The question of “what sugars are reducing sugars” delves into a fundamental aspect of carbohydrate chemistry, with significant implications across various scientific disciplines. While the direct application to the world of drones, flight technology, cameras, drone accessories, aerial filmmaking, or tech and innovation might not be immediately obvious, a closer examination reveals fascinating connections, particularly within the realm of Tech & Innovation, specifically in areas like advanced sensor development and the sophisticated analysis of biological and environmental data.
Reducing sugars are defined by their ability to act as a reducing agent. This property stems from the presence of a free aldehyde or ketone functional group in their open-chain form, which can readily donate electrons to another molecule. This characteristic is not merely an abstract chemical concept; it forms the basis of numerous analytical techniques that can be, and are, integrated into advanced technological systems, including those used in drone-based applications.
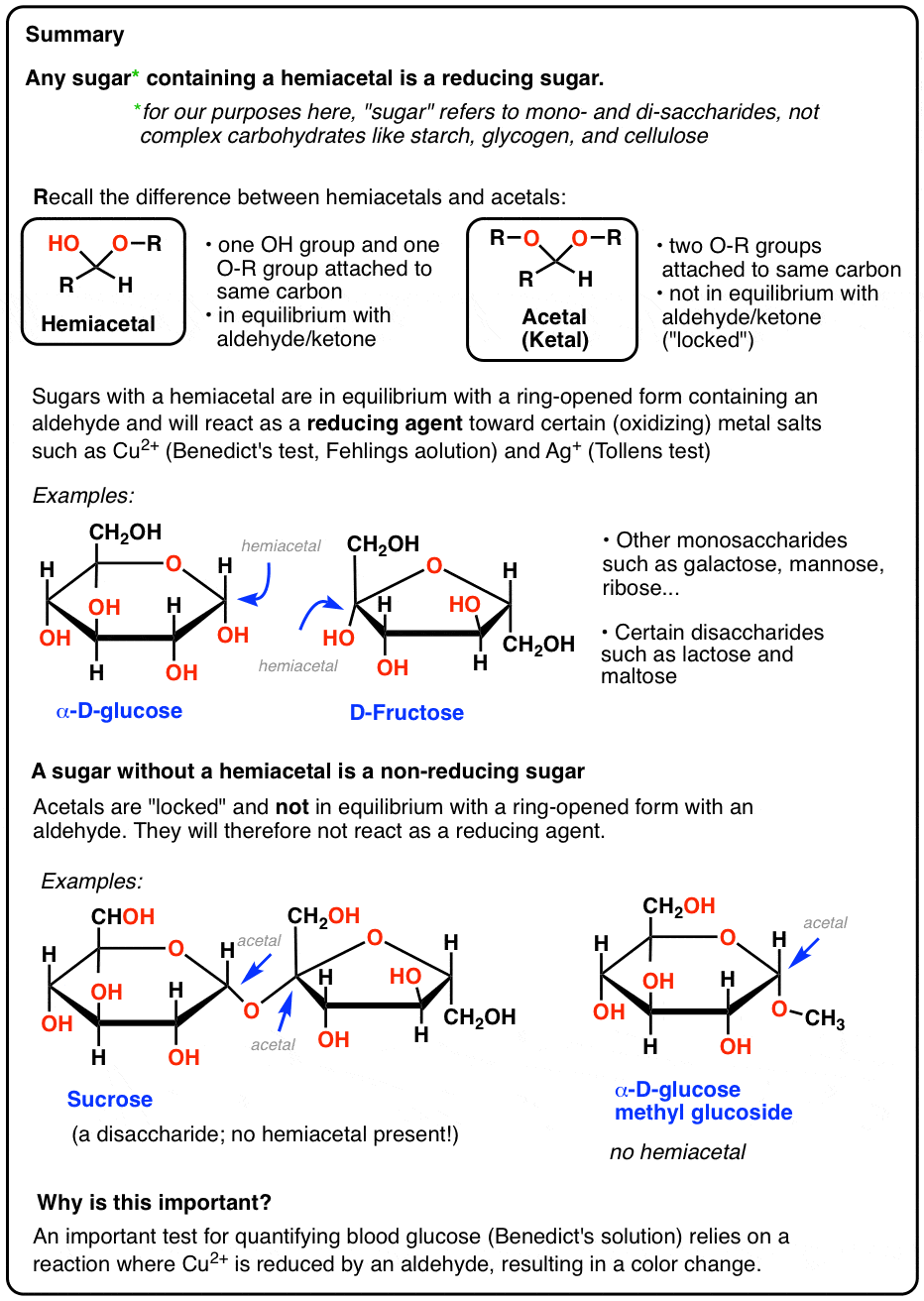
The Chemical Basis of Reducing Sugars
To understand which sugars are reducing sugars, we must first grasp the underlying chemical structure and reactivity. Sugars, or carbohydrates, are broadly classified into monosaccharides and polysaccharides. The critical distinction for reducing sugar status lies within the monosaccharides and the disaccharides formed from them.
