Saturation pressure is a fundamental concept in thermodynamics and fluid dynamics, profoundly impacting a multitude of engineering disciplines, particularly those involving the behavior of gases and liquids. For those involved in the intricate world of flight technology, understanding saturation pressure is not merely academic; it’s crucial for the reliable and efficient operation of sophisticated systems, from atmospheric sensors to onboard propulsion and environmental control. It directly influences the performance of altimeters, the accuracy of weather prediction models used in navigation, and even the integrity of components operating under varying atmospheric conditions.
The Essence of Saturation
At its core, saturation pressure, also known as vapor pressure, refers to the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases (solid or liquid) at a given temperature in a closed system. Imagine a sealed container partially filled with liquid water. As the temperature of the liquid increases, more molecules gain enough kinetic energy to escape from the liquid surface and become vapor. Simultaneously, some vapor molecules will lose energy and condense back into the liquid. Eventually, a state of equilibrium is reached where the rate of evaporation equals the rate of condensation. The pressure exerted by the water vapor at this equilibrium point is the saturation pressure of water at that specific temperature.
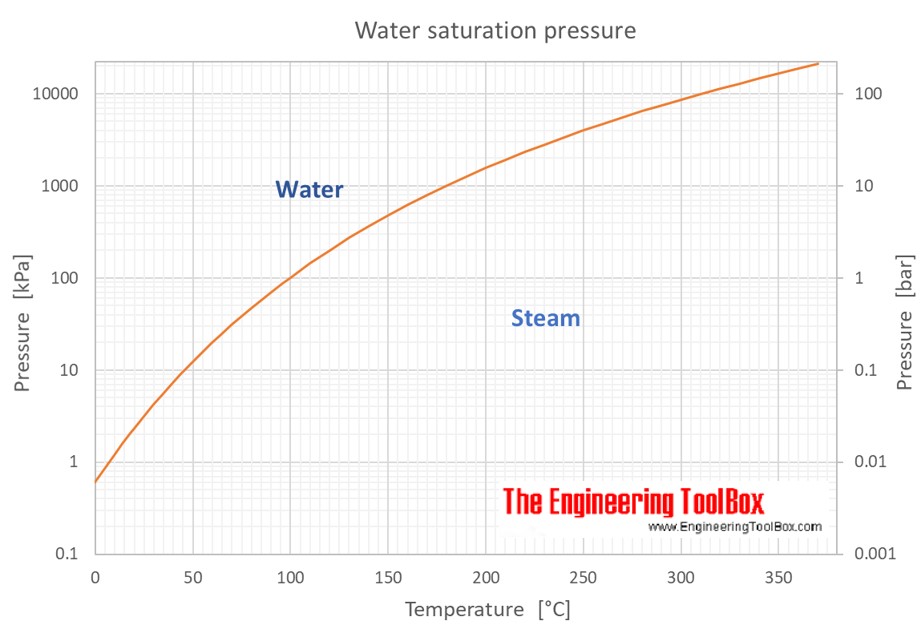
This equilibrium is dynamic; it doesn’t mean that evaporation or condensation has stopped. Instead, the processes are occurring at equal rates, leading to a constant macroscopic state. If more energy is added (temperature increases), the molecules will evaporate faster, and the equilibrium vapor pressure will rise. Conversely, if energy is removed (temperature decreases), condensation will dominate, and the vapor pressure will fall.
Understanding Equilibrium Dynamics
The relationship between temperature and saturation pressure is non-linear and exponential, famously described by the Clausius-Clapeyron relation. This equation quantifies how the saturation pressure of a substance changes with temperature. For many substances, including water, this relationship is characterized by a steep increase in saturation pressure as temperature rises. This is because higher temperatures provide more energy to the molecules, increasing their tendency to transition into the vapor phase.
Consider the phase diagram of a substance. The saturation curve on this diagram represents the locus of points where liquid and vapor coexist in equilibrium. Any point on this curve signifies a specific temperature and its corresponding saturation pressure. Moving away from this curve into the single-phase regions (either purely liquid or purely vapor) signifies conditions where the substance is not at its saturation point.
Factors Influencing Saturation Pressure
While temperature is the primary driver of saturation pressure, it’s important to note that the nature of the substance itself plays a significant role. Different liquids have different intermolecular forces. Substances with weaker intermolecular forces, such as ethanol, tend to have higher saturation pressures at the same temperature compared to substances with stronger forces, like water. This is because less energy is required for their molecules to overcome these forces and enter the vapor phase.
Impurities can also influence saturation pressure, typically by lowering it. This phenomenon is known as boiling point elevation and freezing point depression, which are colligative properties that depend on the concentration of solute particles. In the context of flight technology, understanding these effects is vital when dealing with de-icing fluids or the behavior of atmospheric moisture in the presence of airborne particles.
Saturation Pressure in the Atmosphere
The Earth’s atmosphere is a complex mixture of gases, with water vapor being a crucial component. The behavior of this water vapor is governed by the principles of saturation pressure. Atmospheric saturation pressure refers to the maximum amount of water vapor that the air can hold at a given temperature and pressure.
When the partial pressure of water vapor in the air reaches its saturation pressure at a given temperature, the air is said to be saturated. If more water vapor is added, or if the temperature drops, the excess water vapor will condense into liquid water droplets or ice crystals, forming clouds, fog, or dew. This condensation process is the driving force behind precipitation and is a critical factor in weather forecasting.
Humidity and Saturation
Humidity is a measure of the amount of water vapor in the air, and it is intimately linked to saturation pressure. Relative humidity, a commonly used metric, is the ratio of the actual partial pressure of water vapor to the saturation pressure of water vapor at the same temperature, expressed as a percentage.
