At the heart of bone biology lies a microscopic world of intricate structures and specialized cells, fundamental to skeletal health and function. Among these, lacunae stand out as critical, albeit often overlooked, components. Biologically, lacunae are minute, spindle-shaped cavities within the hard, mineralized matrix of bone tissue, each housing a single osteocyte – the primary cell type responsible for maintaining bone. These osteocytes, nestled within their lacunae, are interconnected by an elaborate network of even finer channels called canaliculi, facilitating nutrient exchange and waste removal, and enabling intercellular communication across the bone matrix. While these biological facts form the bedrock of our understanding, it is through the lens of modern Tech & Innovation that we truly unlock profound insights into lacunae, transforming our ability to visualize, analyze, and ultimately manipulate these structures for improved human health.
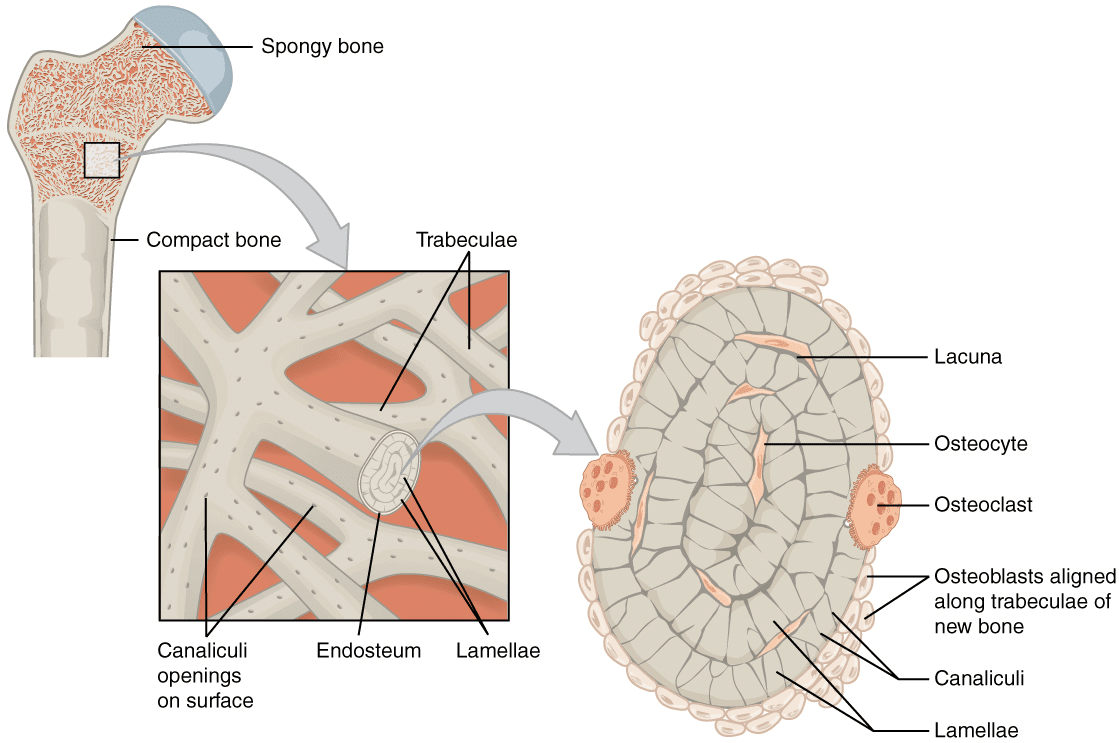
The journey from a rudimentary understanding of bone microstructure to advanced diagnostics and regenerative strategies is paved with technological advancements. From high-resolution imaging that reveals their three-dimensional architecture to sophisticated artificial intelligence algorithms that predict their health implications, and even bio-engineering techniques that aim to replicate them, technology has revolutionized the study of lacunae. This article explores how cutting-edge innovation is pushing the boundaries of our knowledge, offering new perspectives on the vital role of lacunae in bone maintenance, disease progression, and future therapeutic interventions.
Visualizing the Microscopic: Advanced Imaging Techniques for Lacunae
For centuries, our understanding of bone microstructure was largely confined to two-dimensional histological sections, providing snapshots rather than comprehensive views. The inherent limitations of such methods in capturing the true complexity and spatial arrangement of lacunae prompted a relentless pursuit of more advanced imaging technologies. Today, thanks to significant leaps in biomedical imaging, we can peer into the bone matrix with unprecedented clarity, revealing the intricate world of lacunae in exquisite detail.
