The persistent and often unpleasant odor of urine can be a significant nuisance, whether it originates from pets, accidents in the home, or even certain environmental factors. Understanding the chemical composition of urine and the mechanisms behind its odor is the first step to effectively neutralizing it. This article delves into the science behind urine odor and explores various neutralization strategies, focusing on the underlying chemical principles and their practical applications.
The Chemistry of Urine Odor
Urine is primarily composed of water, but it also contains dissolved waste products from metabolic processes, most notably urea. The characteristic smell of fresh urine is typically mild and often described as slightly ammoniacal. However, as urine ages or when exposed to certain conditions, its odor can intensify and change dramatically. This transformation is largely due to bacterial decomposition.

Urea and its Decomposition
Urea (CO(NH₂)₂) is a nitrogenous waste product produced by the liver as a byproduct of protein metabolism. In fresh urine, urea is relatively odorless. However, when bacteria, commonly found in the environment, come into contact with urine, they begin to break down the urea through an enzymatic process called hydrolysis. The enzyme responsible for this is urease, which is produced by many types of bacteria.
The hydrolysis of urea results in the production of ammonia (NH₃) and carbon dioxide (CO₂):
CO(NH₂)₂ + H₂O → 2NH₃ + CO₂
Ammonia is a volatile gas with a pungent, sharp odor. The higher the concentration of ammonia produced, the stronger and more offensive the urine smell becomes. This is why old, dried urine stains are often much more potent than fresh ones.
Other Odorous Compounds
While ammonia is the primary culprit behind the strong urine odor, other compounds can also contribute. These include:
- Mercaptans: Sulfur-containing compounds that can produce foul odors, often described as rotten egg or garlic-like. These can be formed from the breakdown of amino acids present in urine.
- Ketones: In certain metabolic conditions, such as diabetes or during periods of starvation, the body may produce ketones, which can impart a fruity or acetone-like smell to urine.
- Certain foods and medications: Consuming foods like asparagus or certain medications can lead to specific compounds being excreted in urine, causing a temporary, distinct odor.
The interplay of these compounds, especially ammonia and mercaptans, creates the complex and often unpleasant olfactory experience associated with aged urine.
Principles of Odor Neutralization
Neutralizing odor involves either masking the smell, absorbing it, or chemically altering the odor-causing molecules to render them odorless. Effective urine odor neutralization strategies often employ a combination of these principles, targeting the root causes of the smell.
Chemical Neutralization
Chemical neutralizers work by reacting with the odor-causing molecules to transform them into odorless substances. This is often the most effective approach for persistent odors.
Acid-Base Reactions
Ammonia is a basic compound. Therefore, acidic substances can effectively neutralize ammonia through an acid-base reaction. When an acid reacts with ammonia, it forms an ammonium salt, which is typically non-volatile and odorless.
A common example of this is the reaction between ammonia and acetic acid (vinegar):
NH₃ + CH₃COOH → NH₄⁺CH₃COO⁻ (Ammonium acetate)
Ammonium acetate is a salt that does not possess the pungent smell of ammonia. Similarly, other weak acids can be used. However, strong acids should be used with caution as they can be corrosive and may damage surfaces.
Oxidation
Oxidation is another powerful method for breaking down odor-causing molecules. Oxidizing agents chemically react with organic compounds, disrupting their molecular structure and converting them into less volatile and odorless substances like carbon dioxide and water.
-
Hydrogen Peroxide (H₂O₂): A common household oxidizing agent, hydrogen peroxide can effectively break down many organic compounds responsible for urine odor. It decomposes into water and oxygen, leaving no harmful residues. When used on urine stains, it can also help to break down the organic matter that feeds bacteria, further preventing future odor development.
The reaction can be complex but generally involves the oxidation of sulfur-containing compounds and the breakdown of other organic molecules.
-
Ozone (O₃): Ozone is a highly reactive form of oxygen and a potent oxidizing agent. While not typically used in household applications due to its gaseous nature and potential health hazards, ozone generators are used in commercial settings for deep odor elimination. Ozone molecules readily react with and break down odor-causing organic compounds.
-
Potassium Permanganate (KMnO₄): This is a strong oxidizing agent that can be used in diluted solutions. It reacts with many organic compounds, including those that cause urine odor. However, it can stain surfaces purple, so its use is often limited to specific applications and requires careful dilution.
Absorption and Adsorption
Absorption and adsorption involve trapping odor molecules within a porous material, effectively removing them from the air.
-
Activated Carbon: Activated carbon is a highly porous material with a vast surface area. Its porous structure allows it to adsorb (bind to the surface) a wide range of molecules, including those responsible for urine odor. This makes it an effective deodorizer, often found in air filters and odor-absorbing products.
