The Earth’s atmosphere is a complex and dynamic system, and within it lies a crucial component that often goes unnoticed: the ozone layer. This vital shield, primarily situated in the stratosphere, plays an indispensable role in safeguarding life on our planet. While its name might conjure images of a protective blanket, its actual significance extends far beyond mere insulation. Understanding the importance of the ozone layer is paramount to appreciating the delicate balance of our environment and the necessity of its preservation.
The Ozone Layer’s Protective Embrace: Shielding Life from Harmful Radiation
The fundamental importance of the ozone layer lies in its ability to absorb a significant portion of the Sun’s ultraviolet (UV) radiation. This absorption is not a uniform process; rather, ozone molecules are particularly effective at filtering out the most harmful wavelengths of UV light, namely UV-B and UV-C radiation.
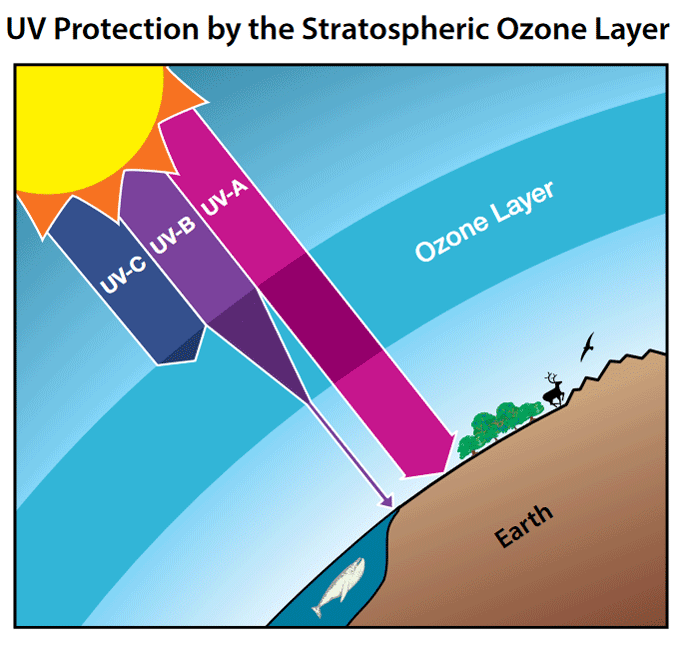
Understanding Ultraviolet Radiation and its Spectrum
To fully grasp the ozone layer’s protective function, it’s essential to understand the different types of UV radiation:
-
UV-A Radiation: This is the least energetic form of UV radiation and has a longer wavelength. While it can penetrate the skin and contribute to premature aging and skin damage, its impact is generally less severe than UV-B and UV-C. The ozone layer absorbs only a small percentage of UV-A.
-
UV-B Radiation: This radiation is more energetic than UV-A and is largely absorbed by the ozone layer. However, a significant portion still reaches the Earth’s surface. UV-B is responsible for sunburn, and prolonged exposure can lead to skin cancer, cataracts, and damage to the immune system. It also affects plant growth and marine ecosystems.
-
UV-C Radiation: This is the most energetic and dangerous form of UV radiation. Fortunately for life on Earth, the ozone layer is an exceptionally effective absorber of UV-C, blocking virtually all of it from reaching the surface. If this radiation were to penetrate, it would be highly damaging to all living organisms.
The Mechanism of Ozone Absorption
Ozone (O3) molecules have a unique molecular structure that makes them highly reactive with UV radiation. When a UV photon strikes an ozone molecule, it provides the energy to break the molecule apart into an oxygen molecule (O2) and a free oxygen atom (O). This process is reversible. The free oxygen atom can then react with another oxygen molecule to form a new ozone molecule, creating a continuous cycle of ozone formation and destruction. This dynamic equilibrium is what allows the ozone layer to effectively absorb UV radiation.
The absorption process can be summarized as follows:
- UV photon strikes an ozone molecule: O3 + UV photon → O2 + O
- A free oxygen atom reacts with an oxygen molecule: O + O2 → O3
While both O3 formation and O3 destruction occur naturally, the net effect of this continuous process is the absorption of UV energy, preventing it from reaching the ground. The concentration of ozone is highest in the stratosphere, typically between 15 and 35 kilometers (9 to 22 miles) above the Earth’s surface, creating the “ozone layer.”