Beyond Traditional Histology: Micro-CT and Synchrotron Imaging
The advent of micro-computed tomography (micro-CT) has been a game-changer in visualizing bone microstructure. Unlike traditional histology which requires tissue processing, embedding, sectioning, and staining—all potentially introducing artifacts—micro-CT offers a non-destructive, three-dimensional reconstruction of bone samples. This technology utilizes X-rays to generate thousands of 2D projection images from various angles around a rotating sample. These projections are then computationally reconstructed into a 3D volume, allowing researchers to accurately identify, quantify, and analyze individual lacunae and their interconnected canalicular networks.
Micro-CT provides crucial metrics such as lacunar density, volume, shape, and spatial distribution, all of which are vital indicators of bone health and remodeling activity. Its ability to provide quantitative data on a large scale makes it indispensable for studying age-related changes, disease progression (like osteoporosis or osteonecrosis), and the efficacy of therapeutic interventions on a structural level. Resolutions can reach down to sub-micrometer levels, making it possible to resolve individual lacunae which typically range from 10-30 micrometers in length.
Pushing the boundaries further, synchrotron X-ray microtomography offers even higher spatial resolution and contrast. Synchrotron facilities produce extremely bright, monochromatic, and coherent X-ray beams, enabling researchers to visualize bone at the sub-micrometer scale with greater precision than conventional micro-CT. This ultra-high resolution is critical for studying the finest details of the lacunar-canalicular network, including the canaliculi themselves (which are only hundreds of nanometers in diameter), and understanding their contribution to nutrient transport and mechanical sensing within the bone matrix. The insights gained from synchrotron imaging are instrumental in understanding fundamental mechanobiology and pathological changes at the cellular level within bone.
Non-Invasive Diagnostics: MRI and Ultrasound Innovations
While micro-CT and synchrotron imaging are powerful research tools, their application in routine clinical diagnostics for assessing lacunae is limited due to factors like invasiveness (biopsy required for ex vivo analysis) or radiation exposure for in vivo studies. However, Magnetic Resonance Imaging (MRI) and advanced ultrasound technologies are rapidly evolving to offer non-invasive methods that could indirectly inform us about the state of lacunae.
Innovations in MRI sequences, such as ultra-short echo time (UTE) imaging, are designed to pick up signals from cortical bone, which is typically “invisible” to standard MRI due to its short T2 relaxation times. By enhancing our ability to image the hard bone matrix, these techniques hold promise for characterizing bone density and microstructure changes associated with conditions that affect lacunar health. While directly resolving individual lacunae in vivo with MRI remains a significant challenge, advancements in high-field MRI and specialized pulse sequences are continually improving its diagnostic potential for assessing overall bone integrity and composition, which is intrinsically linked to the health of its cellular inhabitants.
Similarly, high-frequency ultrasound innovations are being explored for their potential in bone characterization. Traditionally used for imaging soft tissues, specialized quantitative ultrasound techniques are now being developed to assess bone density, elasticity, and microarchitecture. By analyzing the propagation and scattering of high-frequency sound waves through bone, researchers aim to derive parameters that reflect changes in bone quality, potentially including aspects related to the integrity of the lacunar-canalicular system. While still largely in experimental stages for this specific application, the non-ionizing nature and portability of ultrasound make it an attractive candidate for future non-invasive assessments of bone microstructure, opening new avenues for monitoring lacunar health in clinical settings.
The Power of AI and Machine Learning in Bone Analysis
The vast quantities of data generated by advanced imaging techniques present both an opportunity and a challenge. Manually analyzing thousands of 3D images to quantify lacunae is time-consuming, prone to human error, and limits the scale of research. This is where Artificial Intelligence (AI) and Machine Learning (ML) step in, revolutionizing the way we process, interpret, and derive meaning from complex bone microstructure data, particularly concerning lacunae.
