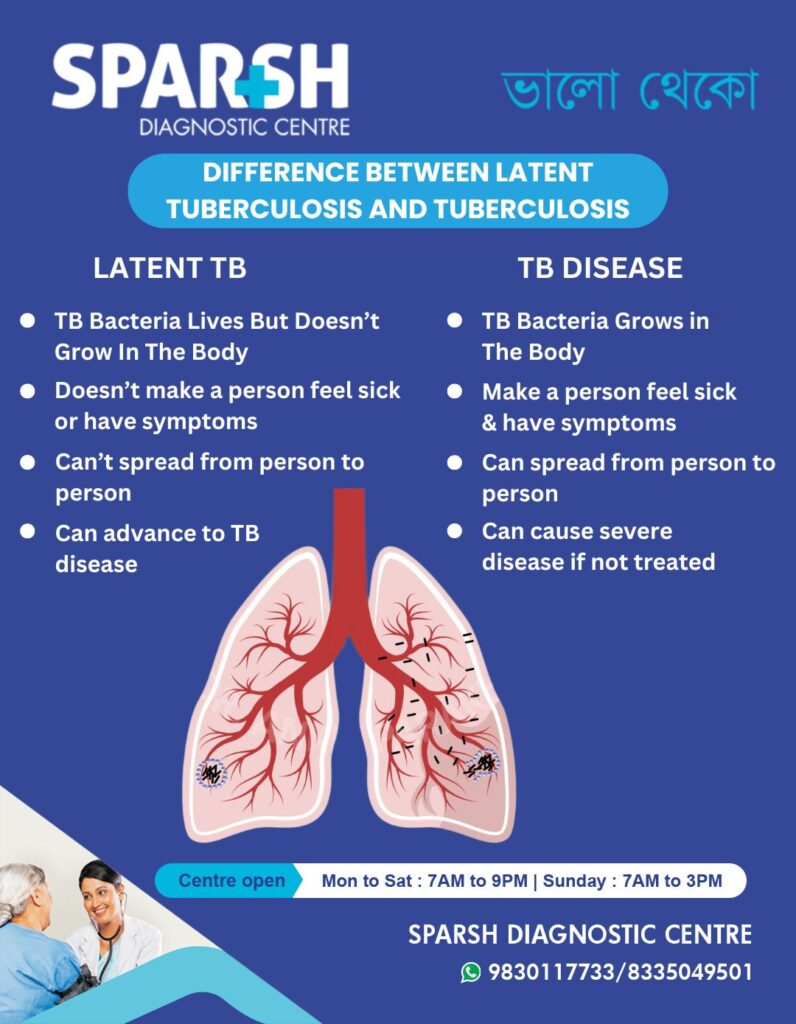
Latent tuberculosis infection (LTBI) is a critical aspect of understanding and ultimately eradicating tuberculosis (TB). While often overshadowed by active TB disease, recognizing and managing LTBI is paramount for public health strategies worldwide. LTBI refers to a state where an individual is infected with Mycobacterium tuberculosis but does not exhibit any symptoms of active disease. Their immune system has effectively contained the bacteria, preventing them from multiplying and causing illness. However, these individuals can still harbor dormant bacteria within their bodies.
The significance of LTBI lies in its potential to reactivate into active TB disease. This reactivation can occur years, or even decades, after the initial infection, often triggered by a weakened immune system. Factors that can compromise immunity and increase the risk of LTBI progression include HIV infection, organ transplantation, treatment with immunosuppressive medications (such as corticosteroids or tumor necrosis factor inhibitors), certain cancers, and conditions like diabetes and malnutrition. Understanding LTBI is therefore not just about identifying current infections but also about mitigating future disease burden.
