Crude oil, a fossil fuel formed over millions of years from the remains of ancient organisms, is far more than just a source of energy. It’s a remarkably versatile raw material that forms the bedrock of countless industries and products that shape our modern lives. While its primary use as fuel for transportation and electricity generation is widely recognized, its true extent as a feedstock for a vast array of materials is often underestimated. From the plastics that encase our electronics to the synthetic fibers in our clothing, and from the asphalt paving our roads to the essential components of life-saving pharmaceuticals, crude oil’s fingerprints are everywhere. This article delves into the complex process of refining crude oil and explores the diverse range of products it yields, highlighting its indispensable role in contemporary society.
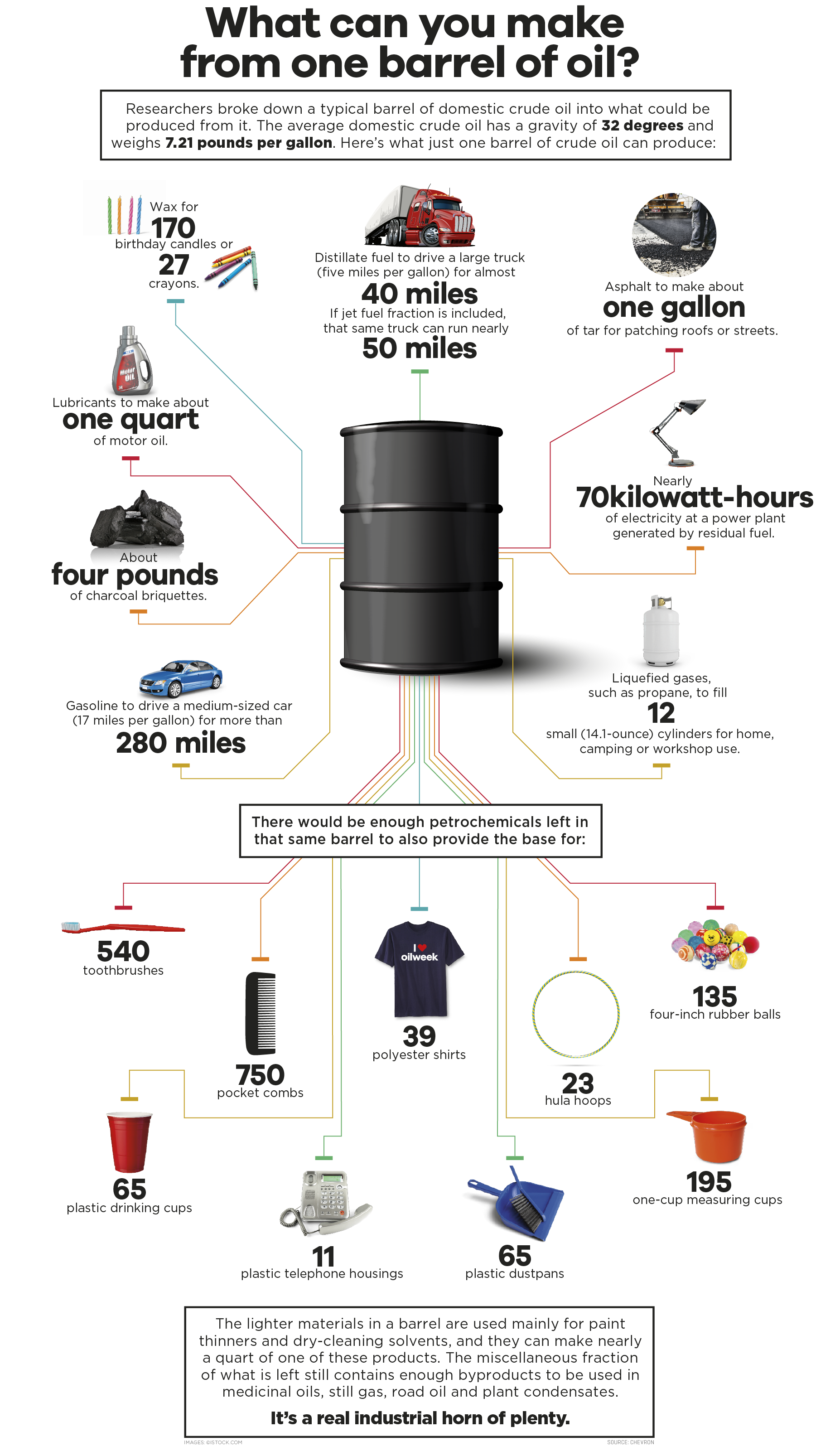
The Alchemy of Refining: Transforming Crude into Usable Components
The journey from a viscous, dark liquid extracted from the earth to the myriad of useful products begins with a sophisticated industrial process known as crude oil refining. This intricate operation involves a series of physical and chemical transformations that separate crude oil into its constituent hydrocarbon components, each with unique properties and applications. The core of this transformation lies in distillation, a process that leverages the different boiling points of these hydrocarbons.