Monosaccharides: The Foundation of Reduction
Monosaccharides are the simplest form of sugars, unable to be hydrolyzed into smaller carbohydrate units. They exist in a cyclic hemiacetal or hemiketal form in solution, but they can also exist in an open-chain form that possesses either an aldehyde group (aldoses) or a ketone group (ketoses). It is this free aldehyde or ketone group that gives them their reducing properties.
Aldoses: The Ubiquitous Reducing Sugars
All aldoses, regardless of their chain length, are reducing sugars. This is because they possess a terminal aldehyde group in their open-chain structure. When these sugars cyclize, they form a hemiacetal. The anomeric carbon, which was the original carbonyl carbon, still retains a hydrogen atom and an –OH group, allowing for the ring to open and close, thus exposing the aldehyde group and enabling it to act as a reducing agent.
- Glucose: Perhaps the most well-known monosaccharide, glucose is an aldohexose. Its cyclic form can revert to the open-chain aldehyde form, making it a potent reducing sugar. This property is vital in biological systems, influencing metabolic pathways and serving as a key indicator in various diagnostic tests.
- Galactose: Another aldohexose, galactose, is structurally similar to glucose. It also possesses a free aldehyde group in its open-chain form and is therefore a reducing sugar.
- Ribose and Deoxyribose: These are aldopentoses, crucial components of RNA and DNA, respectively. Both have free aldehyde groups in their open-chain forms, classifying them as reducing sugars.
Ketoses: A Slightly Different Mechanism
Ketoses are monosaccharides that contain a ketone functional group. In their open-chain form, they possess a ketone group. When they cyclize, they form a hemiketal. Similar to aldoses, the hemiketal can also open to reveal the ketone group, allowing it to act as a reducing agent.
- Fructose: Fructose, an aldohexose, is a common ketose. While its cyclic form is a hemiketal, it readily interconverts with its open-chain form, which contains a ketone group. This allows fructose to also act as a reducing sugar.
Disaccharides: A Tale of Two Bonds
Disaccharides are formed when two monosaccharide units are joined together by a glycosidic bond. Whether a disaccharide is a reducing sugar depends on the nature of this bond and whether it involves the anomeric carbon of one or both monosaccharides.
Reducing Disaccharides: One Free Anomeric Carbon
For a disaccharide to be a reducing sugar, at least one of its constituent monosaccharides must retain a free anomeric carbon, meaning it’s not involved in the glycosidic linkage. This free anomeric carbon can open to reveal an aldehyde or ketone group, enabling the disaccharide to act as a reducing agent.
- Lactose: This disaccharide, composed of glucose and galactose, is a classic example of a reducing sugar. The glycosidic bond connects the anomeric carbon of galactose to the fourth carbon of glucose. This leaves the anomeric carbon of glucose free, allowing it to revert to its open-chain aldehyde form and exhibit reducing properties.
