The question “What is the biochemical oxygen demand?” directly probes a fundamental concept in environmental science, particularly concerning water quality. While not explicitly tied to the listed drone-centric categories, the application of measuring and understanding Biochemical Oxygen Demand (BOD) falls squarely within the realm of Tech & Innovation, specifically Remote Sensing and Environmental Monitoring technologies that can be deployed via aerial platforms. This article will explore BOD through the lens of its measurement, significance, and how advanced technological solutions, including those deployable via drones, contribute to its understanding and management.
Understanding Biochemical Oxygen Demand
Biochemical Oxygen Demand (BOD) is a crucial measure of the amount of dissolved oxygen (DO) needed by aerobic biological organisms to break down organic material present in a given water sample at a certain temperature over a specific time period. Essentially, it quantifies the oxygen consumed by microorganisms as they decompose organic pollutants. A higher BOD value indicates a greater amount of organic pollution, which can deplete the dissolved oxygen levels in water bodies. This depletion can be detrimental to aquatic life, leading to suffocation and potentially fish kills.
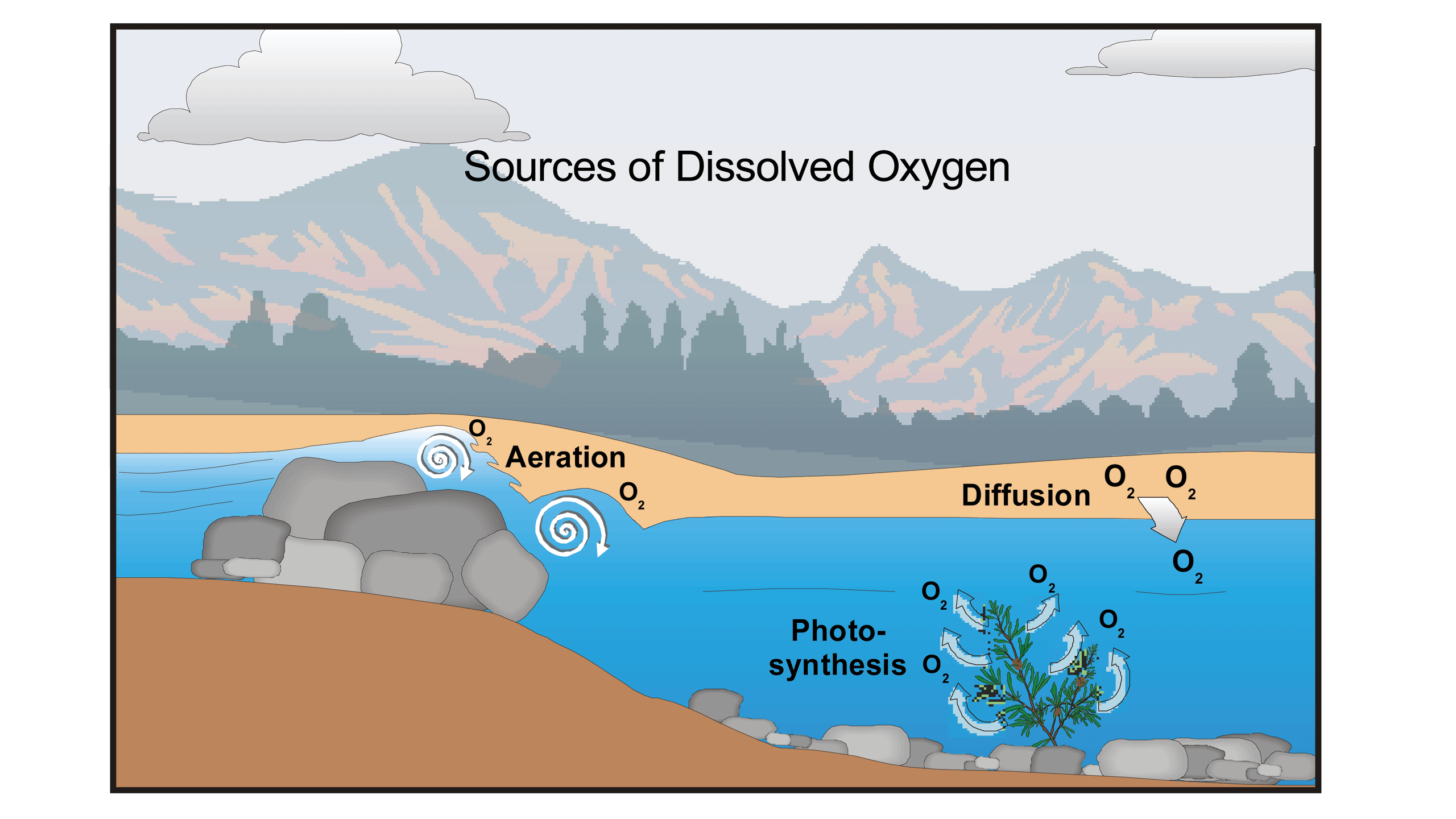
The Decomposition Process
When organic matter enters a water system, such as from wastewater discharge, agricultural runoff, or natural decay of plant and animal material, it serves as a food source for aerobic bacteria and other microorganisms. These organisms utilize dissolved oxygen in the water to metabolize and break down the complex organic compounds into simpler inorganic substances like carbon dioxide and water. This biological process is central to the concept of BOD.
