In the intricate world of flight technology, where every component plays a critical role in performance and reliability, understanding the fundamental science behind these elements is paramount. While the term “reduction agent” might sound like it belongs solely in a chemistry textbook, its principles are deeply embedded in the very heart of modern drone accessories—specifically, the batteries that power these advanced aerial vehicles. To fully appreciate the capabilities and limitations of your drone’s energy source, it’s essential to grasp the concept of a reduction agent and its counterpart in electrochemical reactions.
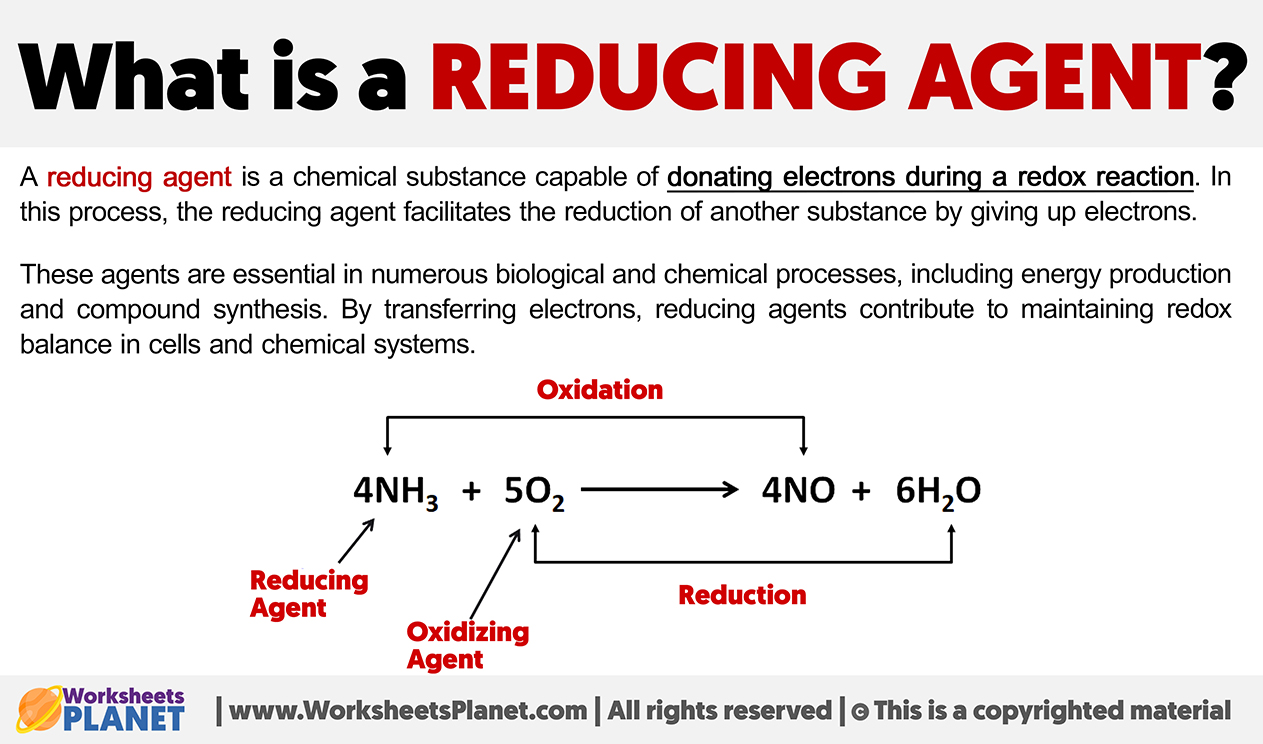
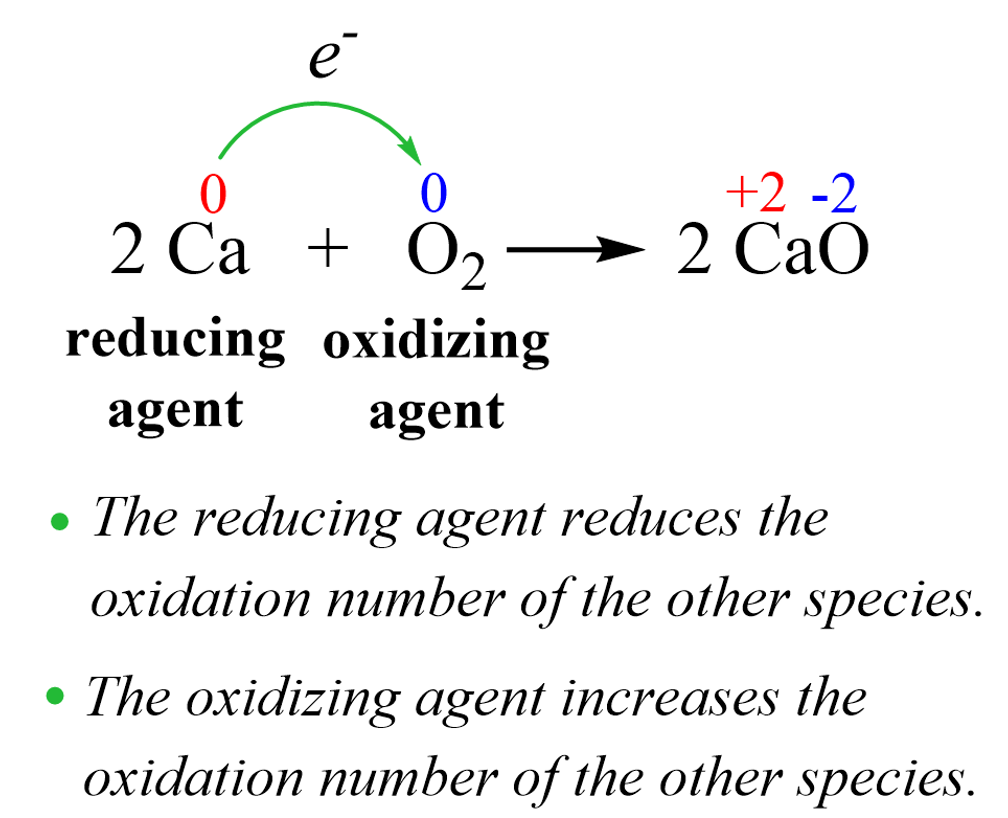
At its core, a reduction agent is a chemical species that facilitates the reduction of another chemical species by losing its own electrons. This process is part of a larger, fundamental chemical reaction known as a redox reaction, a portmanteau for “reduction-oxidation.” In any redox reaction, one substance undergoes oxidation (loses electrons), and another undergoes reduction (gains electrons). The substance that loses electrons and gets oxidized is the reduction agent because it causes the reduction of the other substance. Conversely, the substance that gains electrons and gets reduced is the oxidation agent because it causes the oxidation of the other substance. This delicate ballet of electron transfer is precisely what generates the electrical energy that brings your drone to life.
The Electrochemistry of Drone Batteries: A Redox Symphony
Modern drones predominantly rely on lithium-ion (Li-ion) batteries due to their excellent energy density-to-weight ratio, high voltage per cell, and relatively low self-discharge rate. The operation of a Li-ion battery is a classic example of a carefully controlled redox reaction. Inside every Li-ion cell, there are three primary components: an anode (negative electrode), a cathode (positive electrode), and an electrolyte that separates them while allowing lithium ions to pass.
