Adenosine Triphosphate (ATP) is not merely a molecule; it is the universal energy currency of life. Its seemingly simple structure belies its profound importance, underpinning virtually every cellular process that requires energy. From the microscopic twitch of a muscle fiber to the complex biochemical reactions that drive thought, ATP is the power source. Understanding its molecular nature is key to appreciating its role in biological systems and, by extension, to sparking innovation in fields that seek to harness or replicate biological efficiency.
The Molecular Architecture of ATP
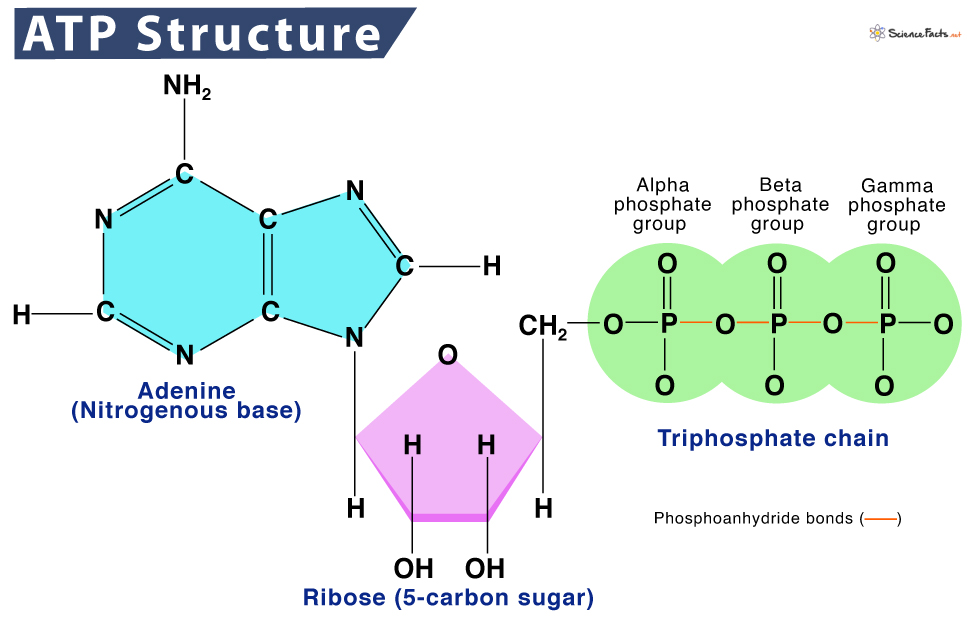
ATP’s significance stems directly from its unique chemical structure, which allows it to store and release energy efficiently. This structure can be broken down into three main components: adenine, ribose, and a triphosphate chain.

Adenine: The Nitrogenous Base
Adenine is a purine, a type of nitrogenous base characterized by a double-ring structure. In ATP, this adenine molecule is attached to a ribose sugar. Adenine is a fundamental building block of nucleic acids, DNA and RNA, highlighting ATP’s connection to the genetic machinery of the cell. Its presence in ATP serves as a recognition site for enzymes and other cellular components involved in energy transfer. The specific arrangement and chemical properties of adenine are crucial for ATP’s ability to interact with various biological catalysts and energy-harnessing machinery within the cell.
Ribose: The Five-Carbon Sugar
Ribose is a five-carbon sugar, classifying it as a pentose. It forms the central part of the ATP molecule, linking the adenine base to the triphosphate chain. Ribose is also a component of RNA, further emphasizing the close relationship between energy metabolism and genetic information processing. The hydroxyl (-OH) groups on the ribose sugar are involved in forming the ester bonds that attach the phosphate groups. The orientation and configuration of the ribose molecule are essential for the correct positioning of the phosphate groups, which is critical for energy release.
The Triphosphate Chain: The Powerhouse of Energy
The defining feature of ATP is its chain of three phosphate groups. These phosphate groups are linked together by high-energy phosphodiester bonds. It is within these bonds, particularly the bonds connecting the second and third phosphate groups (the beta and gamma phosphates, respectively), that the energy is stored. When ATP is hydrolyzed – meaning a water molecule is used to break one of these bonds – a significant amount of energy is released, converting ATP into Adenosine Diphosphate (ADP) and an inorganic phosphate ion (Pi).
- Beta-Phosphate: This bond, between the alpha and beta phosphates, is also high-energy, and its cleavage yields ADP and Pi.
- Gamma-Phosphate: The bond between the beta and gamma phosphates is the most frequently cleaved in cellular energy transfer. Its hydrolysis releases the largest amount of readily usable energy, transforming ATP into ADP and inorganic phosphate. The release of energy is a result of the electrostatic repulsion between the negatively charged phosphate groups and the inherent instability of the strained bond.
The sequential removal of phosphate groups from ATP can also occur. While ADP can be further hydrolyzed to Adenosine Monophosphate (AMP) and another inorganic phosphate, this process releases less energy and is less common for direct cellular work. The primary cycle of energy currency involves the interconversion of ATP and ADP.
ATP Synthesis: Recharging the Energy Currency
The continuous demand for energy necessitates a constant supply of ATP. This ATP is synthesized through various metabolic pathways, with the primary goal being the reattachment of a phosphate group to ADP. This process requires an input of energy, which is harvested from the breakdown of nutrients or from light energy.
