The materials science behind silicone implants is a fascinating intersection of chemistry, engineering, and medicine. While the term “silicone implant” is often used broadly, it encompasses a range of sophisticated materials designed for biocompatibility, durability, and aesthetic outcomes. The core component, silicone, is a polymer known for its unique properties that make it ideal for a variety of medical applications, from reconstructive surgery to cosmetic enhancements. Understanding the composition of these implants requires delving into the specific types of silicones used, their manufacturing processes, and the additional materials that contribute to their structure and performance.
![]()
The Chemistry of Silicone: A Polymer Backbone
At its heart, silicone is not a single substance but a family of synthetic polymers. The fundamental building block of all silicones is a repeating chain of silicon-oxygen atoms, known as a siloxane backbone. This inorganic backbone sets silicones apart from organic polymers, which are primarily composed of carbon chains. The silicon-oxygen bond is exceptionally strong and stable, contributing to the inherent resilience and inertness of silicone materials.
Siloxane Chains and Their Properties
The siloxane backbone is represented by the formula [R₂SiO]n, where ‘Si’ represents silicon, ‘O’ represents oxygen, and ‘R’ represents organic side groups. These ‘R’ groups are typically methyl groups (CH₃), but they can be varied to modify the properties of the resulting silicone. The nature and arrangement of these side groups are critical in determining the silicone’s viscosity, flexibility, thermal stability, and chemical resistance. For example, in medical-grade silicones, these side groups are carefully chosen to minimize reactivity with biological tissues.
The chain length (represented by ‘n’) also plays a significant role. Longer siloxane chains generally lead to more viscous and flexible silicones, while shorter chains result in more rigid materials. The cross-linking between these polymer chains is another crucial factor. By introducing cross-links, manufacturers can create a network structure that significantly enhances the material’s strength, elasticity, and resistance to deformation.
Types of Silicones Used in Implants
Medical-grade silicones are a specific subset of these polymers, engineered to meet stringent safety and performance standards. They are characterized by their high purity, inertness, and excellent biocompatibility. There are several key forms of silicone utilized in implants:
-
Silicone Gel: This is perhaps the most common form. Silicone gel used in implants is typically a cohesive or semi-cohesive material. This means the silicone molecules are cross-linked in such a way that the gel maintains its shape and integrity, even if the implant shell is breached. Older generations of silicone gel were more fluid and could “bleed” out, but modern cohesive gels are designed to stay within the implant shell, reducing the risk of migration and improving the predictability of implant shape. The ‘R’ groups on the siloxane backbone are often engineered to provide a specific degree of softness and compliance.
-
Silicone Elastomers: These are cross-linked silicone polymers that behave like rubber. They are highly flexible, elastic, and durable. Silicone elastomers form the outer shell of most silicone implants. The degree of cross-linking and the specific ‘R’ groups used determine the elastomer’s tensile strength, tear resistance, and elongation properties. This ensures the shell can withstand the stresses of implantation and everyday movement without rupturing.
-
Liquid Silicone Rubber (LSR): This is a two-part, platinum-cured silicone that is processed through injection molding. It offers precise control over the final shape and consistency of the implant. LSR is known for its high purity and excellent biocompatibility, making it a preferred material for many implantable devices.
The Manufacturing Process: From Raw Materials to Medical Devices
The transformation of raw silicone compounds into implantable medical devices is a complex and highly controlled process, adhering to strict regulatory guidelines to ensure patient safety. This process involves meticulous formulation, polymerization, shaping, and rigorous quality control.
Polymerization and Cross-linking
The initial stage involves the synthesis of the silicone polymers. This typically starts with silicon metal, which is reacted with methyl chloride to produce methylchlorosilanes. These silanes are then hydrolyzed to form silanols, which subsequently polymerize to create the siloxane backbone.
The process of creating the desired silicone gel or elastomer involves controlled polymerization and cross-linking. For silicone gels, a low-viscosity silicone fluid is mixed with cross-linking agents and catalysts. Upon heating, these components react, forming a three-dimensional network that traps the fluid and creates the cohesive gel. The degree of cross-linking is precisely controlled to achieve the desired firmness and cohesiveness.

For silicone elastomers used in implant shells, similar polymerization and cross-linking reactions occur, but with a higher degree of cross-linking to create a more robust and elastic material. The specific curing method – whether it’s peroxide curing or, more commonly for medical devices, platinum-catalyzed addition curing – is chosen to ensure purity and minimize the formation of byproducts. Platinum-cured silicones are favored for their superior biocompatibility and stability.
Shaping and Shell Construction
Once the silicone gel or elastomer is formulated, it must be shaped into the final implant form. This is a critical step that demands precision and consistency.
-
Shell Molding: The outer shell of a silicone implant is typically formed using injection molding or compression molding techniques with silicone elastomers. Molds are intricately designed to achieve the exact shape, size, and surface texture required for the implant. For textured implants, the mold surface itself is textured to impart these properties to the shell.
-
Gel Filling: After the shell is molded, it is meticulously filled with the prepared silicone gel. This process requires specialized equipment to ensure the shell is completely and uniformly filled without introducing air bubbles or contaminants. The cohesive nature of modern silicone gels is crucial here, preventing leakage during the filling process.