Fractional Distillation: The Primary Separation Technique
Fractional distillation is the cornerstone of crude oil refining. Crude oil is heated to high temperatures and then introduced into a distillation column. As the vapor rises, it cools, and different hydrocarbons condense at specific temperature ranges, separating into distinct fractions. Lighter, more volatile hydrocarbons with lower boiling points rise higher in the column, while heavier, less volatile hydrocarbons with higher boiling points condense lower down.
- Gases: At the very top of the column, gases like methane, ethane, propane, and butane are collected. These are often used as liquefied petroleum gas (LPG) for heating and cooking, or as feedstocks for the petrochemical industry.
- Naphtha: This fraction, which boils at temperatures between gasoline and kerosene, is a crucial intermediate product. It’s primarily used as a feedstock for producing gasoline through further processing (reforming) and as a key ingredient in the production of petrochemicals, forming the basis for plastics and other organic chemicals.
- Gasoline (Petrol): This is one of the most well-known products, refined to specific octane ratings for use in internal combustion engines. Its production is a major focus of most refineries.
- Kerosene: This fraction, also known as paraffin, is used as jet fuel, for heating and lighting, and as a solvent.
- Diesel Fuel: Heavier than kerosene, diesel fuel powers diesel engines found in trucks, buses, trains, and some passenger cars.
- Lubricating Oils: Further down the column, heavier fractions yield lubricating oils used in engines, machinery, and industrial applications to reduce friction.
- Fuel Oil: This viscous oil is used as fuel for ships, industrial furnaces, and power plants.
- Bitumen (Asphalt): At the very bottom of the distillation column are the heaviest, least volatile residues. Bitumen is the primary component of asphalt used for paving roads and roofing.
Secondary Processing: Enhancing and Creating New Products
While fractional distillation separates crude oil into basic fractions, further processing is often necessary to enhance the quality of these products, increase their yield, or convert them into more valuable substances. These secondary processes are crucial for meeting the diverse demands of the market.
- Cracking: This process breaks down larger, heavier hydrocarbon molecules into smaller, lighter ones. Thermal cracking uses high temperatures and pressures, while catalytic cracking employs catalysts to achieve the same result at lower temperatures. Cracking significantly increases the yield of gasoline and other lighter fuels from heavier crude oil fractions.
- Reforming: This process rearranges the molecular structure of hydrocarbon molecules to increase their octane rating, making them suitable for use as high-performance gasoline. This is particularly important for producing premium unleaded fuels.
- Alkylation and Polymerization: These processes combine smaller hydrocarbon molecules to create larger ones, often to produce high-octane gasoline components or valuable petrochemical feedstocks. Polymerization, in particular, is crucial for creating the long chains of molecules that form plastics.
- Treating: Various treating processes are employed to remove impurities such as sulfur, nitrogen, and metals from the refined products. This is essential for meeting environmental regulations and improving product quality. For example, desulfurization is critical to reduce emissions of sulfur dioxide when fuels are burned.
The Petrochemical Revolution: From Fuel to Foundation Materials
The true extent of crude oil’s utility is revealed when we consider the petrochemical industry. Petrochemicals are chemical products derived from petroleum. While many of the fractions from crude oil distillation can be used directly as fuels, they also serve as fundamental building blocks for an astounding array of synthetic materials. This transformation is achieved through processes that break down or rearrange hydrocarbon molecules to create simpler compounds, which are then used to synthesize more complex substances.
Olefins and Aromatics: The Cornerstones of Petrochemicals
The primary petrochemical feedstocks are derived from naphtha and lighter fractions through processes like steam cracking. This process involves heating hydrocarbons in the presence of steam to very high temperatures, breaking them down into smaller, more reactive molecules. The most important products of steam cracking are olefins and aromatics.
- Olefins: These are hydrocarbons containing at least one carbon-carbon double bond. The most significant olefins are:
- Ethylene: The most produced organic compound in the world. It is the primary building block for polyethylene, the most common plastic used in everything from plastic bags and bottles to films and pipes. Ethylene is also used to produce ethylene oxide (used in antifreeze and detergents), vinyl chloride (for PVC), and ethylbenzene (for styrene).
- Propylene: The second most important olefin, used to make polypropylene (a versatile plastic used in packaging, textiles, and automotive parts), acrylonitrile (for synthetic fibers and plastics), and propylene oxide (for polyurethanes).
- Butadiene: A key ingredient in the production of synthetic rubber, used in tires and other rubber products.
- Aromatics: These are hydrocarbons containing one or more benzene rings. The main aromatics are:
- Benzene: A fundamental building block for a vast range of chemicals. It’s used to produce styrene (for polystyrene plastics and synthetic rubber), phenol (for resins and disinfectants), and cyclohexane (for nylon).
- Toluene: Used as a solvent and in the production of benzene, TNT, and other chemicals.
- Xylenes: Used in solvents and in the production of polyester fibers and films.
Polymers and Plastics: Shaping Our Modern World

The olefins and aromatics derived from crude oil are the direct precursors to the vast family of polymers and plastics that permeate our daily lives. Polymerization is the chemical process of linking together many small molecules (monomers) to form a long chain molecule (a polymer).