The decomposition typically proceeds in two phases:
- Carbonaceous BOD (cBOD): This phase involves the microbial oxidation of carbon-containing organic compounds. It is the primary component measured in standard BOD tests.
- Nitrogenous BOD (nBOD): This phase occurs after the carbonaceous phase and involves the oxidation of nitrogen-containing compounds, such as ammonia, by nitrifying bacteria. While it also consumes oxygen, it is often measured separately or accounted for in longer-term BOD tests.
Factors Influencing BOD
Several environmental factors significantly influence the rate and extent of BOD in a water body:
- Temperature: Microorganisms are more active at higher temperatures, leading to a faster rate of decomposition and thus a higher BOD. Conversely, colder temperatures slow down microbial activity, reducing BOD. Standard BOD tests are conducted at a controlled temperature of 20°C to ensure comparability.
- Dissolved Oxygen Concentration: The availability of dissolved oxygen is critical. If DO levels are too low, aerobic decomposition cannot occur efficiently, and anaerobic processes may take over, producing different byproducts and potentially toxic substances.
- Presence of Microorganisms: A sufficient population of active aerobic microorganisms is necessary for decomposition. Seeding the sample with a known quantity of microorganisms can be done if the natural microbial population is insufficient or inhibited.
- Type and Concentration of Organic Matter: Different types of organic compounds vary in their biodegradability. Readily biodegradable compounds will exert a higher BOD over a shorter period compared to more recalcitrant or complex organic matter. The concentration of organic matter directly dictates the oxygen demand.
- Light Exposure: Sunlight can inhibit the activity of some microorganisms and can also promote the growth of photosynthetic organisms that produce oxygen. Therefore, light can indirectly influence BOD.
- pH: Microbial activity is optimal within a certain pH range. Extreme pH values can inhibit or kill microorganisms, affecting the BOD measurement.
The Significance of BOD in Water Quality Assessment
BOD is a critical parameter for assessing the health and quality of aquatic ecosystems. It provides valuable insights into the potential impact of organic pollution on water bodies.
Impact on Aquatic Life
The most direct consequence of high BOD is the depletion of dissolved oxygen in water. Aquatic organisms, including fish, invertebrates, and other wildlife, rely on dissolved oxygen for respiration. As BOD increases due to pollution, DO levels drop. When DO falls below critical thresholds, it can lead to:
- Stress and Reduced Growth: Even moderate DO depletion can stress aquatic organisms, impairing their growth, reproduction, and ability to evade predators.
- Suffocation and Mortality: Critically low DO levels can cause widespread suffocation and mortality, leading to significant ecological damage.
- Shift in Species Composition: Organisms sensitive to low DO will disappear, while more tolerant species may dominate, leading to a loss of biodiversity and a simplified ecosystem structure.
- Anaerobic Conditions: In extreme cases, the water can become anoxic (virtually devoid of oxygen), leading to the production of hydrogen sulfide (which smells like rotten eggs) and other toxic byproducts of anaerobic decomposition, further rendering the water uninhabitable for most aquatic life.
Wastewater Treatment Efficiency
BOD is a primary indicator used to monitor the effectiveness of wastewater treatment processes. Municipal and industrial wastewater often contains high concentrations of organic pollutants. Treatment plants are designed to remove these pollutants, reducing the BOD of the effluent before it is discharged into natural water bodies. Regular BOD testing of influent (incoming wastewater) and effluent (treated wastewater) allows operators to:
- Assess Treatment Performance: A significant reduction in BOD from influent to effluent indicates that the treatment process is working effectively.
