The QuantiFERON-TB Gold Plus (QFT-Gold Plus) is a modern, in-vitro diagnostic blood test designed to detect latent tuberculosis infection (LTBI). Unlike traditional skin tests, it utilizes advanced immunological principles to identify the body’s immune response to Mycobacterium tuberculosis (M. tuberculosis), the bacterium responsible for tuberculosis (TB). This test offers a more precise and reliable method for diagnosing LTBI, particularly in individuals where traditional methods might be less accurate or pose logistical challenges.
The development of QFT-Gold Plus represents a significant leap forward in TB diagnostics. For decades, the Tuberculin Skin Test (TST) was the primary tool for identifying latent TB. However, the TST has several limitations, including the potential for false-positive results due to BCG vaccination or exposure to non-tuberculous mycobacteria, and a need for a follow-up visit to interpret the results. QFT-Gold Plus aims to overcome these limitations by offering a single-visit blood draw and a more specific detection of the immune response.

Understanding Latent Tuberculosis Infection (LTBI)
Latent tuberculosis infection is a crucial concept when discussing the QFT-Gold Plus test. It’s important to differentiate LTBI from active TB disease.
The Nature of Latent TB
LTBI refers to a state where an individual has been infected with M. tuberculosis but does not currently have active TB disease. In this state, the bacteria are present in the body in an inactive form, typically contained by the immune system. Individuals with LTBI are not contagious and do not exhibit symptoms of active TB. However, the bacteria can remain dormant for years and, under certain conditions, can reactivate and lead to active TB disease.
The World Health Organization (WHO) estimates that about a quarter of the world’s population has latent TB infection. While most infected individuals will never develop active TB, a small percentage, if left untreated, will progress to the disease. Factors that can increase the risk of LTBI progressing to active TB include weakened immune systems due to conditions like HIV/AIDS, diabetes, malnutrition, or the use of immunosuppressant medications, as well as age (very young or elderly individuals).
Why Detecting LTBI is Important
Detecting and treating LTBI is a critical public health strategy for preventing the progression to active TB disease. Treating LTBI can reduce the incidence of new TB cases and contribute to the global effort to eliminate TB. Individuals with LTBI who are at high risk of developing active TB are prime candidates for preventive therapy. Identifying these individuals through tests like QFT-Gold Plus allows for timely intervention, which can significantly alter the course of the infection and protect both the individual and the community.
The Science Behind QuantiFERON-TB Gold Plus
QFT-Gold Plus leverages a sophisticated understanding of the immune system’s response to M. tuberculosis. The test is based on the principle of measuring the release of specific immune signaling molecules called interferons.
Interferon-Gamma Release Assays (IGRAs)
QuantiFERON-TB Gold Plus is classified as an Interferon-Gamma Release Assay (IGRA). IGRAs work by stimulating blood cells with specific antigens that mimic those found in M. tuberculosis. In individuals who have been infected with M. tuberculosis, their T-cells (a type of white blood cell) will recognize these antigens and release interferon-gamma (IFN-γ) when exposed to them in a laboratory setting. The amount of IFN-γ released is then measured.