-
Baking Soda (Sodium Bicarbonate – NaHCO₃): While primarily known for its mild alkalinity and ability to absorb moisture, baking soda also has some odor-absorbing properties. It can react with acidic odor compounds and absorb some volatile organic compounds. Its effectiveness is moderate compared to activated carbon but it is a safe and readily available option.
Enzymatic Breakdown
Enzymatic cleaners utilize specific enzymes to break down the organic molecules in urine, such as urea, proteins, and fats, into simpler, odorless compounds. These are particularly effective for biological stains and odors.
-
Urease: As mentioned earlier, urease breaks down urea into ammonia and carbon dioxide. Enzymatic cleaners often contain urease or similar enzymes to target the initial breakdown of urea, preventing the formation of strong ammonia smells.
-
Protease: Enzymes like protease break down proteins into amino acids.
-
Lipase: Enzymes like lipase break down fats.
By breaking down the various components of urine, enzymatic cleaners effectively eliminate the source of the odor and prevent its recurrence. They are considered a safe and environmentally friendly option for dealing with pet stains and other biological odors.
Practical Applications and Solutions
Based on the chemical principles discussed, several practical methods and products can be employed to neutralize urine odor.

Household Remedies
For immediate or minor odor issues, common household items can offer effective solutions.
Vinegar
White vinegar is an acidic solution that can neutralize the alkaline ammonia present in urine.
- Method: Mix equal parts white vinegar and water. Spray the solution liberally onto the affected area, let it sit for 10-15 minutes, and then blot it up with a clean cloth. For older or dried stains, a stronger concentration or repeated applications may be necessary. Ensure the area is well-ventilated during use.
Baking Soda
Baking soda is a versatile deodorizer that can be used in several ways.
- Method 1 (Dry application): Sprinkle a generous amount of baking soda directly onto the dry urine stain or area. Allow it to sit for several hours or overnight to absorb odors. Then, vacuum it up thoroughly.
- Method 2 (Paste): Mix baking soda with a small amount of water to form a paste. Apply the paste to the stain, let it dry, and then brush or vacuum it away.
Hydrogen Peroxide
A 3% hydrogen peroxide solution can be effective for breaking down odor-causing compounds.
- Method: After cleaning the urine stain with soap and water, lightly mist the area with a 3% hydrogen peroxide solution. Do not saturate the area, as peroxide can sometimes bleach fabrics. Test in an inconspicuous spot first. Allow it to air dry. This is particularly effective for pet accidents.
Commercial Odor Neutralizers
A wide range of commercial products are available, formulated specifically for urine odor neutralization. These often leverage advanced chemical formulations.
Enzymatic Cleaners
These are the gold standard for biological odors like urine. They contain a blend of enzymes designed to break down urea, proteins, and fats.
- Application: Follow the product instructions carefully. Typically, the cleaner is applied directly to the affected area, allowed to dwell for a specified time to allow the enzymes to work, and then blotted or rinsed. For deep-set odors, multiple applications or a more concentrated formula might be needed. Look for products specifically labeled for pet stains or urine.
Oxidizing Cleaners
These products use oxidizing agents like hydrogen peroxide or peroxyacetic acid to break down odor molecules.
- Features: They are often fast-acting and can provide immediate odor relief. Some may also have bleaching properties, so caution should be exercised on colored fabrics.
Adsorbent Products
These products, often containing activated carbon or zeolites, work by trapping odor molecules.
- Usage: They are often used in the form of sprays, gels, or solid beads. They are good for ongoing odor control in areas where urine odors may linger, such as litter boxes or kennels.
Professional Cleaning Methods
For severe or persistent urine odor issues, professional cleaning services may be necessary.
Ozone Treatment
Professional ozone generators can effectively de-odorize entire rooms or spaces by breaking down odor molecules in the air and on surfaces. This is a powerful but potentially hazardous treatment that requires professional operation and proper ventilation afterwards.
Specialized Chemical Treatments
Professional cleaners may use more potent chemical solutions and advanced equipment to deeply penetrate and neutralize odors in carpets, upholstery, and structural elements.
Prevention and Ongoing Management
The most effective way to deal with urine odor is to prevent it from occurring in the first place.
Prompt Cleanup
Cleaning up urine accidents immediately is crucial. The longer urine remains on a surface, the more time bacteria have to break down urea and produce strong ammonia odors. Blot up as much liquid as possible before applying any cleaning solution.
Addressing the Source
If pet accidents are a recurring problem, it’s important to address the underlying cause. This could involve:
- Training: Reinforcing house training for pets.
