid chain: A crucial innovation in semaglutide’s design is the attachment of a fatty acid derivative (stearoyl-glutamic acid) to the molecule. This acylation allows semaglutide to bind to albumin, a protein abundant in the bloodstream. This binding forms a complex that shields the semaglutide molecule from enzymatic breakdown and reduces its renal clearance. The result is a significantly extended half-life, allowing for once-weekly administration.
This sophisticated molecular engineering represents a prime example of how advancements in medicinal chemistry and protein engineering can translate into groundbreaking therapeutic solutions. The ability to design and synthesize such complex molecules with precise pharmacological profiles is a testament to the power of applied science and technological innovation.
The Application of Advanced Delivery Systems
Beyond the molecule itself, the effectiveness and patient experience of Ozempic are also enhanced by innovative drug delivery systems. The development of injectable formulations that are stable, easy to administer, and designed for long-term use has been crucial to its widespread adoption and clinical success.
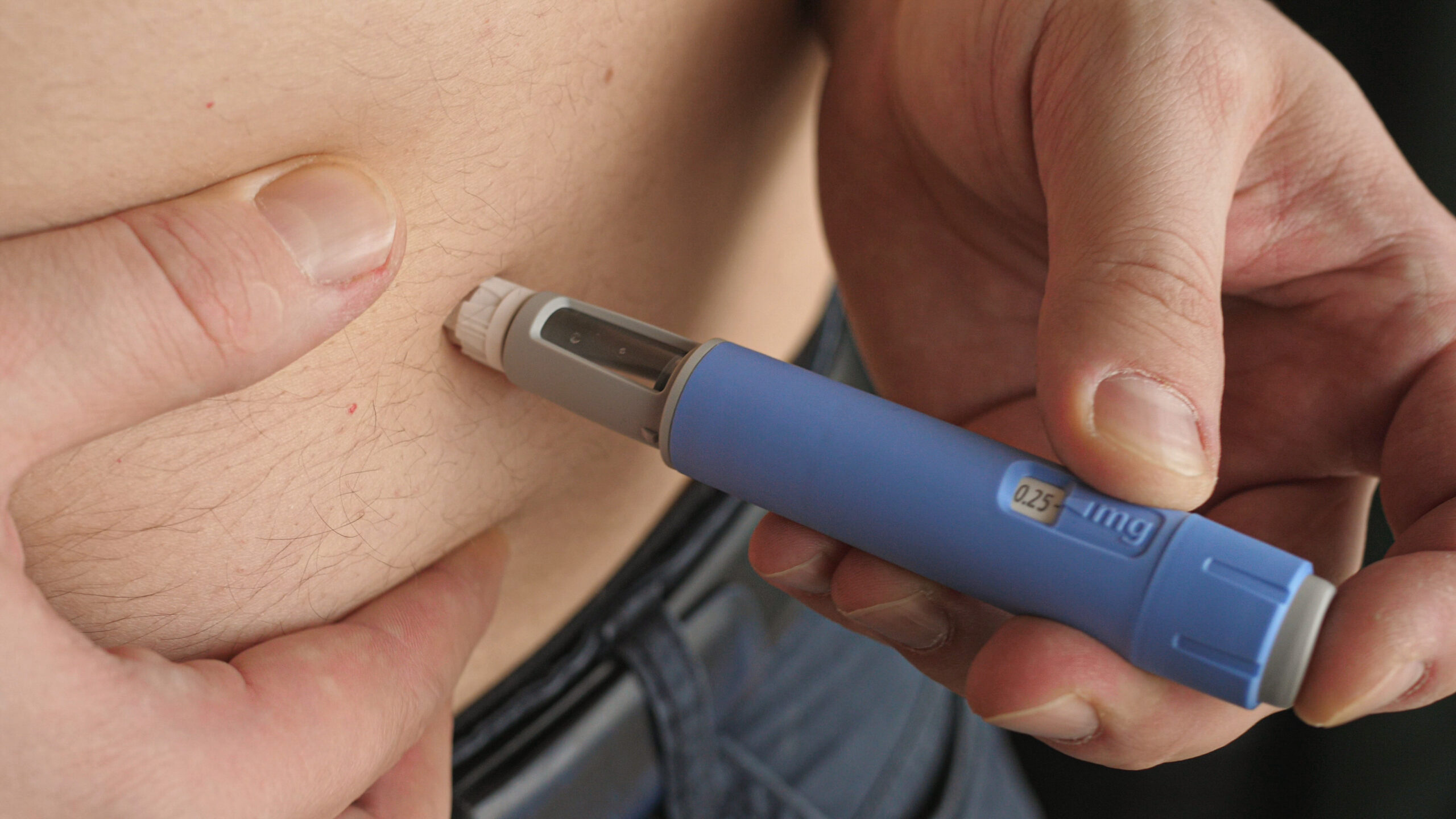
Precision Injection: The Pen Device Technology
Ozempic is typically administered via a pre-filled injection pen. This delivery system itself embodies technological advancement designed for user convenience and therapeutic precision.