- Maltose: Also known as malt sugar, maltose is composed of two glucose units linked by an $alpha(1 to 4)$ glycosidic bond. The anomeric carbon of one glucose unit is involved in the linkage, but the anomeric carbon of the other glucose unit remains free. Thus, maltose is a reducing sugar.
Non-Reducing Disaccharides: No Free Anomeric Carbon
If both anomeric carbons of the constituent monosaccharides are involved in the glycosidic linkage, then neither can open to reveal an aldehyde or ketone group. Such disaccharides are not reducing sugars.
- Sucrose: Commonly known as table sugar, sucrose is formed from glucose and fructose. The glycosidic bond in sucrose links the anomeric carbon of glucose to the anomeric carbon of fructose. Since both anomeric carbons are involved, sucrose does not have a free aldehyde or ketone group and is therefore a non-reducing sugar.
Polysaccharides: Generally Non-Reducing
Polysaccharides are long chains of monosaccharide units linked by glycosidic bonds. While they are formed from reducing sugars, the vast majority of internal glycosidic bonds are between an anomeric carbon and a hydroxyl group on another monosaccharide. This means that there are typically very few, if any, free anomeric carbons available for reduction.
- Starch and Glycogen: These are polymers of glucose. While the terminal glucose unit on each chain has a free anomeric carbon and can act as a reducing agent, their contribution is negligible in comparison to the sheer number of non-reducing internal linkages. Thus, for practical purposes, starch and glycogen are considered non-reducing.
- Cellulose: Also a polymer of glucose, cellulose is linked by $beta(1 to 4)$ glycosidic bonds. Similar to starch and glycogen, the terminal glucose units might possess some reducing potential, but the overall polysaccharide is generally classified as non-reducing.
The Significance of Reducing Sugars in Analytical Technology
The ability of reducing sugars to donate electrons is not just a chemical curiosity; it forms the bedrock of numerous analytical tests and technologies that are increasingly integrated into various scientific and industrial applications, including those relevant to advanced tech.
Benedict’s Test and Fehling’s Test: Classic Analytical Approaches
Historically, the identification and quantification of reducing sugars relied on qualitative and quantitative chemical tests that exploit their reducing properties.
Benedict’s Test
Benedict’s test uses a solution containing copper(II) sulfate, sodium carbonate, and sodium citrate. When heated with a reducing sugar, the copper(II) ions ($Cu^{2+}$) are reduced to copper(I) ions ($Cu^{+}$), which precipitate as copper(I) oxide ($Cu_2O$). This precipitate is brick-red in color, indicating the presence of a reducing sugar. The intensity of the color is proportional to the concentration of the reducing sugar, allowing for semi-quantitative analysis.
Fehling’s Test
Fehling’s test is similar in principle to Benedict’s test, also utilizing a copper(II) complex that is reduced by reducing sugars to form a brick-red precipitate of copper(I) oxide. Fehling’s solution consists of two parts: Fehling’s solution A (copper(II) sulfate) and Fehling’s solution B (alkaline solution of potassium sodium tartrate). These are mixed just before use. Like Benedict’s test, it provides a visual indication of reducing sugar presence and can be adapted for quantitative measurements.
Modern Applications and Technological Integration
While Benedict’s and Fehling’s tests are foundational, the underlying principle of redox reactions involving reducing sugars has been adapted and miniaturized for more sophisticated analytical platforms. These advancements have direct implications for the development of intelligent systems and sensors that might be deployed in diverse technological fields.
Biosensors and Electrochemical Detection
The ability of reducing sugars to participate in redox reactions makes them ideal candidates for detection via electrochemical biosensors. In such systems, an enzyme that specifically reacts with a particular reducing sugar (e.g., glucose oxidase for glucose) is immobilized onto an electrode. The enzymatic reaction produces a substance that is electrochemically active, or the sugar itself might be directly oxidized electrochemically.
- Glucose Meters: The most ubiquitous example is the modern glucose meter used by individuals with diabetes. These devices employ electrochemical sensors that quantify blood glucose levels by measuring the current produced from the oxidation of glucose, a reducing sugar. This technology has seen remarkable miniaturization and integration, paving the way for non-invasive or minimally invasive sensing.