Deconstructing Oxidation and Reduction in a Li-ion Cell
During discharge, when the battery is providing power to the drone, the following simplified processes occur:
- Anode (Negative Electrode): Typically made from graphite or a silicon-carbon composite, the anode is where the oxidation reaction takes place. Lithium atoms, which are stored within the anode structure, lose electrons and become lithium ions (Li+). These electrons travel through the external circuit (powering the drone) to the cathode. Since the anode material is losing electrons, it is undergoing oxidation, and therefore, the anode material itself acts as the reduction agent. It gives up its electrons to facilitate the reduction reaction at the cathode.
- Cathode (Positive Electrode): Composed of various lithium metal oxides (e.g., LiCoO2, LiFePO4, LiMn2O4, or NMC combinations), the cathode is where the reduction reaction occurs. Lithium ions (Li+) from the anode travel through the electrolyte and intercalate (insert themselves) into the cathode material. Simultaneously, electrons arriving from the external circuit combine with these lithium ions and the cathode material, causing the cathode material to gain electrons and undergo reduction. The cathode material is the oxidation agent because it accepts electrons, causing the anode to oxidize.
- Electrolyte: This is a non-aqueous solvent containing lithium salts. Its primary role is to provide a medium for lithium ions to move between the anode and cathode during charging and discharging, ensuring electrical neutrality within the cell. The electrolyte itself does not participate directly in the redox reactions by gaining or losing electrons.
During charging, the process is reversed. An external power source forces electrons back into the anode, and lithium ions de-intercalate from the cathode, travel through the electrolyte, and re-intercalate into the anode, restoring the battery to its charged state.