Automating Detection and Quantification
One of the most immediate and impactful applications of AI in bone analysis is the automated detection, segmentation, and quantification of lacunae from high-resolution images. Deep learning models, particularly convolutional neural networks (CNNs), can be trained on large datasets of annotated bone scans (e.g., micro-CT or synchrotron images) to accurately identify individual lacunae. These algorithms can then precisely measure their dimensions (length, width, volume), determine their spatial orientation, calculate their density per unit volume, and even assess their connectivity within the canalicular network.
The benefits of this automation are multifaceted: it dramatically increases the speed of analysis, ensures objectivity and consistency across studies, and allows researchers to process datasets that would be impossible to handle manually. Moreover, AI can often detect subtle patterns and variations in lacunar morphology that might be imperceptible to the human eye, potentially leading to the discovery of new biomarkers for bone diseases. This objective, high-throughput analysis is crucial for large-scale epidemiological studies, drug screening, and the rapid evaluation of new bone-targeting therapies.
Predictive Analytics for Bone Health and Disease
Beyond mere quantification, AI and ML algorithms are being leveraged for powerful predictive analytics in the realm of bone health. By integrating lacunar characteristics (derived from automated analysis) with clinical data, patient demographics, genetic markers, and lifestyle factors, machine learning models can be trained to predict various outcomes.
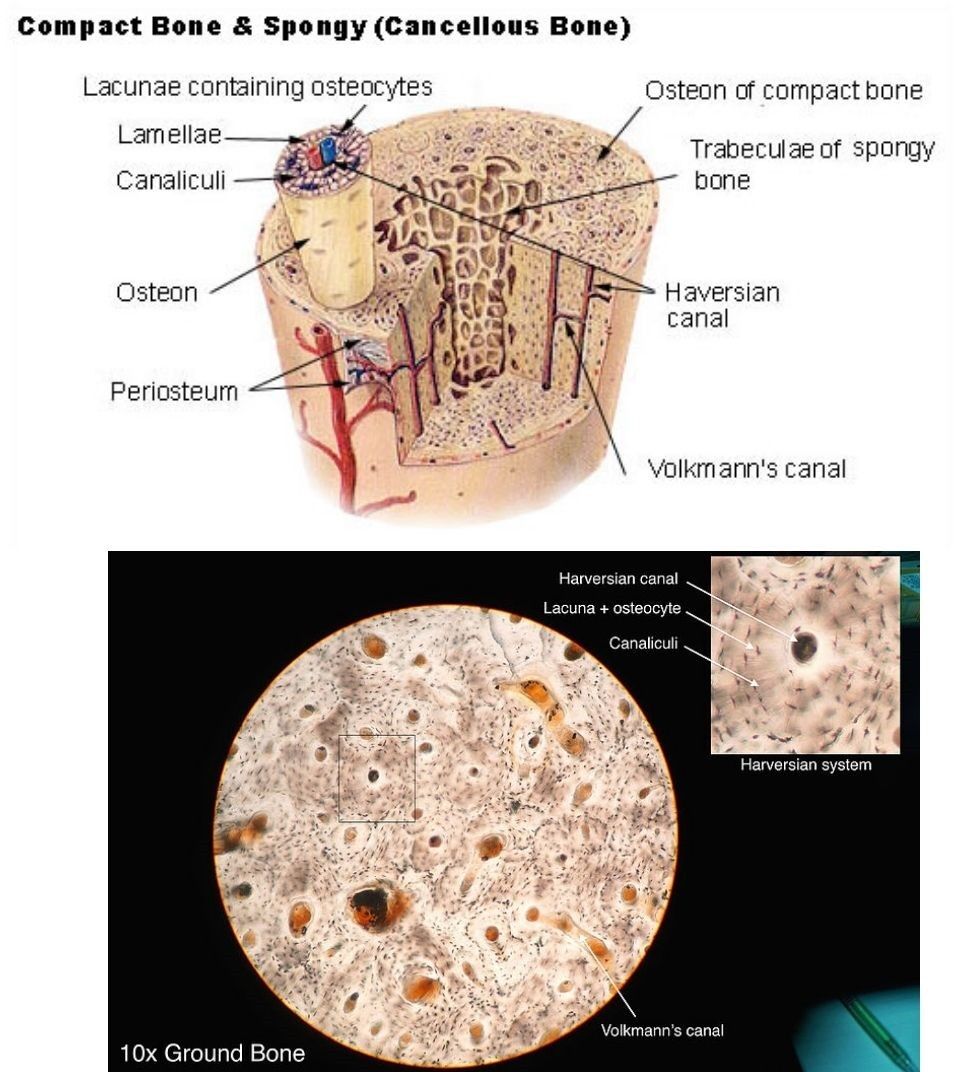
For example, models can predict an individual’s risk of developing osteoporosis, the likelihood of fracture, or the progression rate of diseases like osteonecrosis or osteoarthritis, based on specific lacunar signatures. Changes in lacunar size, shape, or density might serve as early indicators of metabolic dysfunction within bone, allowing for earlier intervention. Furthermore, AI can help tailor personalized treatment strategies by predicting a patient’s response to specific drugs or interventions based on their unique bone microstructure profile. This move towards personalized medicine, where therapeutic decisions are guided by an individual’s specific cellular and microstructural characteristics, represents a significant paradigm shift, offering more effective and targeted treatments for bone disorders.