Understanding the Infection Process
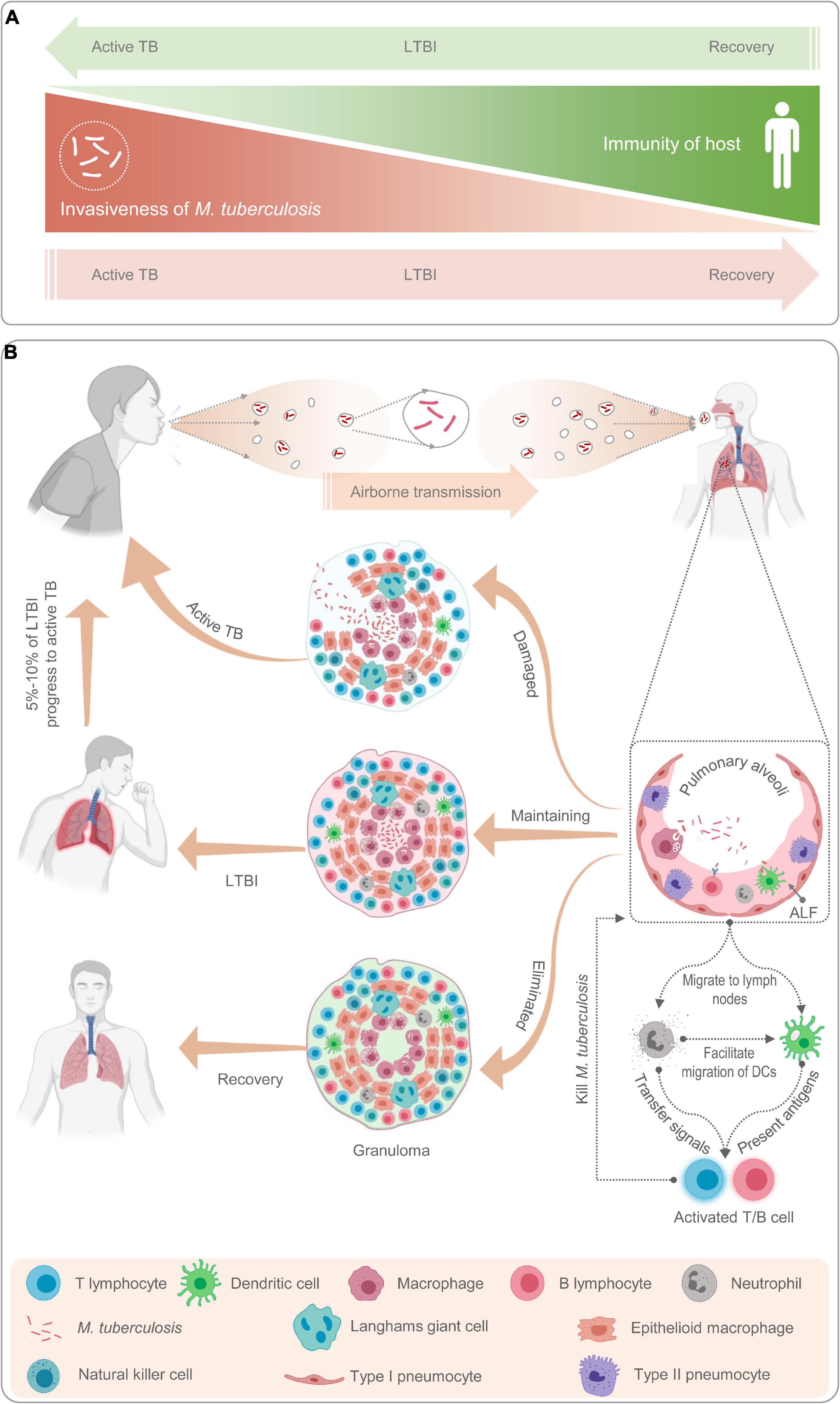
The journey from exposure to Mycobacterium tuberculosis to LTBI involves a complex interplay between the bacteria and the host’s immune system. When an individual inhales infectious aerosolized droplets containing live TB bacteria, these microorganisms typically travel to the lungs.
Initial Exposure and Immune Response
Upon reaching the alveoli, the tiny air sacs in the lungs where gas exchange occurs, the TB bacteria begin to multiply. The body’s first line of defense, primarily the innate immune system, attempts to contain this invasion. Macrophages, a type of white blood cell, engulf the bacteria. However, Mycobacterium tuberculosis has evolved sophisticated mechanisms to survive and replicate within these macrophages.
As the infection progresses, the adaptive immune system is activated. T lymphocytes, crucial components of cell-mediated immunity, are stimulated to recognize and target the infected cells. This immune response leads to the formation of granulomas, which are organized collections of immune cells, including macrophages, lymphocytes, and epithelioid cells, that encapsulate the bacteria. These granulomas act as a containment barrier, preventing further bacterial spread.