The Impact of Ozone Depletion: A Global Health and Environmental Crisis
The recognition of the ozone layer’s importance was amplified by the alarming discovery of ozone depletion, particularly the formation of the “ozone hole” over Antarctica. This depletion, primarily caused by human-made chemicals, has profound implications for global health and ecosystems.
Skin Cancer and Other Health Risks
The most direct consequence of increased UV-B radiation reaching the Earth’s surface due to ozone depletion is a rise in the incidence of skin cancers, including melanoma, basal cell carcinoma, and squamous cell carcinoma. UV-B radiation damages the DNA in skin cells, leading to mutations that can result in cancerous growth. Beyond skin cancer, excessive UV-B exposure can also lead to:
- Cataracts: Clouding of the eye’s lens, leading to impaired vision and blindness.
- Suppressed Immune System: Weakening the body’s defenses, making individuals more susceptible to infections and diseases.
- Photoaging: Premature aging of the skin, characterized by wrinkles, age spots, and loss of elasticity.
Ecosystem Disruption and Biodiversity Loss
The impact of increased UV radiation extends far beyond human health, posing significant threats to terrestrial and aquatic ecosystems.
-
Terrestrial Ecosystems: Plants are particularly sensitive to UV-B radiation. Excessive exposure can impair photosynthesis, reduce growth rates, alter flowering times, and affect crop yields. This can have cascading effects throughout food webs, impacting herbivores and ultimately the entire ecosystem. Certain plant species are more vulnerable than others, leading to shifts in plant community composition and potential biodiversity loss.
-
Aquatic Ecosystems: Marine life, especially phytoplankton, which forms the base of most aquatic food webs, is highly susceptible to UV-B radiation. Phytoplankton absorb CO2 and produce oxygen, making them crucial for the planet’s health. Damage to phytoplankton populations can disrupt marine food webs, impacting fish, marine mammals, and seabirds. UV-B radiation can also harm fish larvae, shrimp, and other aquatic organisms, affecting their development and survival.
Material Degradation
Beyond biological impacts, increased UV radiation can also degrade certain materials, such as plastics, paints, and rubber. This can lead to their premature breakdown and reduced lifespan, causing economic losses and requiring more frequent replacement.
The Role of Human Activities: The Culprits Behind Ozone Depletion
For decades, the scientific community has worked to understand the causes of ozone depletion. It was unequivocally established that human-produced chemicals, primarily chlorofluorocarbons (CFCs) and halons, were the main culprits.
Chlorofluorocarbons (CFCs) and Halons: Persistent Pollutants

CFCs are synthetic chemical compounds that were widely used in the mid-20th century as refrigerants, propellants in aerosol cans, blowing agents in foam production, and solvents. Halons were primarily used in fire extinguishers. These compounds are remarkably stable in the lower atmosphere, meaning they don’t break down easily. This stability allows them to persist for decades, even centuries, and gradually migrate to the stratosphere.
The Stratospheric Breakdown: Unleashing Ozone-Destroying Agents
Once in the stratosphere, CFCs and halons are exposed to intense UV radiation. This radiation breaks them down, releasing highly reactive chlorine and bromine atoms. These atoms then act as catalysts in a series of chemical reactions that destroy ozone molecules.
The simplified cycle of ozone depletion by chlorine is as follows:
- Chlorine atom reacts with an ozone molecule: Cl + O3 → ClO + O2
- Chlorine monoxide reacts with a free oxygen atom: ClO + O → Cl + O2
In this cycle, a single chlorine atom can destroy thousands of ozone molecules before it is eventually removed from the stratosphere. Bromine atoms are even more efficient at destroying ozone than chlorine atoms.
The Ozone Hole Phenomenon: A Stark Warning
The “ozone hole” observed over Antarctica is a dramatic manifestation of ozone depletion. During the Antarctic spring (September to October), specific meteorological conditions, including extremely cold temperatures and polar stratospheric clouds, create an environment where chlorine and bromine atoms are released and can efficiently destroy ozone. This leads to a significant thinning of the ozone layer in this region. Similar, though less pronounced, depletion has been observed over the Arctic.