- 100% Relative Humidity: When relative humidity is 100%, the air is saturated. Any further increase in water vapor content or decrease in temperature will lead to condensation.
- Below 100% Relative Humidity: The air can hold more water vapor.
- Above 100% Relative Humidity (Supersaturation): This is a transient and unstable state where the air holds more water vapor than it theoretically can at saturation. It typically requires the presence of condensation nuclei (like dust or salt particles) for condensation to occur efficiently.
For flight operations, understanding atmospheric humidity and its relationship to saturation pressure is essential for several reasons. It influences:

- Visibility: High humidity, especially when close to saturation, can lead to fog or low clouds, significantly reducing visibility for aircraft.
- Icing Conditions: In colder atmospheric layers, when the air is near saturation with respect to liquid water, aircraft surfaces can become coated with supercooled water droplets that freeze upon contact, leading to dangerous ice accumulation.
- Aerodynamic Performance: Ice accretion can alter the airfoil shape, reducing lift and increasing drag, potentially leading to loss of control.
- Sensor Accuracy: Many atmospheric sensors, including those used for altitude and weather measurement, are affected by humidity. For instance, altimeters can experience errors due to variations in air density, which is influenced by moisture content.
Dew Point Temperature
The dew point temperature is the temperature to which air must be cooled, at constant pressure and water vapor content, to reach saturation. At the dew point, the partial pressure of water vapor equals the saturation pressure at that temperature. Knowing the dew point is a direct indicator of the amount of moisture in the air. For example, a higher dew point signifies more water vapor content. This is crucial for pilots to assess the risk of fog formation or carburetor icing (in piston-engine aircraft).
Applications in Flight Technology
The principles of saturation pressure are woven into the fabric of modern flight technology, influencing everything from the design of aircraft components to the interpretation of environmental data.
Altimetry and Pressure Sensors
Barometric altimeters, a cornerstone of aviation navigation, function by measuring atmospheric pressure. The relationship between altitude and atmospheric pressure is well-established, but this relationship is influenced by temperature and humidity. For accurate altitude readings, especially in critical phases of flight, altimeters often incorporate temperature and humidity compensation. The concept of saturation pressure is indirectly involved because the density of air, which affects pressure readings, is dependent on its moisture content. Denser, moister air will have a slightly different pressure profile than dry air at the same altitude and temperature.
Advanced pressure sensors used in research aircraft or for atmospheric sensing must account for the vapor pressure of any fluids or gases they are measuring. In a sealed sensor, the presence of water vapor, for instance, will exert its own partial pressure, influencing the overall measured pressure. Understanding saturation pressure allows engineers to calibrate these sensors accurately or to design systems that prevent condensation within sensitive components.
Environmental Control Systems (ECS) and Cabin Pressurization
Modern aircraft maintain a pressurized cabin environment for passenger comfort and safety, simulating an altitude lower than the aircraft’s actual flight altitude. The ECS is responsible for regulating the temperature and humidity within the cabin. This involves complex interplay of air conditioning, heating, and humidification/dehumidification processes.
The saturation pressure of water vapor at various temperatures within the ECS dictates how much moisture the air can hold. For example, to dehumidify the air, it is cooled below its dew point, causing water to condense out. The rate at which this condensation occurs, and the achievable humidity levels, are directly related to the saturation pressure of water vapor at the prevailing temperatures within the system. Conversely, humidifying the air involves adding water vapor until the desired relative humidity is reached, which is again defined relative to the saturation pressure.
Propulsion Systems and Fuel Management
While less direct, saturation pressure can play a role in certain aspects of propulsion. In piston engines, particularly in older designs, carburetor icing can occur when humid air passes through the Venturi of the carburetor. The expansion and cooling of air in the Venturi can drop its temperature below its dew point, leading to condensation and the formation of ice, which can block fuel flow. Understanding the saturation pressure of water vapor at the ambient temperature and pressure helps in assessing the risk of such icing conditions.
In jet engines, while operating at much higher temperatures, the properties of air and its components at extreme conditions are critical. However, the principles of phase changes and vapor pressures are fundamental to understanding the thermodynamics of combustion and the behavior of fuel-air mixtures under various pressures and temperatures.
Weather Radar and Remote Sensing
Weather radar systems, essential for aviation safety, detect precipitation by transmitting radio waves and analyzing the reflected signals. The reflectivity of radar signals is influenced by the size, phase (liquid or ice), and concentration of water particles in the atmosphere. Understanding the conditions under which water vapor will condense into these particles, which is directly governed by saturation pressure and temperature, is crucial for interpreting radar data and forecasting weather phenomena.
Remote sensing instruments on aircraft, used for atmospheric research or Earth observation, also rely on understanding the radiative properties of water vapor. The absorption and emission of electromagnetic radiation by water vapor are strongly dependent on its concentration and phase, which are, in turn, dictated by atmospheric temperature and pressure relative to saturation.

Conclusion
Saturation pressure is a fundamental thermodynamic property that governs the behavior of phase transitions, particularly between liquid and vapor states. For flight technology professionals, grasping this concept is indispensable. It underpins our understanding of atmospheric phenomena, the accurate functioning of critical flight instruments, the design of life-support systems, and the interpretation of complex weather data. From ensuring safe visibility for pilots to designing efficient environmental control systems, the subtle yet pervasive influence of saturation pressure is a constant consideration in the pursuit of safer, more efficient, and more advanced aviation. The ability to predict and manage conditions related to water vapor saturation is a hallmark of sophisticated engineering in the aerospace domain.
