The Microscopic Menace: Understanding Trypanosoma brucei
Trypanosoma brucei is not a subject typically found in discussions about cutting-edge technology, flight, or visual capture. However, its profound impact on human and animal health, and the ongoing scientific endeavors to combat it, position it squarely within the realm of Tech & Innovation. The development of diagnostic tools, innovative treatment strategies, advanced surveillance methods, and ultimately, the pursuit of eradication, all hinge on technological breakthroughs and scientific ingenuity. This single-celled parasite, responsible for a devastating disease, serves as a stark reminder of how technology and innovation are crucial not just for advancement, but for safeguarding life itself.
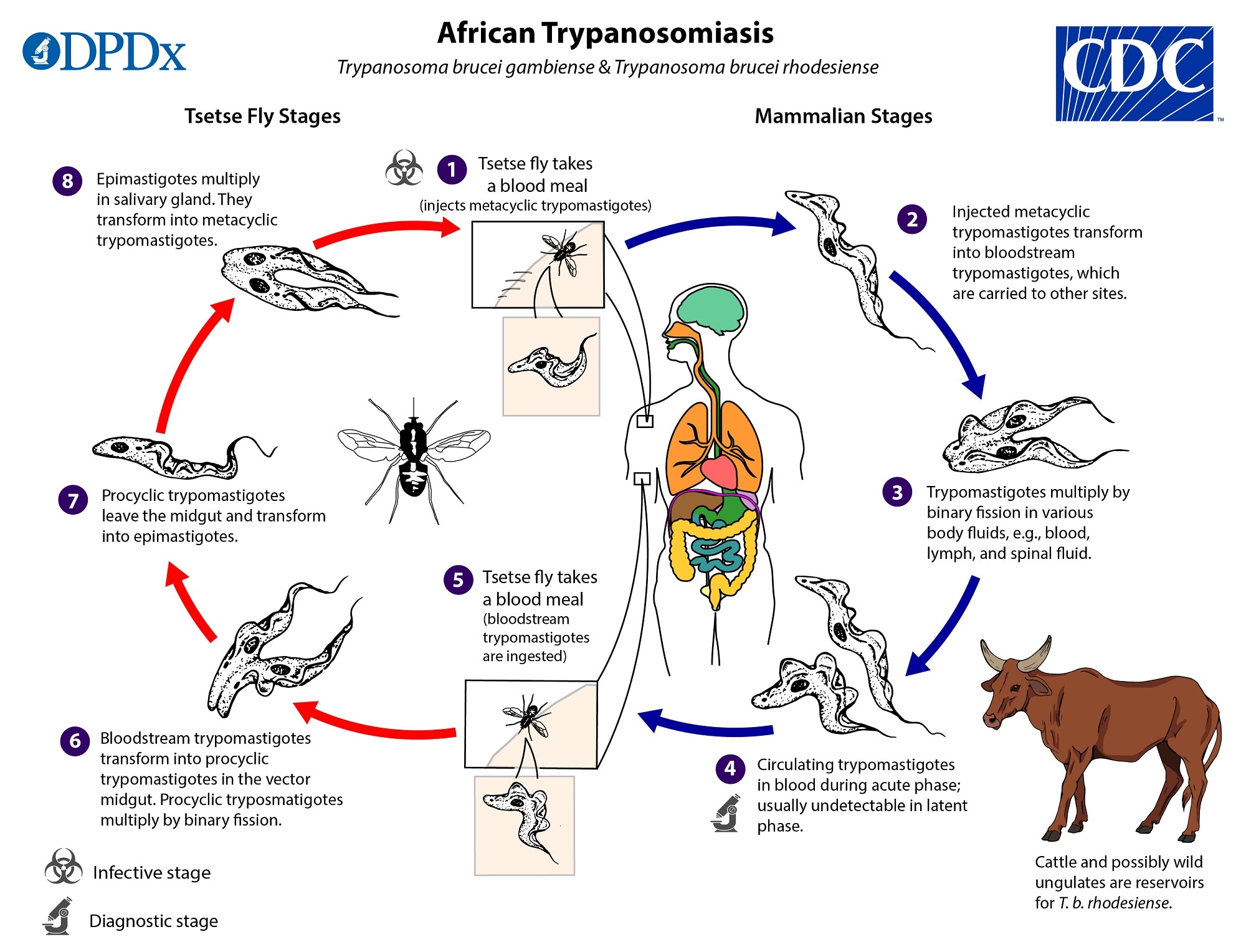
The Biological Profile of a Parasite
At its core, Trypanosoma brucei is a eukaryotic protozoan, a microscopic organism that relies on a host for survival and reproduction. It belongs to the genus Trypanosoma, a group of flagellated protozoa characterized by a single, whip-like appendage called a flagellum, which it uses for motility. Trypanosoma brucei is the causative agent of human African trypanosomiasis, also known as sleeping sickness.