- Medical Issues: Consulting a veterinarian to rule out any medical conditions causing incontinence.
- Behavioral Issues: Addressing anxiety or territorial marking.
Ventilation
Good ventilation helps to dissipate odors and reduce their concentration in the air. Opening windows and using fans can be beneficial, especially when dealing with fresh urine.
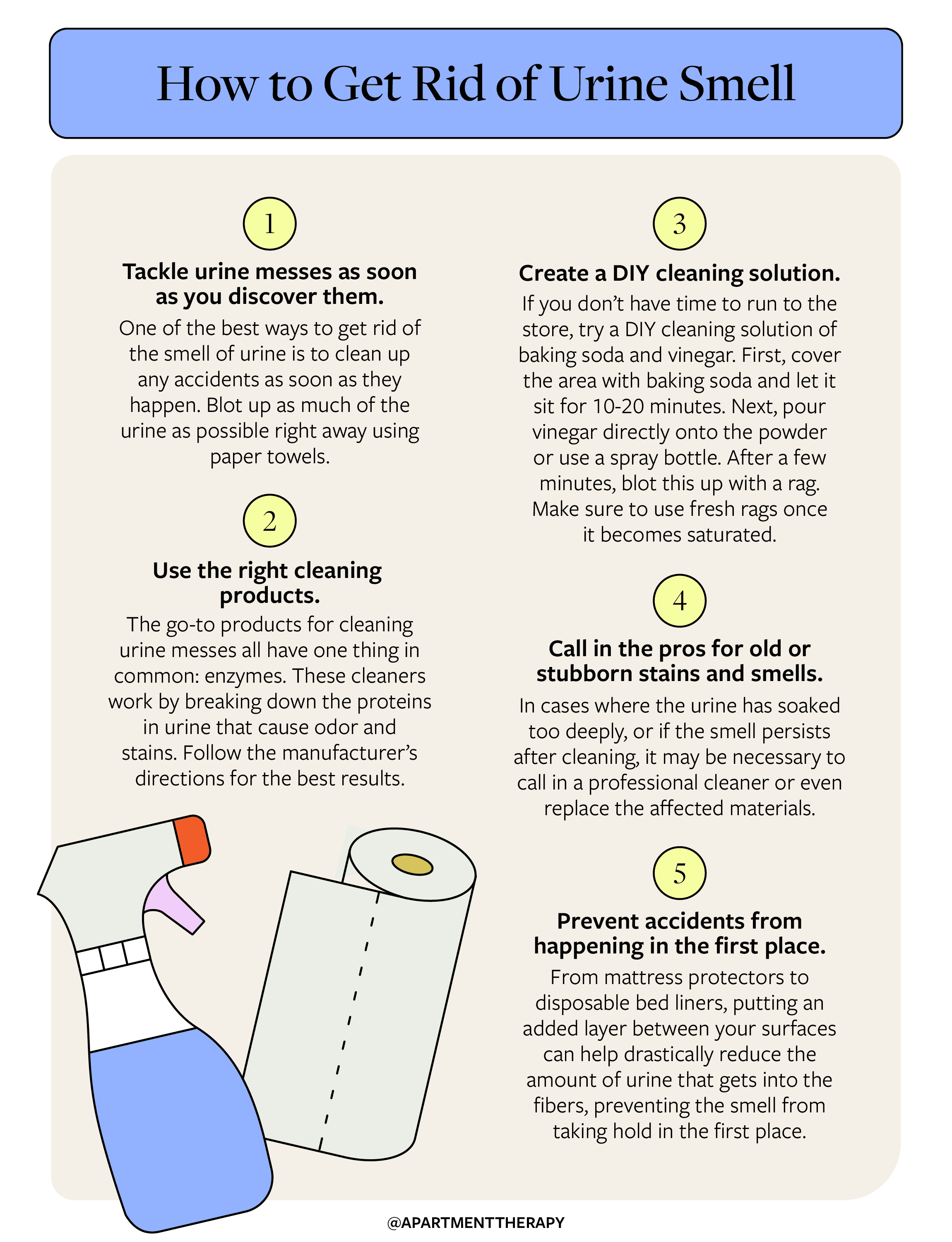
Regular Cleaning
Regular cleaning of areas prone to urine odor, such as pet bedding, litter boxes, and bathrooms, can prevent the buildup of odor-causing compounds.
By understanding the chemistry of urine odor and employing the appropriate neutralization strategies, it is possible to effectively eliminate these unpleasant smells and maintain a fresh, clean environment. Whether opting for simple household remedies or advanced commercial solutions, a systematic approach targeting the root cause of the odor will yield the best results.