Global Efforts and Successes: The Montreal Protocol and Beyond
The scientific understanding of ozone depletion spurred unprecedented global cooperation to address the issue. The development and implementation of international agreements have been remarkably successful in mitigating the threat.
The Montreal Protocol on Substances that Deplete the Ozone Layer
Signed in 1987, the Montreal Protocol is a landmark international treaty designed to phase out the production and consumption of ozone-depleting substances (ODS). It is widely considered one of the most successful environmental agreements in history. Under the protocol, signatory nations committed to a schedule for reducing and eventually eliminating the use of CFCs, halons, and other ODS.
The protocol’s success can be attributed to several factors:
- Strong Scientific Consensus: Clear and compelling scientific evidence guided the protocol’s development and implementation.
- Phased Approach: The protocol allowed for a gradual phase-out, giving industries time to develop and adopt alternatives.
- Flexibility and Adaptability: The protocol has been amended and adjusted over time to incorporate new scientific findings and address emerging challenges.
- Financial Mechanisms: The Multilateral Fund for the Implementation of the Montreal Protocol provides financial assistance to developing countries to help them meet their obligations.
The Emergence of Alternatives and Technological Innovation
The phase-out of ODS spurred significant innovation in finding safer alternatives. Industries developed new refrigerants, propellants, and solvents that do not harm the ozone layer. While some early alternatives had their own environmental concerns (e.g., some hydrofluorocarbons, HFCs, are potent greenhouse gases), the focus has continued to shift towards more sustainable solutions.
Technological advancements have also played a role in monitoring and understanding the ozone layer. Satellites equipped with sophisticated instruments continuously track ozone concentrations, providing crucial data for assessing the effectiveness of the Montreal Protocol and identifying any remaining challenges. Remote sensing technologies allow scientists to measure ozone levels globally, detect depletion events, and refine atmospheric models.
The Ongoing Importance of Ozone Layer Protection: A Continuous Commitment
While the Montreal Protocol has been a resounding success, the work of protecting the ozone layer is far from over. The ozone layer is still recovering, and several factors necessitate continued vigilance and commitment.
The Slow Pace of Stratospheric Recovery
Ozone-depleting substances have long atmospheric lifetimes. Even with the complete cessation of production and use, it will take many decades for the ozone layer to fully recover to its pre-depletion levels. This means that for the foreseeable future, the Earth will remain more vulnerable to UV radiation than it was in the past.
Emerging Threats and Challenges
Despite the success of the Montreal Protocol, there are ongoing challenges and potential new threats:
- Illegal Production and Trade: The black market for ODS can undermine the protocol’s effectiveness.
- Unexpected Emissions: In recent years, scientists have detected unexpected emissions of certain controlled ODS, highlighting the need for continued monitoring and enforcement.
- Climate Change Interactions: Climate change itself can influence stratospheric temperatures and circulation patterns, which can, in turn, affect the rate of ozone recovery. For example, a warming lower atmosphere can lead to a cooling upper atmosphere (stratosphere), which can slow down ozone depletion in some regions but potentially exacerbate it in others.
- New Chemical Risks: As new chemicals are developed and introduced into the atmosphere, ongoing scientific assessment is crucial to ensure they do not pose a threat to the ozone layer.

The Indispensable Role in a Healthy Planet
The importance of the ozone layer cannot be overstated. It is a critical component of Earth’s life-support system, protecting us from the damaging effects of solar UV radiation. Its preservation is essential for:
- Human Health: Preventing widespread skin cancer, cataracts, and immune system suppression.
- Ecosystem Stability: Ensuring the health and productivity of terrestrial and aquatic ecosystems, from the smallest phytoplankton to the largest mammals.
- Agricultural Productivity: Safeguarding food security by protecting crops from UV damage.
- Biodiversity: Maintaining the intricate web of life that enriches our planet.
The success of the Montreal Protocol serves as a powerful testament to what humanity can achieve when we unite with a shared purpose and act upon scientific evidence. The ongoing protection of the ozone layer requires continued international cooperation, robust scientific monitoring, and a commitment to sustainable practices. By understanding and valuing the importance of this invisible shield, we invest in the health of our planet and the future of all living beings.