- Optimize Treatment Processes: If BOD levels remain high, adjustments can be made to the treatment process, such as increasing aeration time or optimizing microbial populations, to improve efficiency.
- Ensure Regulatory Compliance: Environmental regulations often set strict limits on the BOD levels of discharged wastewater. Monitoring BOD is essential for compliance and avoiding penalties.
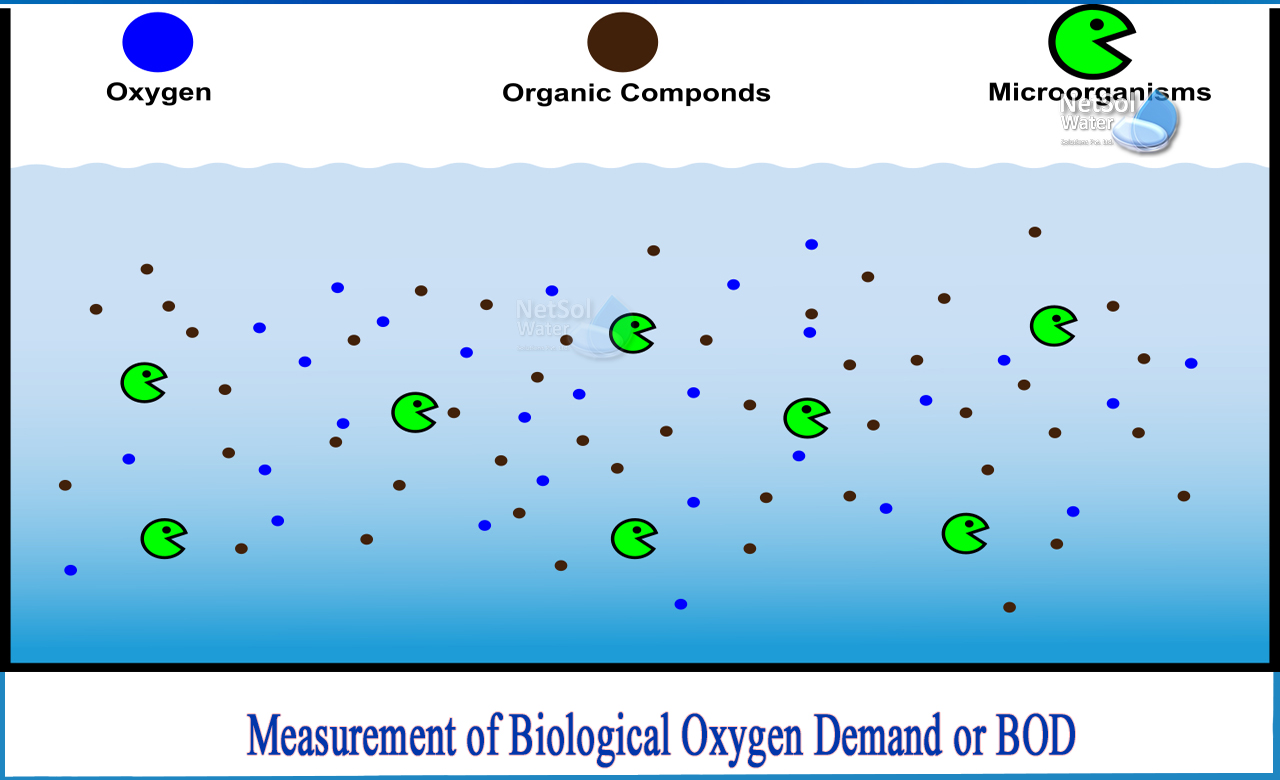
Identifying Pollution Sources
By measuring BOD at various points in a river or lake, scientists can help identify the sources and extent of organic pollution. A localized increase in BOD downstream of a suspected discharge point can strongly suggest that the discharge is the primary contributor to the pollution. This information is crucial for targeted remediation efforts and enforcement actions.
Measuring Biochemical Oxygen Demand
The standard method for measuring BOD involves a laboratory procedure that incubates a water sample under specific conditions for a set period, typically five days, hence the common term “BOD5.”
The Standard BOD Test Procedure
-
Sample Collection: Water samples are collected from the water body or wastewater stream. Care is taken to avoid introducing air into the sample, which could artificially inflate the initial DO reading.
-
Dissolved Oxygen Measurement (Initial): The initial dissolved oxygen concentration of the water sample is measured immediately after collection.