Morphology and Life Cycle
T. brucei exists in several morphological forms throughout its complex life cycle, which involves two hosts: the tsetse fly (Glossina genus) and a vertebrate host, typically humans or other mammals.
- Tsetse Fly Stage: Within the tsetse fly, the trypanosomes undergo development. They begin as procyclic trypomastigotes in the midgut of the fly. These then migrate to the salivary glands, where they differentiate into epimastigotes and finally into metacyclic trypomastigotes. These metacyclic forms are the infective stage transmitted to a new vertebrate host during a blood meal by the fly.
- Vertebrate Host Stage: Once injected into the vertebrate host, the metacyclic trypomastigotes transform into bloodstream trypomastigotes. This is the form that circulates in the blood and lymph of the host. These slender, elongated cells are characterized by a kinetoplast (a dense granule containing mitochondrial DNA), a nucleus, and a flagellum that runs along the body and emerges at the anterior end. The bloodstream form is responsible for the pathogenesis of the disease. In some cases, particularly in the final stages of the disease, trypanosomes can cross the blood-brain barrier and invade the central nervous system, leading to the neurological symptoms characteristic of sleeping sickness.
Genetic Adaptability: A Major Challenge
One of the most significant challenges in combating T. brucei is its extraordinary ability to evade the host’s immune system. This is achieved through a sophisticated mechanism known as antigenic variation.
- Antigenic Variation: The surface of the bloodstream trypanosome is covered by a dense coat of proteins called the Variant Surface Glycoprotein (VSG). This VSG coat is highly immunogenic, meaning it elicits a strong immune response from the host, leading to the production of antibodies. However, T. brucei possesses a vast repertoire of genes encoding different VSGs, estimated to be hundreds, if not thousands. When the host’s immune system begins to mount an effective response against the current VSG, the trypanosome can switch to expressing a different VSG from its genetic library. This allows the parasite to continually present a “new” surface to the immune system, effectively remaining one step ahead of the host’s defense mechanisms and leading to chronic, relapsing infections. This genetic adaptability is a prime example of biological innovation driving survival and a critical area where technological solutions are sought.
The Impact: Sleeping Sickness and Nagana
The presence of Trypanosoma brucei in a vertebrate host leads to significant disease burdens. The disease manifests differently depending on the subspecies of T. brucei and the host.
Human African Trypanosomiasis (HAT)
Human African Trypanosomiasis (HAT) is a severe, neglected tropical disease that is fatal if left untreated. It is caused by two subspecies of T. brucei:
- Trypanosoma brucei gambiense: This subspecies is responsible for the vast majority (over 95%) of HAT cases and causes a chronic form of the disease that can progress over months or even years. It is primarily found in West and Central Africa. Initial symptoms include fever, headaches, joint pain, and swollen lymph nodes (known as Winterbottom’s sign in the posterior cervical chain). As the disease progresses into the second, neurological stage, it affects the central nervous system, leading to confusion, personality changes, sleep disturbances (hence “sleeping sickness”), coma, and ultimately death.