- Enzyme-Linked Assays: While not strictly redox-based, many enzyme-linked assays for carbohydrate analysis utilize the enzymatic breakdown of sugars, which can be coupled to downstream reactions that produce a measurable signal, often involving colorimetric or electrochemical detection.
Spectrophotometric Analysis and Imaging
The colorimetric changes observed in classic tests like Benedict’s and Fehling’s can be measured precisely using spectrophotometers. This allows for highly accurate quantification of reducing sugar concentrations.
- Automated Analyzers: In laboratory settings, automated analyzers utilize spectrophotometric principles to process large numbers of samples rapidly, determining reducing sugar levels in various matrices.
- Potential for Drone-Based Environmental Monitoring: Imagine a drone equipped with advanced sensor packages. If the objective is to monitor water quality for specific agricultural pollutants or biological markers, and if certain indicators involve changes in reducing sugar levels within a sample, then miniaturized electrochemical or optical sensors capable of detecting these changes could be integrated. This could allow for in-situ analysis of water bodies without the need for physical sample collection and laboratory processing.
Reducing Sugars in the Context of Advanced Technology
The chemistry of reducing sugars, while seemingly esoteric, underpins technologies that are increasingly sophisticated and integrated into our daily lives, particularly within the domains of cutting-edge innovation.
Precision Agriculture and Food Quality Control
In sectors like agriculture and food production, the ability to accurately measure sugar content is crucial for determining ripeness, quality, and shelf-life.
- Crop Health Monitoring: Changes in the metabolic profile of plants, including sugar levels, can be indicative of stress, disease, or nutrient deficiencies. Advanced sensors, potentially integrated into agricultural drones, could be developed to non-destructively assess crop health by detecting subtle changes in leaf or fruit surface chemistry, which might correlate with altered reducing sugar concentrations.
- Food Processing and Quality Assurance: In food manufacturing, maintaining consistent sugar profiles is vital for taste, texture, and stability. Real-time monitoring of reducing sugars during processing can ensure product quality and safety. This could involve inline sensors within processing plants that leverage electrochemical or optical detection principles derived from reducing sugar chemistry.
Biomedical Diagnostics and Research
The role of reducing sugars, particularly glucose, in human health is paramount. The analytical methods developed for their detection have revolutionized medical diagnostics.
- Diabetes Management: As mentioned, glucose meters are indispensable tools. Future innovations might involve wearable sensors or implantable devices that continuously monitor glucose levels, providing real-time feedback and enabling more precise management of diabetes.
- Disease Biomarkers: Beyond glucose, other reducing sugars or their metabolic byproducts can serve as biomarkers for various diseases. The development of sensitive and specific sensors for these compounds, enabled by understanding their chemical properties, is an ongoing area of research with immense potential for early disease detection.
Environmental Sensing and Bio-Monitoring
The application of reducing sugar detection extends to environmental monitoring, where biological processes often involve these compounds.
- Wastewater Treatment Monitoring: Biological wastewater treatment processes rely on microbial activity, which involves the metabolism of various organic compounds, including sugars. Sensors that can detect specific reducing sugars could provide insights into the efficiency of these processes or indicate the presence of particular organic loads.
- Ecological Studies: In aquatic or soil ecosystems, the concentration of reducing sugars can be an indicator of microbial activity, decomposition rates, or the availability of nutrients. Drones equipped with specialized sensors could potentially be used for large-scale, non-invasive ecological surveys, mapping areas with high or low reducing sugar indicators to understand ecosystem health.
In conclusion, the question of “what sugars are reducing sugars” opens a window into a fundamental aspect of carbohydrate chemistry. This understanding is not confined to academic laboratories; it forms the crucial scientific basis for a wide array of analytical technologies that are increasingly being integrated into advanced systems. From sophisticated biosensors to potential applications in environmental monitoring and precision agriculture, the ability to detect and quantify reducing sugars continues to drive innovation and enhance our capacity to understand and interact with the complex world around us.