Oxidative Phosphorylation: The Major ATP Producer
The vast majority of ATP produced in aerobic organisms is generated through oxidative phosphorylation. This complex process occurs within the mitochondria, the powerhouses of the cell. It involves a series of electron transfer reactions along the electron transport chain (ETC), ultimately leading to the pumping of protons (H+) across the inner mitochondrial membrane. This creates an electrochemical gradient, a form of potential energy.
- Electron Transport Chain (ETC): Specialized protein complexes embedded in the inner mitochondrial membrane accept high-energy electrons, primarily from NADH and FADH2 (electron carriers generated during the breakdown of glucose and other fuel molecules). As electrons move through the chain, energy is released.
- Proton Gradient: The released energy is used to actively pump protons from the mitochondrial matrix into the intermembrane space, creating a high concentration of protons in the intermembrane space compared to the matrix.
- ATP Synthase: This remarkable molecular machine acts like a tiny turbine. Protons flow back into the matrix down their concentration gradient through a channel in ATP synthase. This flow of protons drives the rotation of a part of ATP synthase, providing the mechanical energy needed to catalyze the synthesis of ATP from ADP and Pi. This is a prime example of chemiosmosis, where a chemical gradient is converted into mechanical and then chemical energy.
Substrate-Level Phosphorylation: A Direct Energy Transfer
While oxidative phosphorylation is the most prolific ATP producer, substrate-level phosphorylation provides a more direct method of ATP synthesis. This process occurs in the cytoplasm during glycolysis and in the mitochondrial matrix during the Krebs cycle. In substrate-level phosphorylation, a high-energy phosphate group is directly transferred from a substrate molecule to ADP, forming ATP. This reaction is catalyzed by specific enzymes.
- Glycolysis: This initial stage of cellular respiration breaks down glucose into pyruvate, producing a net gain of ATP through substrate-level phosphorylation.
- Krebs Cycle (Citric Acid Cycle): Further breakdown of pyruvate derivatives in the mitochondria generates ATP via substrate-level phosphorylation, along with electron carriers for oxidative phosphorylation.
Photophosphorylation: ATP from Light
In photosynthetic organisms (plants, algae, and some bacteria), ATP is also generated using light energy. This process, called photophosphorylation, occurs within chloroplasts. Light energy is captured by pigments like chlorophyll and used to excite electrons. These energized electrons then move through an electron transport chain, similar in principle to oxidative phosphorylation, generating a proton gradient across the thylakoid membrane. This gradient drives ATP synthase to produce ATP. This remarkable ability to convert light energy into chemical energy is a cornerstone of most ecosystems on Earth, directly or indirectly providing the energy that fuels life.
The Role of ATP in Cellular Functions and Bio-Inspired Innovation
ATP’s role as the universal energy currency means it is indispensable for a vast array of cellular activities. Its efficient storage and release of energy have inspired numerous avenues of scientific inquiry, including those that might eventually impact technological innovation.
Driving Cellular Processes
Every energy-requiring process within a cell relies on ATP. This includes:
- Muscle Contraction: The sliding of actin and myosin filaments in muscle cells is powered by ATP hydrolysis, enabling movement.
- Active Transport: Moving molecules across cell membranes against their concentration gradients requires energy supplied by ATP. This is crucial for nutrient uptake and waste removal.
- Biosynthesis: The synthesis of complex molecules like proteins, nucleic acids, and lipids from simpler precursors demands significant energy input from ATP.
- Nerve Impulse Transmission: The establishment and maintenance of ion gradients across nerve cell membranes, which are essential for transmitting electrical signals, are ATP-dependent.
- Cell Signaling: Many signaling pathways that regulate cellular behavior involve ATP or its derivatives, and the energy expenditure for signal transduction is significant.

Potential for Bio-Inspired Technologies
While ATP itself is not directly incorporated into technological devices, the principles of its function and the efficiency of biological energy transfer are subjects of intense research that can inform technological development.
- Energy Storage and Conversion: Understanding how biological systems, particularly mitochondria, achieve such high efficiency in energy conversion and storage could inspire novel approaches to battery design and energy harvesting technologies. Bio-mimicking the electron transport chain or ATP synthase could lead to more efficient and sustainable energy solutions.
- Biomimetic Materials: The self-assembly and energy transfer mechanisms inherent in ATP-dependent biological processes could inspire the creation of new smart materials capable of responding to stimuli or performing complex functions with minimal external energy input.
- Sustainable Energy Generation: The principles of photophosphorylation, which convert light into chemical energy, are foundational to developing more efficient solar energy technologies. Furthermore, understanding biological catalysts involved in ATP synthesis could lead to improved biofuel production or other bio-based energy generation methods.
- Micro-Robotics and Nanotechnology: The miniaturization and efficiency of cellular energy use are aspirational goals for micro-robotics and nanotechnology. If scientists can replicate the energy management strategies of biological cells, it could enable smaller, more autonomous, and longer-lasting microscopic devices.
In conclusion, ATP is a nucleoside triphosphate, a molecule whose structure of adenine, ribose, and a triphosphate chain allows it to serve as the primary energy currency of all known life. Its synthesis through pathways like oxidative phosphorylation, substrate-level phosphorylation, and photophosphorylation ensures a continuous supply of energy for cellular work. While ATP itself is a biological entity, the fundamental principles of its energy storage and release, and the efficiency of biological energy transfer systems, offer a rich source of inspiration for advancing various fields of technological innovation, pushing the boundaries of what is possible in energy, materials science, and beyond.