- Trypanosoma brucei rhodesiense: This subspecies causes a more acute and rapidly progressing form of HAT, typically leading to death within weeks or months if untreated. It is mainly found in East Africa. The neurological stage can be reached more quickly, with symptoms like confusion, delirium, and coma developing relatively rapidly.
The economic and social impact of HAT is devastating, affecting primarily rural populations in sub-Saharan Africa. It leads to reduced agricultural productivity, loss of livelihoods, and significant healthcare costs.
Animal African Trypanosomiasis (AAT)
Trypanosoma brucei also infects livestock, causing Animal African Trypanosomiasis (AAT), commonly known as nagana. This disease significantly impacts cattle, sheep, goats, and pigs, leading to reduced productivity, infertility, abortion, and death. Nagana poses a major constraint on livestock development and food security in affected regions. The presence of nagana also indirectly impacts human health by influencing tsetse fly populations, which can then transmit HAT to humans.
Technological Innovations in Detection and Diagnosis
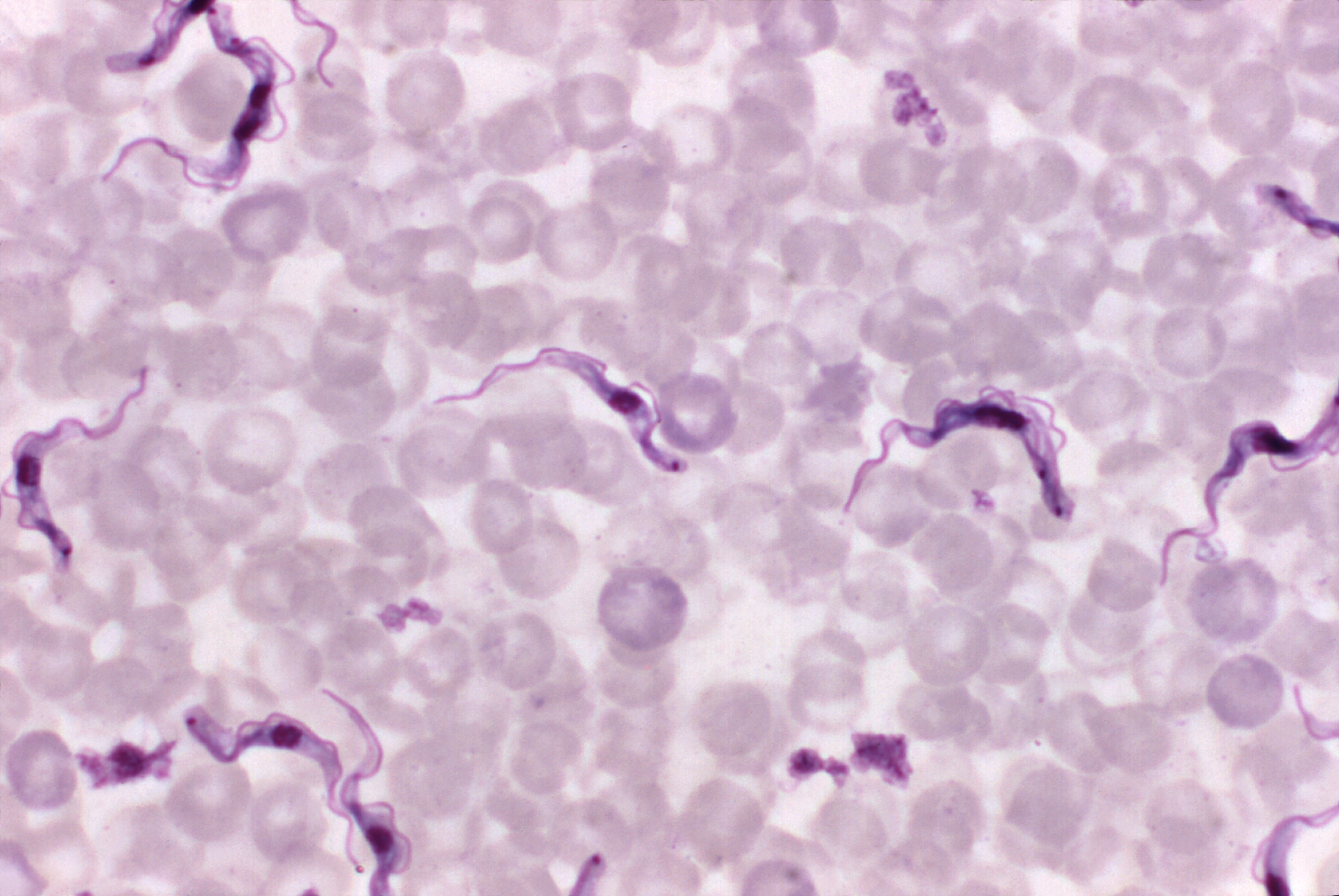
The effective control and eventual eradication of T. brucei hinges on the development and deployment of accurate, rapid, and accessible diagnostic tools. Traditional diagnostic methods often involve microscopy, which can be time-consuming, require specialized expertise, and lack sensitivity in early stages or low parasitemia. The innovation in this field has focused on overcoming these limitations.
Advanced Diagnostic Platforms
- Rapid Diagnostic Tests (RDTs): The development of RDTs has been a significant leap forward. These tests, often based on immunochromatographic principles, detect the presence of specific antibodies against T. brucei or parasite antigens in blood or serum. RDTs offer a portable, easy-to-use solution that can be deployed in remote settings, significantly improving case detection in primary healthcare facilities. Innovations continue to refine the sensitivity and specificity of these tests.
- Molecular Diagnostics: Polymerase Chain Reaction (PCR) and its variants have revolutionized the detection of T. brucei. PCR amplifies specific DNA sequences of the parasite, allowing for highly sensitive and specific detection, even at very low parasite loads. This is particularly valuable for early diagnosis, identifying asymptomatic carriers, and confirming diagnoses where other methods are inconclusive. Innovations in microfluidics and point-of-care PCR devices are making these technologies more accessible and faster.