- Polyethylene (PE): Derived from ethylene, PE is ubiquitous. Its different forms (HDPE, LDPE, LLDPE) offer varied properties, making it suitable for everything from grocery bags and milk jugs to piping and toys.
- Polypropylene (PP): Made from propylene, PP is known for its strength, heat resistance, and flexibility. It’s found in automotive parts, carpets, furniture, and food packaging.
- Polyvinyl Chloride (PVC): Produced from vinyl chloride (derived from ethylene and chlorine), PVC is a durable and versatile plastic used in pipes, window frames, flooring, and electrical insulation.
- Polystyrene (PS): Made from styrene, PS is commonly used in disposable cutlery, CD cases, insulation foam (Styrofoam), and packaging.
- Polyethylene Terephthalate (PET): A polyester formed from ethylene glycol and terephthalic acid (both petrochemical derivatives), PET is widely used for beverage bottles and synthetic fibers (polyester).
- Nylon: A family of synthetic polymers produced from monomers like adipic acid and hexamethylenediamine, both derived from petrochemicals. Nylon is renowned for its strength and elasticity, used in textiles, ropes, and industrial components.
- Polyurethanes: These versatile polymers, created using isocyanates and polyols (both petrochemicals), are found in foams for furniture and insulation, coatings, adhesives, and elastic fibers.
Beyond Plastics: The Diverse Applications of Crude Oil Products
The impact of crude oil extends far beyond the realm of plastics. The refined fractions and petrochemical derivatives form the basis of countless other essential products that underpin our infrastructure, health, and comfort. Understanding these applications highlights the pervasive nature of crude oil in modern industrial economies.
Synthetic Fibers and Textiles
The clothing we wear, the carpets we walk on, and the upholstery in our homes are often made from synthetic fibers derived from crude oil. These fibers offer advantages like durability, wrinkle resistance, and affordability.
- Polyester: As mentioned, PET is a key component of polyester fibers, widely used in clothing, home furnishings, and industrial textiles due to its strength and resilience.
- Nylon: Its high tensile strength makes it ideal for hosiery, activewear, ropes, and even parachute fabric.
- Acrylic: Produced from acrylonitrile, acrylic fibers offer a wool-like feel and are used in sweaters, blankets, and carpets.
- Spandex (Elastane): Known for its exceptional elasticity, spandex is made from polyurethane and is blended with other fibers to create stretch fabrics for activewear and form-fitting clothing.
Solvents, Detergents, and Fertilizers
Many everyday cleaning products and agricultural essentials owe their existence to crude oil. Petrochemical processes create the chemical building blocks for these vital substances.
- Solvents: Various hydrocarbon fractions and their derivatives, such as toluene, xylene, and acetone, are used as powerful solvents in paints, varnishes, adhesives, and industrial cleaning agents.
- Detergents: The surfactants that enable soaps and detergents to lift dirt and grease are often synthesized from petrochemical intermediates like linear alkylbenzene (LAB).
- Fertilizers: While not a direct product of distillation, natural gas (often found alongside oil deposits) and some heavier oil fractions are critical feedstocks for producing ammonia, a key component of nitrogen-based fertilizers essential for modern agriculture.
Pharmaceuticals and Cosmetics
The pharmaceutical and cosmetic industries rely heavily on petrochemical derivatives for synthesizing active ingredients, excipients, and functional components.
- Pharmaceuticals: Many life-saving drugs, from aspirin to complex antibiotics and chemotherapy agents, have synthetic pathways that begin with petrochemical intermediates. These provide the precise molecular structures needed for therapeutic effects.
- Cosmetics: Petrochemicals are used to create a wide range of ingredients in cosmetics, including emollients, emulsifiers, preservatives, fragrances, and colorants. Vaseline (petroleum jelly) itself is a direct petroleum byproduct.

Other Essential Products
The list of products derived from crude oil continues to expand, encompassing numerous other categories:
- Asphalt and Bitumen: As mentioned, the heaviest residues are used for road construction and roofing, forming the durable surfaces we rely on daily.
- Waxes: Paraffin wax, derived from crude oil, is used in candles, food packaging, and polishes.
- Lubricants: Beyond simple oils, a vast array of specialized lubricants for automotive, industrial, and aviation uses are refined from crude oil.
- Pesticides and Herbicides: Many agricultural chemicals designed to protect crops are synthesized using petrochemical feedstocks.
- Adhesives and Sealants: The bonding agents and protective sealants used in construction and manufacturing often have petrochemical origins.
In conclusion, crude oil is a fundamental resource that fuels our world in more ways than one. Its transformation through sophisticated refining and petrochemical processes yields an astonishing diversity of products, from the fuels that power our transportation to the advanced materials that form the backbone of modern manufacturing, healthcare, and consumer goods. While the world increasingly seeks sustainable alternatives, understanding the pervasive and essential role of crude oil in our current technological and material landscape remains critically important.