Latency vs. Active Disease
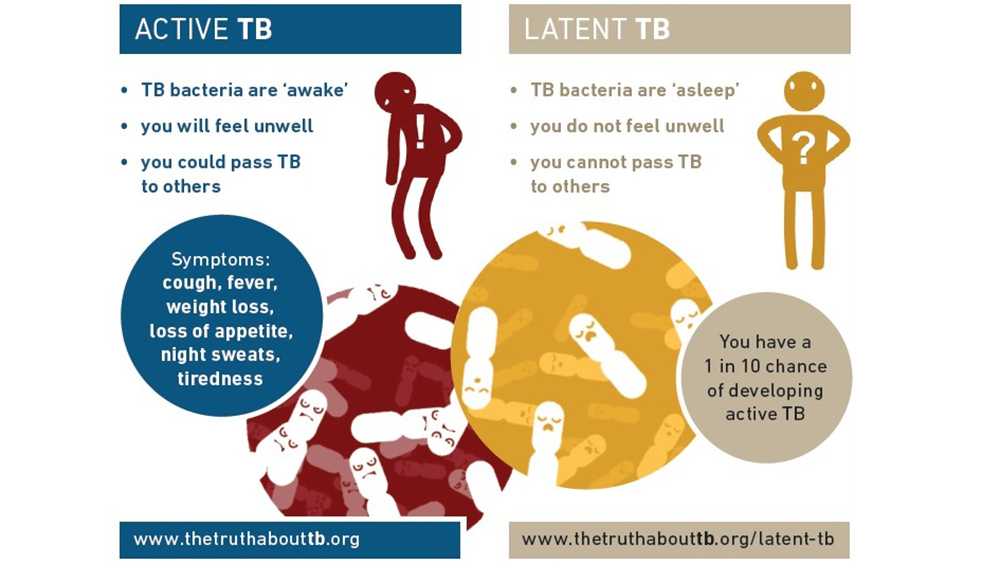
In individuals with a robust immune system, the granulomas successfully wall off the TB bacteria, leading to a state of latency. The bacteria remain viable but are metabolically dormant, meaning they are not actively replicating. This is LTBI. The individual experiences no symptoms, is not infectious to others, and a standard chest X-ray may appear normal.
However, if the immune system weakens, the containment provided by the granulomas can falter. The dormant bacteria can then reactivate, begin to multiply uncontrollably, and break free from the granuloma. This leads to active TB disease, where the bacteria can damage lung tissue and spread to other parts of the body. Symptoms of active TB include persistent cough, fever, night sweats, unexplained weight loss, and fatigue.
Diagnosis of Latent Tuberculosis Infection
Diagnosing LTBI presents a unique challenge. Since individuals with LTBI are asymptomatic and their chest X-rays are typically normal, traditional methods used for diagnosing active TB are not sufficient. The goal of LTBI diagnosis is to identify individuals who are infected but not yet ill, thereby allowing for preventive treatment.
Interferon-Gamma Release Assays (IGRAs)
Interferon-gamma release assays (IGRAs) are the preferred diagnostic tools for LTBI in many high-income countries. These blood tests measure the release of interferon-gamma (IFN-γ), a cytokine produced by T lymphocytes when they are exposed to specific TB antigens.
The process involves drawing a blood sample from the individual. This blood is then incubated with two specific TB antigens: early secretory antigenic target 6 (ESAT-6) and culture filtrate protein 10 (CFP-10). If the individual has been infected with TB bacteria, their T cells will release IFN-γ in response to these antigens. The amount of IFN-γ released is then measured.
There are two main types of IGRAs: QuantiFERON-TB Gold Plus (QFT-Plus) and T-SPOT.TB. Both assays have high specificity, meaning they are less likely to produce false positive results in individuals who have been vaccinated with the BCG vaccine (which can cause false positives with the older TST). However, IGRAs, like other LTBI tests, can have false negatives, particularly in individuals with severely compromised immune systems. It is important to note that a positive IGRA result indicates TB infection but does not distinguish between LTBI and active TB disease.