- Serological Assays: Beyond basic RDTs, more sophisticated serological assays like Enzyme-Linked Immunosorbent Assays (ELISAs) and Indirect Immunofluorescence Assays (IIFAs) offer higher sensitivity and specificity for detecting antibodies. These are crucial for epidemiological surveillance and confirming diagnoses.
- Biosensors: The exploration of biosensor technology holds promise for the future. These devices integrate biological recognition elements with physical transducers to detect the presence of T. brucei or its components, offering the potential for real-time, on-site detection with minimal sample processing.
Innovations in Treatment and Drug Development
The treatment of sleeping sickness has historically been challenging, with drugs that were often toxic, difficult to administer, and increasingly ineffective due to drug resistance. The innovation in this area is critical for improving patient outcomes and reducing the burden of disease.
Refined Therapeutic Regimens
- Early-Stage Treatment: For HAT caused by T. brucei gambiense, early-stage treatment (before the central nervous system is significantly affected) typically involves pentamidine. While effective, pentamidine has side effects and requires injectable administration.
- Late-Stage Treatment: For late-stage HAT, melarsoprol, an arsenic-based compound, was the mainstay for decades. However, melarsoprol is highly toxic, causing severe neurological side effects, including encephalopathy and death in a significant percentage of patients. Its use has been significantly curtailed due to this toxicity.
- Newer Drug Combinations: Significant innovation has led to the development of improved treatment regimens. Eflornithine, originally developed for cancer chemotherapy, was found to be effective against T. brucei gambiense in the late stage and is less toxic than melarsoprol. However, it requires intravenous administration over extended periods. The combination of eflornithine and NECT (nifurtimox-eflornithine combination therapy) has become the preferred treatment for late-stage T. brucei gambiense HAT, offering improved efficacy and reduced toxicity compared to melarsoprol. Nifurtimox, another drug, is effective against early-stage T. brucei rhodesiense and also used in combination with eflornithine.