- Pre-filled cartridges: These cartridges contain a precise dose of the medication, eliminating the need for manual measurement and reducing the risk of dosing errors. This is a significant improvement over traditional vials and syringes.
- Adjustable dose dials: The pen devices allow users to select their prescribed dose with a simple turning mechanism, ensuring accuracy. This is particularly important as patients may require dose titration.
- Integrated needle system: The needles are typically very fine and short, designed for subcutaneous injection with minimal discomfort. The pen design often incorporates features to ensure the needle is properly attached and to provide audible or tactile feedback during injection.
- Discreet and portable: The pen format makes the medication easy to carry and administer discreetly, which can be a significant factor for patients managing chronic conditions.
The design and manufacturing of these injection pens require advanced polymer science, precision engineering, and sterile manufacturing processes. The goal is to create a device that is not only functional but also intuitive and empowering for patients, encouraging adherence to treatment regimens.
Beyond Subcutaneous: Exploring Future Delivery Modalities
While the pen remains the primary delivery method, ongoing research and technological innovation are exploring other potential avenues for GLP-1 receptor agonists like semaglutide. This includes the development of oral formulations, which have faced significant challenges due to the peptide nature of GLP-1 and its susceptibility to degradation in the gastrointestinal tract.
The development of an oral formulation of semaglutide (Rybelsus) represents a major technological breakthrough. This was achieved through the use of an absorption enhancer, specifically sodium N-[8-(2-hydroxybenzoyl)amino]caprylate (SNAC). SNAC facilitates the passage of semaglutide across the intestinal wall and protects it from enzymatic degradation in the stomach. This innovation has opened up new possibilities for administering peptide-based therapeutics, potentially reducing the burden of daily injections for patients.
The Broader Technological and Societal Impact
The success of Ozempic extends beyond its immediate clinical benefits, highlighting broader trends in technological innovation within healthcare and its societal implications.
The Rise of Personalized Medicine and Targeted Therapies
Ozempic is a prime example of a targeted therapy. Its mechanism of action is precisely tailored to a specific biological pathway, offering a more personalized approach to managing type 2 diabetes and obesity compared to some traditional broad-spectrum treatments. This trend towards highly specific, mechanism-based interventions is a hallmark of modern pharmaceutical innovation. The ability to understand complex biological systems at a molecular level and then design drugs to selectively modulate them represents a significant leap forward.
Data-Driven Drug Development and Real-World Evidence
The development and ongoing monitoring of drugs like Ozempic are increasingly reliant on advanced data analytics and the collection of real-world evidence (RWE). Clinical trials, while rigorous, are controlled environments. To understand the true impact and long-term effectiveness of a drug, researchers analyze data from electronic health records, insurance claims, and patient-reported outcomes. This data-driven approach allows for:
- Refinement of treatment protocols: Identifying optimal dosing strategies and patient populations that benefit most.