-
Dilution: If the expected BOD is high, the sample is diluted with a known volume of oxygen-saturated dilution water. This ensures that sufficient DO is available throughout the incubation period for the microorganisms to act upon the organic matter without becoming depleted. The dilution water is typically prepared by aerating tap water or distilled water and is often seeded with a small amount of settled wastewater or surface water to provide a healthy microbial population.
-
Incubation: The diluted (or undiluted) sample is placed in a BOD bottle and incubated in a dark, temperature-controlled incubator at 20°C for five days. The darkness prevents photosynthetic activity from contributing to the DO levels.
-
Dissolved Oxygen Measurement (Final): After five days, the final dissolved oxygen concentration of the incubated sample is measured.
-
Calculation: The BOD5 is calculated using the following formula:
$BOD5 = frac{(DO{initial} – DO_{final}) times text{Dilution Factor}}{text{Volume of Sample Used}}$
Where:
- $DO_{initial}$ is the initial dissolved oxygen concentration of the diluted sample.
- $DO_{final}$ is the final dissolved oxygen concentration of the diluted sample after five days of incubation.
- Dilution Factor is the ratio of the total volume of the diluted sample to the volume of the original sample used.
Limitations of the Standard BOD Test
While the standard BOD5 test is widely used, it has certain limitations:
- Time-Consuming: A five-day incubation period makes it unsuitable for real-time monitoring or rapid decision-making.
- Temperature Sensitivity: Results are highly dependent on maintaining a precise incubation temperature.
- Microbial Sensitivity: The test results can be affected by the presence of toxic substances that inhibit microbial activity or by an insufficient or overly dominant microbial population.
- Doesn’t Measure All Organic Matter: It primarily measures readily biodegradable organic matter. Recalcitrant organic compounds, which may still impact water quality over longer periods, are not fully accounted for.
Technological Innovations in BOD Measurement and Monitoring
Recognizing the limitations of traditional methods, technological advancements are providing more efficient and sophisticated ways to measure and monitor BOD, including applications in remote sensing.
Rapid BOD Measurement Methods
Researchers have developed faster methods to estimate BOD, often by correlating with other parameters or using biochemical reactions that occur more quickly. These include:
- Respirometric Methods: These methods measure the oxygen uptake rate directly, often over shorter periods, and extrapolate to estimate BOD. Some advanced respirometers can provide results within hours.
- DO-Stat Systems: These automated systems maintain a constant DO level in the sample during incubation by adding pure oxygen, allowing for faster and more consistent measurements.