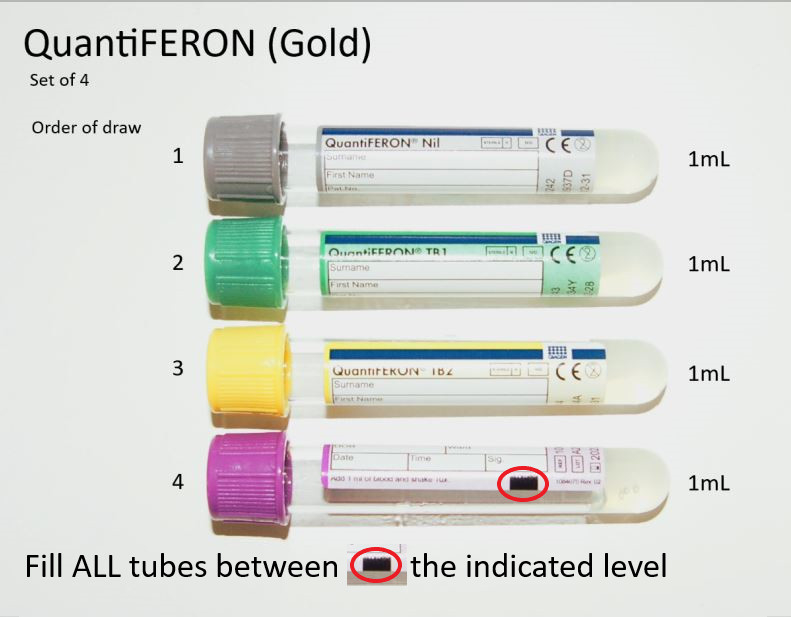
The QFT-Gold Plus test utilizes a specific set of proprietary antigens designed to be highly specific to M. tuberculosis. This specificity helps to minimize cross-reactivity with other mycobacteria, such as those found in the environment or from BCG vaccination. The test includes two specific TB antigen tubes (TB1-Aurora and TB2-Viruses) and one negative control tube. The TB antigen tubes are designed to stimulate cell-mediated immune responses to specific TB antigens. A positive result is indicated by a significant release of IFN-γ in response to these TB antigens, above a certain threshold, while no significant release is observed in the negative control.
How the Test is Performed
The QFT-Gold Plus test is a simple, single-visit blood test. Here’s a general overview of the procedure:
-
Blood Draw: A sample of blood is drawn from the patient, similar to a routine blood test.
-
Incubation: Immediately after collection, the blood is incubated in a laboratory at body temperature (37°C or 98.6°F) for 16 to 24 hours. During incubation, the blood is exposed to:
- TB Antigen Tube 1 (TB1-Aurora): Contains specific TB antigens (ESAT-6 and CFP-10).
- TB Antigen Tube 2 (TB2-Viruses): Contains the same TB antigens as TB1-Aurora, plus a third antigen (Rv2654). This tube is designed to elicit a response from both CD4+ and CD8+ T-cells.
- Nil Control Tube: Does not contain any TB antigens. This tube serves as a baseline to ensure that the laboratory conditions and the patient’s immune cells are functioning normally and to detect any background IFN-γ release.
- Mitogen Control Tube (often included as a separate tube or within the kit): Contains a mitogen (e.g., phytohemagglutinin) that stimulates a broad range of T-cells. This tube helps confirm that the patient’s immune cells are capable of responding to stimulation.
-
Measurement: After incubation, the amount of interferon-gamma released into the plasma from each tube is measured using an enzyme-linked immunosorbent assay (ELISA) technique. The ELISA quantifies the concentration of IFN-γ in each sample.
-
Interpretation: The results are interpreted based on the levels of IFN-γ detected in the TB antigen tubes compared to the Nil control. A significant increase in IFN-γ in the TB antigen tubes, above a defined cutoff, indicates a positive result, suggesting LTBI. The results from both TB antigen tubes are considered, with specific algorithms for interpretation.

Advantages of QuantiFERON-TB Gold Plus
The QFT-Gold Plus test offers several significant advantages over traditional methods for detecting latent TB infection, contributing to its widespread adoption in clinical practice.
Enhanced Specificity and Accuracy
One of the primary advantages of QFT-Gold Plus is its enhanced specificity. The proprietary antigens used in the test are highly specific to M. tuberculosis and have a lower likelihood of cross-reacting with BCG vaccine strains or common environmental mycobacteria. This significantly reduces the incidence of false-positive results often seen with the TST, especially in populations that have received BCG vaccination. The ability to differentiate between infection with M. tuberculosis and exposure to other mycobacteria leads to more accurate diagnoses.
The inclusion of two TB antigen tubes in QFT-Gold Plus, each containing a slightly different combination of antigens, is designed to stimulate a broader range of immune responses. This dual antigen stimulation aims to capture a more comprehensive immune response, potentially increasing sensitivity and providing a more robust confirmation of infection. This nuanced approach contributes to the overall accuracy of the test.