Tuberculin Skin Test (TST)
The tuberculin skin test (TST), also known as the Mantoux test, is another widely used method for diagnosing LTBI. It is an older test but remains a valuable tool, especially in resource-limited settings where IGRAs may not be readily available.
The TST involves injecting a small amount of purified protein derivative (PPD) tuberculin intradermally, usually on the forearm. PPD contains proteins derived from Mycobacterium tuberculosis. If an individual has been infected with TB bacteria, their immune system will have developed T cells that recognize these proteins.
After 48 to 72 hours, the injection site is checked for a reaction. A raised, hardened bump (an induration) indicates a positive TST. The size of the induration that is considered positive varies depending on the individual’s risk factors for TB exposure or progression. For example, a smaller induration may be considered positive in individuals with a high risk of exposure or progression, while a larger induration is needed for a positive result in lower-risk individuals.
Challenges with the TST include the possibility of false positives due to BCG vaccination or exposure to non-tuberculous mycobacteria. False negatives can occur due to improper testing technique, weakened immune systems, or recent TB infection where the immune response has not yet developed. Like IGRAs, a positive TST result indicates TB infection but does not differentiate between LTBI and active TB.
Treatment of Latent Tuberculosis Infection
The primary goal of treating LTBI is to prevent the progression to active TB disease. This preventive therapy, also known as preventive treatment or isoniazid preventive therapy (IPT), is a crucial component of global TB control efforts. By treating individuals with LTBI, the risk of them developing active TB later in life is significantly reduced.
Rifampin Preventive Therapy (RPT)
While isoniazid (INH) has been the cornerstone of LTBI treatment for decades, shorter-course regimens are increasingly being used, offering improved adherence and reduced treatment duration. Rifampin preventive therapy (RPT) is one such option.
RPT typically involves taking rifampin, a potent antibiotic, for a shorter duration, often four months. This regimen has shown to be highly effective in preventing TB disease and is particularly beneficial for individuals who may not tolerate isoniazid or who have been exposed to drug-resistant TB. It is also a preferred option for certain populations, such as children and individuals living with HIV.
Isoniazid Preventive Therapy (IPT)
Isoniazid preventive therapy (IPT) has long been the standard of care for LTBI. It involves taking isoniazid, an antibiotic specifically effective against TB, daily or twice weekly for a duration of six to nine months. IPT has been proven to reduce the risk of developing active TB by approximately 60-90% in individuals with LTBI.
However, IPT can be associated with side effects, most notably liver toxicity (hepatitis). Therefore, individuals undergoing IPT require regular monitoring for signs and symptoms of liver damage, including jaundice, fatigue, and abdominal pain. Alcohol consumption should be minimized during IPT due to an increased risk of liver toxicity.
Combination Therapies
In certain situations, combination therapies may be recommended for LTBI. For example, in individuals with a higher risk of developing drug-resistant TB, a combination of isoniazid and rifampin may be prescribed. Newer regimens are also being investigated to further improve treatment effectiveness and shorten durations. The choice of treatment regimen depends on various factors, including the individual’s risk profile, potential drug interactions, and the availability of specific medications.
Public Health Implications and Future Directions
Addressing LTBI is a critical, yet often complex, public health imperative. Effective strategies for identifying and treating LTBI are essential for achieving the global goal of TB elimination.
Screening and Targeted Interventions
Identifying individuals at highest risk of LTBI progression is key to successful interventions. This includes close contacts of individuals with active TB, people living with HIV, individuals on immunosuppressive therapy, and populations residing in high-prevalence settings. Targeted screening programs allow for the proactive identification of infected individuals before they develop active disease.
Challenges and Innovations
Despite advancements, significant challenges remain in managing LTBI. These include the stigma associated with TB infection, which can lead to reluctance in seeking testing and treatment; the need for long treatment durations, impacting adherence; and the complexities of drug resistance.
Future directions in LTBI management focus on developing more effective and shorter treatment regimens, improving diagnostic tools that can differentiate between LTBI and active TB, and enhancing public awareness and education. The development of novel vaccines that can prevent initial infection or prevent progression from LTBI to active TB is also a crucial area of research. By a comprehensive approach encompassing diagnosis, treatment, and ongoing research, the burden of TB can be substantially reduced, ultimately working towards its global eradication.