- Detection of rare side effects: Uncovering less common adverse events that may not be apparent in smaller clinical trials.
- Understanding long-term efficacy: Assessing how the drug performs in diverse populations over extended periods.
This integration of data science into the drug lifecycle is transforming how medications are developed, approved, and utilized.
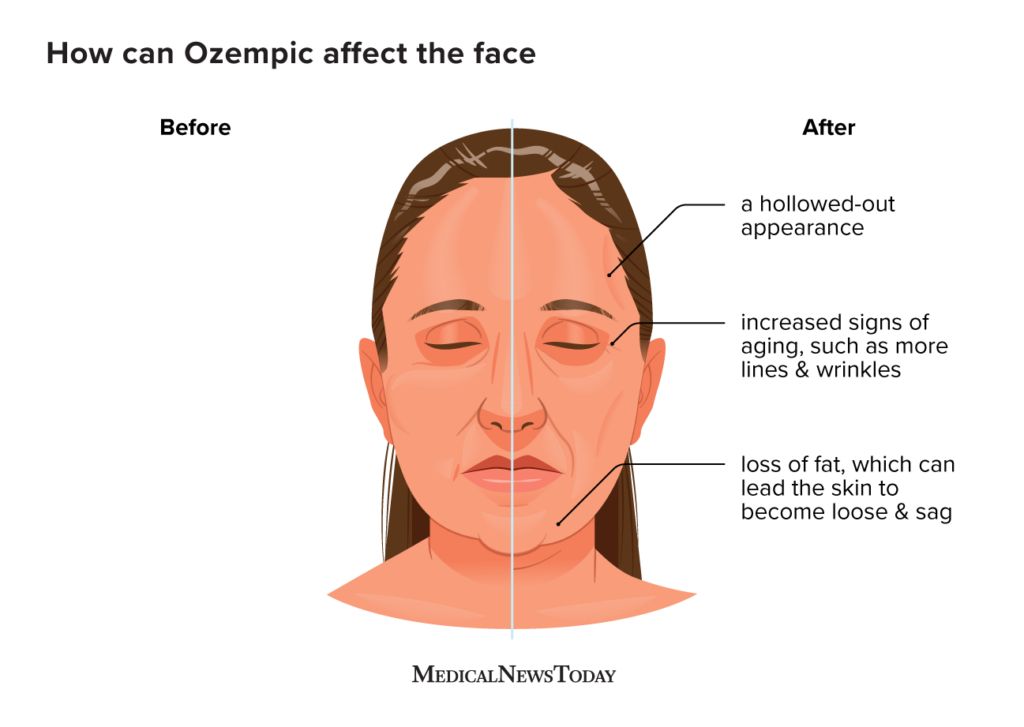
The Evolving Landscape of Weight Management
The approval and subsequent widespread use of Ozempic for weight management have sparked significant discussion and highlighted the technological advancements in addressing obesity. For decades, obesity has been treated with a combination of diet, exercise, and some medications with limited efficacy and significant side effects. The efficacy of semaglutide in promoting substantial weight loss represents a paradigm shift, driven by a deeper understanding of the neurobiological pathways that regulate appetite and energy balance. This success is paving the way for further innovation in the development of next-generation weight management therapies.
However, this technological advancement also brings societal challenges, including issues of access, affordability, and the ethical considerations surrounding the use of powerful medications for aesthetic or non-life-threatening conditions. The technological prowess that brings these innovations to fruition must be matched by thoughtful societal and ethical frameworks.
In conclusion, Ozempic is more than just a medication; it is a product of sophisticated technological innovation. From the intricate molecular engineering of semaglutide to the user-friendly delivery systems, its development embodies the cutting edge of pharmaceutical science and engineering. As research continues, we can anticipate further advancements driven by the same spirit of innovation, promising new frontiers in the treatment of metabolic diseases and beyond.