Convenience and Efficiency
QFT-Gold Plus offers a significant improvement in convenience for both patients and healthcare providers.
- Single-Visit Test: Unlike the TST, which requires a second visit for interpretation of the skin reaction, QFT-Gold Plus can be completed with a single blood draw. This saves time and resources, reduces patient inconvenience, and minimizes the risk of patients not returning for a follow-up appointment, thus improving completion rates for testing.
- Objective Measurement: The test provides an objective, quantitative measurement of IFN-γ levels, eliminating the subjective interpretation required for the TST, which can be influenced by the skill of the reader. This objectivity contributes to standardization and reproducibility of results.
- No Booster Effect: The TST can sometimes elicit a “booster effect” in individuals who have been previously exposed to mycobacteria, leading to a positive reaction on a subsequent test that might not reflect true recent infection. QFT-Gold Plus does not cause this booster effect, making it more reliable for serial testing or in situations where previous TB exposure is unknown.
Broader Applicability
The advantages of QFT-Gold Plus make it suitable for a wider range of individuals and clinical scenarios. It is particularly beneficial in populations where BCG vaccination is common or where TST results are difficult to interpret. This includes:
- BCG-vaccinated individuals: QFT-Gold Plus is preferred over TST in these groups due to its specificity.
- Individuals at low risk of TB transmission: When the probability of TB infection is low, a highly specific test is crucial to avoid misdiagnosis.
- Settings with limited resources for follow-up visits: The single-visit nature of the test makes it ideal for various healthcare settings.
- Individuals with a history of TST anergy: Some individuals may have impaired immune responses that lead to false-negative TST results. QFT-Gold Plus can be a more sensitive option in these cases.
Interpretation and Clinical Applications
The interpretation of QFT-Gold Plus results is critical for guiding clinical decisions. The test is used in various settings to identify individuals who may benefit from treatment for latent TB infection.
Understanding Test Results
A QFT-Gold Plus result is typically reported as either “Positive,” “Negative,” or “Indeterminate.”
- Positive Result: Indicates that the individual likely has a latent TB infection. This means they have been infected with M. tuberculosis, but do not currently have active TB disease. A positive result warrants further evaluation to determine if preventive treatment is necessary.
- Negative Result: Suggests that the individual is unlikely to have a latent TB infection. However, a negative result does not completely rule out TB infection, especially in individuals with severe immunosuppression or very recent exposure.
- Indeterminate Result: Means the test could not be reliably interpreted. This can occur due to various reasons, such as problems with blood collection, incubation, or a lack of response in the mitogen control tube (if performed), indicating a problem with the patient’s immune cells or laboratory procedure. In such cases, the test may need to be repeated.
Clinical Scenarios for QFT-Gold Plus Use
The QFT-Gold Plus test plays a vital role in several clinical applications:
- Contact Investigations: When an individual is diagnosed with active TB disease, their close contacts are tested for LTBI. QFT-Gold Plus is a preferred tool for these investigations due to its accuracy and ability to identify infected contacts who may be asymptomatic.
- Screening of High-Risk Populations: Individuals at higher risk of developing TB disease or exposure are often screened for LTBI. This includes healthcare workers, residents of long-term care facilities, individuals with HIV infection, organ transplant recipients, and those on immunosuppressive therapy.
- Pre-employment and Pre-operative Screening: In some professions or before certain medical procedures, screening for LTBI may be required. QFT-Gold Plus provides a reliable method for such screenings.
- Monitoring Treatment Efficacy: While not its primary indication, serial testing with IGRAs might be considered in some research settings or specific clinical scenarios to monitor immune responses to treatment, though this is less common than its diagnostic use.
The decision to test for LTBI and the interpretation of results should always be made by a qualified healthcare professional, considering the individual’s clinical history, risk factors, and local public health guidelines. Treatment of LTBI is typically recommended for individuals who are at high risk of progressing to active TB disease.