-
Curing and Finishing: Following filling, the implant undergoes a final curing process to ensure the cross-linking is complete and the material properties are optimized. This is often followed by a washing and sterilization process to prepare the implant for surgical use.
Beyond Silicone: Additional Components and Considerations
While silicone is the primary material, other components and manufacturing considerations contribute to the safety and efficacy of silicone implants. The choice of materials, the design of the implant, and the manufacturing environment all play vital roles.
Purity and Biocompatibility Standards
The paramount concern in the manufacturing of medical implants is biocompatibility – the ability of the material to coexist with biological tissues without eliciting an adverse immune response or toxic reaction. Medical-grade silicones undergo extensive testing to ensure their purity. This includes rigorous analysis to detect and quantify any residual monomers, catalysts, or byproducts from the manufacturing process. Regulatory bodies like the FDA (Food and Drug Administration) in the United States and similar organizations globally set strict standards for the purity and biocompatibility of materials used in medical implants.
Surface Texturing
Some silicone implants feature textured surfaces. This texturing can be achieved through various methods, including roughening the mold surface during the molding process, creating a porous silicone surface, or applying a fine silicone powder. The rationale behind texturing is to reduce the incidence of capsular contracture, a complication where scar tissue forms a tight capsule around the implant, causing hardening and distortion. The textured surface is believed to promote a less organized adherence of scar tissue, allowing for more tissue integration and potentially reducing the risk of displacement.
Saline-Filled Implants (A Comparative Note)
It is worth noting that while the focus here is on silicone implants, there are also saline-filled breast implants. These implants consist of a silicone elastomer shell filled with sterile saline solution. The shell is essentially the same type of silicone elastomer used in silicone gel implants, but the filling is a simple salt-water solution. This comparison highlights that while the outer casing might share similarities, the internal composition significantly differentiates implant types.
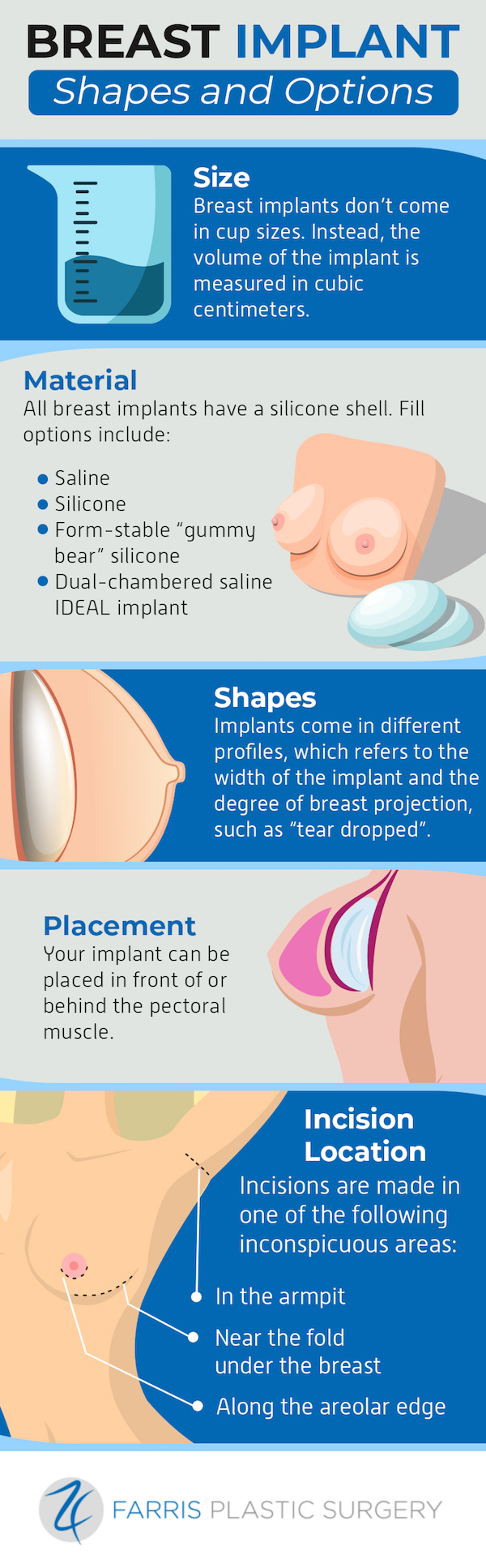
Quality Control and Sterilization
Throughout the entire manufacturing process, stringent quality control measures are implemented. This includes inspections at various stages, material testing, and final product evaluation. Implants are designed for single use and are sterilized before being packaged. The sterilization method is chosen to effectively eliminate microorganisms without compromising the integrity or properties of the silicone materials. Common sterilization methods include autoclaving (steam sterilization) or ethylene oxide gas sterilization, depending on the specific product and its packaging.
In conclusion, silicone implants are sophisticated medical devices made primarily from highly purified, medical-grade silicones. Their composition relies on the unique properties of siloxane polymers, meticulously engineered and processed to ensure safety, durability, and optimal performance. From the strength of the siloxane backbone to the controlled cross-linking of polymers and the precision of molding techniques, every aspect of their manufacture is geared towards providing a biocompatible and reliable solution for a range of medical and aesthetic applications.