The Critical Role of Anode Materials as Reduction Agents
The efficiency and performance of a drone battery are heavily dependent on the characteristics of its reduction agent—the anode material. Graphite has historically been the preferred choice due to its stability, affordability, and ability to reversibly intercalate lithium ions. However, as drone technology demands longer flight times, faster charging, and greater power output, the search for superior reduction agents is a continuous area of innovation in battery research.
Graphite: The Established Reduction Agent

Graphite’s layered structure provides excellent sites for lithium ion storage. When a Li-ion battery discharges, the lithium atoms within the graphite lose an electron (oxidize), transforming into Li+ ions. This electron then travels through the drone’s circuitry to power its motors, flight controller, and cameras. The capacity of a graphite anode is limited by how many lithium ions it can store per unit mass, typically around 372 mAh/g (milliampere-hours per gram). This intrinsic chemical property dictates a fundamental limit on a standard Li-ion battery’s energy density.
Advancing Reduction Agents: The Promise of Silicon
One of the most promising next-generation reduction agents for drone batteries is silicon. Silicon has a theoretical capacity nearly ten times higher than graphite (around 4200 mAh/g), meaning it can store significantly more lithium ions per unit mass. This translates directly into batteries with much higher energy density, potentially allowing for drones with substantially extended flight times without increasing battery size or weight.
However, silicon’s use as a reduction agent comes with challenges:
- Volume Expansion: During lithiation (when lithium ions are absorbed), silicon anodes can expand by up to 300-400%. This massive volume change can cause the anode material to pulverize, leading to a loss of electrical contact and rapid capacity degradation.
- Solid Electrolyte Interphase (SEI) Layer Instability: Repeated expansion and contraction can continuously rupture and reform the SEI layer, a passivation layer that forms on the anode surface. This process consumes active lithium, reducing the battery’s lifespan.
Researchers are working on innovative solutions to these problems, such as using silicon nanoparticles, nanowires, or composites, as well as new electrolyte formulations and binder materials, to stabilize the silicon anode and harness its potential as a superior reduction agent.
Impact on Drone Performance and Longevity
The quality and stability of the reduction agent directly influence key performance metrics for drone batteries:
- Energy Density: A more efficient reduction agent (like silicon) allows for greater energy storage in a smaller, lighter package, directly translating to longer flight times or the ability to carry heavier payloads.
- Power Density: The rate at which the reduction agent can release electrons (i.e., its oxidation kinetics) impacts the battery’s power output. High power density is crucial for responsive throttle control, rapid acceleration, and executing demanding maneuvers.
- Cycle Life: The ability of the reduction agent to undergo repeated oxidation and reduction cycles without significant degradation is vital for battery longevity. Issues like volume expansion or SEI layer instability, which compromise the reduction agent, lead to a reduction in the number of charge/discharge cycles a battery can endure before its capacity drops below acceptable levels.
- Safety: The stability of the reduction agent at various temperatures and states of charge is critical for preventing thermal runaway, a condition where the battery rapidly overheats and can catch fire. Research into safer reduction agents and battery architectures is continuous.
Protecting Your Battery’s Reduction Agent: Best Practices
Understanding the role of the reduction agent also provides insights into proper battery care, which directly impacts its lifespan and performance. The goal is to preserve the integrity of the anode material to ensure it can efficiently store and release lithium ions and electrons over many cycles.
- Avoid Deep Discharges: Constantly draining your battery to 0% can stress the anode material, potentially leading to increased degradation of the reduction agent and the formation of irreversible chemical compounds that reduce capacity.
- Moderate Charging: While fast charging is convenient, it can generate heat and stress the anode, especially if the battery chemistry is not optimized for it. Slower charging rates generally extend the life of the reduction agent.
- Storage Conditions: Store batteries at a moderate state of charge (e.g., 50-60%) and at cool temperatures. Extreme heat or cold can accelerate the degradation of the anode and other battery components.
- Balanced Charging: For multi-cell drone batteries, always use a balance charger. This ensures that each cell, and thus its respective reduction agent, is charged and discharged evenly, preventing individual cells from being overstressed.
In conclusion, while “reduction agent” might seem like an abstract chemical term, its practical implications are fundamental to the operation, performance, and longevity of the batteries that power your drones. From the graphite in today’s ubiquitous Li-ion cells to the promising silicon materials of tomorrow, the efficiency and stability of these electron-donating substances are at the very heart of aerial innovation. A deeper appreciation for this chemical marvel not only enriches your understanding of drone technology but also empowers you to make informed decisions regarding battery care and future tech adoption.