- Oral Treatments: A major focus of ongoing innovation is the development of safe, effective, and orally administered drugs for all stages of HAT. This would dramatically simplify treatment delivery, reduce the need for hospitalization, and improve patient adherence. Several promising drug candidates are in various stages of preclinical and clinical development.
Surveillance, Control, and Eradication Strategies: The Role of Technology
Beyond diagnosis and treatment, controlling and ultimately eradicating T. brucei requires sophisticated strategies that leverage technological advancements for population-level interventions.
Vector Control Innovations
The tsetse fly is the vector responsible for transmitting T. brucei. Innovations in vector control are crucial for interrupting transmission.
- Insecticide-Treated Targets (ITTs): These are passive traps impregnated with insecticide. They attract tsetse flies with visual and olfactory cues, leading to their death upon contact. ITTs are cost-effective and can be deployed strategically in high-risk areas.
- Sterile Insect Technique (SIT): This environmentally friendly approach involves releasing large numbers of sterilized male tsetse flies into the wild. When these sterile males mate with wild females, they produce no offspring, thereby reducing the overall fly population over time. Advances in mass rearing and sterilization techniques are making SIT more feasible.
- Genetic Control Strategies: Research is ongoing into developing genetic strategies to control tsetse fly populations, such as using gene drives or modifying the flies to be refractory to the parasite. While these are still in early stages of development, they represent potential revolutionary tools for future eradication efforts.
Disease Surveillance and Mapping
Technological innovations are transforming how we monitor the spread of HAT and nagana.
- Geographic Information Systems (GIS): GIS technology allows for the mapping and analysis of disease prevalence, vector distribution, and risk factors. This information is vital for identifying hotspots, allocating resources effectively, and planning targeted interventions.
- Remote Sensing and Satellite Imagery: These technologies can provide valuable data on land use, environmental conditions, and human population movements, which can be correlated with the distribution of tsetse flies and disease risk. This helps in understanding the ecological drivers of transmission.
- Mobile Health (mHealth) Applications: The use of smartphones and mobile applications for data collection by healthcare workers in the field is streamlining surveillance efforts. This includes real-time reporting of case numbers, treatment outcomes, and vector control activities, enabling faster response times and better data management.
Data Integration and Predictive Modeling
The future of combating T. brucei lies in the intelligent integration of data from various sources and the application of sophisticated modeling.
- Big Data Analytics: Combining diagnostic data, treatment records, vector surveillance information, and environmental data allows for the identification of complex patterns and trends that might not be apparent otherwise.
- Predictive Modeling: Developing predictive models that can forecast disease outbreaks or identify areas at high risk of future transmission is a key area of innovation. These models can inform proactive public health interventions and resource allocation.
- Artificial Intelligence (AI) and Machine Learning (ML): AI and ML algorithms are increasingly being used to analyze large datasets, identify novel diagnostic markers, predict drug efficacy, and optimize vector control strategies. For instance, AI can be used to analyze images from microscopy for faster parasite identification or to predict optimal locations for deploying tsetse fly traps.

The Future: Towards Eradication Through Sustained Innovation
The battle against Trypanosoma brucei is a testament to the power of scientific inquiry and technological innovation. From understanding the intricate biology of a microscopic parasite to developing sophisticated diagnostic tools, life-saving drugs, and targeted control strategies, progress has been driven by relentless pursuit of knowledge and the application of novel technologies. The ongoing research into oral medications, novel vector control methods, and advanced data analytics points towards a future where this devastating disease could be not just managed, but eradicated. This ongoing fight underscores the critical role of continued investment in R&D and interdisciplinary collaboration, harnessing the full spectrum of technological advancements to protect human and animal health.