- Chemical Oxygen Demand (COD) as an Indicator: While not a direct measure of BOD, COD measures the total amount of oxygen required to chemically oxidize all organic matter (biodegradable and non-biodegradable). There is often a correlation between BOD and COD, allowing for rapid COD measurements to provide an indirect estimate of BOD.
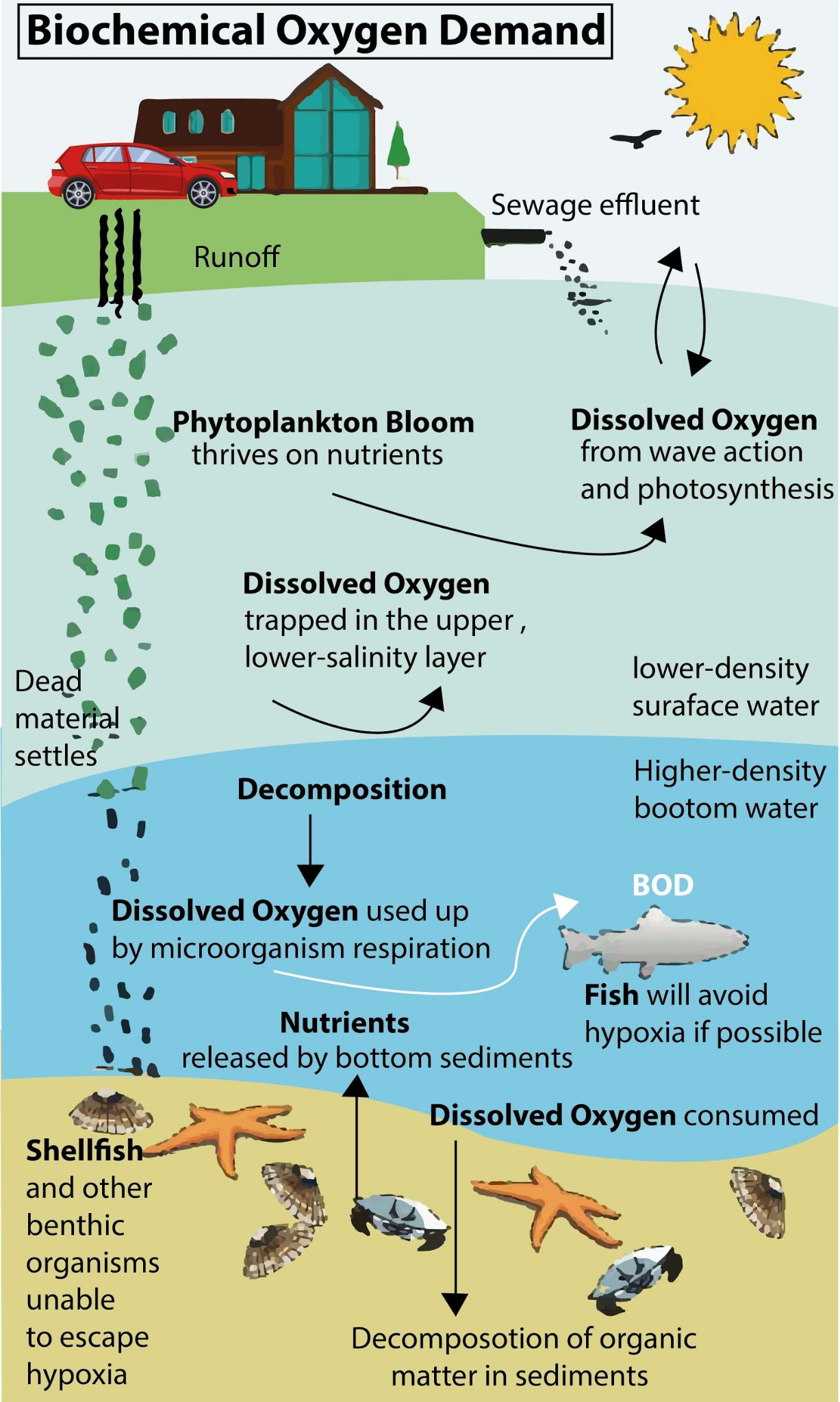
Remote Sensing and Drone Applications
The increasing sophistication of sensor technology and unmanned aerial vehicles (UAVs), commonly known as drones, is opening new avenues for environmental monitoring, including aspects related to water quality and the assessment of factors influencing BOD.
- Water Quality Sensor Deployment: Drones equipped with specialized sensors can collect samples from hard-to-reach areas or survey large bodies of water efficiently. While a full laboratory BOD test still requires sample collection, drones can facilitate the process.
- Proxy Parameter Sensing: Drones can carry sensors that measure parameters directly related to organic pollution and its impact, which can then be used to infer BOD levels. These include:
- Turbidity Sensors: High turbidity can indicate increased organic particulate matter, which contributes to BOD.
- Chlorophyll-a Sensors: Elevated chlorophyll-a levels often signal eutrophication, which is driven by nutrient enrichment and can lead to increased organic load and BOD.
- Spectrophotometers (Multispectral/Hyperspectral): These sensors can detect the spectral signatures of various organic pollutants in the water column. Advanced algorithms can then be used to estimate the concentration of these pollutants, providing an indirect indication of potential BOD.
- Temperature Sensors: Drones can map surface water temperatures, crucial for understanding how temperature influences the actual BOD exerted in the environment.
- Mapping and Spatial Analysis: Drones provide high-resolution aerial imagery that can be used to map pollution plumes, identify sources of runoff, and assess the overall health of aquatic ecosystems. This spatial data, combined with localized sensor readings, can help predict areas with higher BOD.
- Accessibility to Remote Areas: For monitoring remote lakes, rivers, or coastal areas, drones offer a cost-effective and rapid solution compared to traditional boat-based surveys or manual sampling. This allows for more frequent and comprehensive data collection, which is vital for understanding dynamic water quality changes.
While direct drone-based measurement of BOD is still an evolving field and typically relies on collecting physical samples for laboratory analysis, the integration of advanced sensors on UAV platforms is revolutionizing how we approach environmental monitoring. By providing comprehensive spatial data and enabling efficient collection of proxy indicators, drone technology significantly enhances our ability to understand, predict, and manage the impact of organic pollution, and by extension, the critical parameter of Biochemical Oxygen Demand. This innovative approach underscores the growing synergy between environmental science and advanced technological capabilities.