Computational Modeling and Simulation of Lacunar Networks
Understanding the static morphology of lacunae is one thing; comprehending their dynamic role within the living bone is another. Computational modeling and simulation offer a powerful virtual laboratory to explore the complex biomechanical and biochemical functions of lacunar networks, providing insights that are difficult or impossible to obtain through experimental methods alone.
Biomechanical Simulation and Stress Distribution
Bone is a dynamic tissue that constantly adapts to mechanical loads. Osteocytes within their lacunae are considered the primary mechanosensors of bone, detecting changes in mechanical stress and initiating remodeling responses. Computational models allow researchers to create highly detailed, patient-specific virtual representations of bone architecture, incorporating the precise geometry and spatial arrangement of individual lacunae and their interconnecting canaliculi.
Using techniques like finite element analysis (FEA), these models can simulate the distribution of mechanical forces and strains throughout the bone matrix at a microscopic level. By applying virtual loads, researchers can predict how different stresses are transmitted through the lacunar-canalicular network, how osteocytes within their lacunae perceive these stresses, and how these signals might influence overall bone strength, adaptation, and fracture risk. This allows for a deeper understanding of mechanotransduction – the process by which mechanical stimuli are converted into biochemical signals – and its disruption in various bone diseases. Furthermore, these simulations can be used to test the biomechanical effects of new implant designs or therapeutic interventions on the bone microstructure, including the load-bearing capacity and potential impact on lacunar health.
Fluid Flow and Nutrient Transport Dynamics
Beyond mechanical sensing, the lacunar-canalicular network is crucial for the transport of nutrients, oxygen, and signaling molecules to and from the osteocytes, as well as for the removal of waste products. Given the dense, avascular nature of the bone matrix, this microfluidic system is vital for osteocyte viability and bone homeostasis. Computational fluid dynamics (CFD) models are employed to simulate the intricate patterns of fluid flow within these nanoscale channels.
These models consider factors such as pressure gradients generated by mechanical loading, the viscosity of interstitial fluid, and the geometry of the canaliculi to predict flow rates and nutrient concentrations around individual osteocytes. Understanding these dynamics is critical, as disruptions in fluid flow (e.g., due to microcracks, aging, or reduced mechanical loading) can lead to osteocyte apoptosis (cell death), which is a significant factor in the initiation and progression of bone diseases like osteoporosis and osteonecrosis. By simulating these processes, researchers can investigate how pathological conditions affect nutrient delivery and waste clearance, and explore potential interventions to restore healthy fluid flow, thereby improving osteocyte survival and overall bone health.
Future Frontiers: Bio-Engineering and Regenerative Approaches
The profound insights gained from advanced imaging, AI, and computational modeling of lacunae are not merely academic; they are paving the way for revolutionary advancements in bio-engineering and regenerative medicine. The ultimate goal is not just to understand lacunae but to harness this knowledge to repair, regenerate, and even create functional bone tissue.
3D Bioprinting of Bone Scaffolds
One of the most exciting areas of innovation is 3D bioprinting, which allows for the creation of complex, multi-cellular tissue constructs with precise architectural control. Advances in bioinks and bioprinting technologies are enabling the fabrication of biomimetic bone scaffolds that closely mimic the natural microstructure of bone, including the sophisticated lacunar-canalicular network.
Researchers are developing techniques to bioprint scaffolds with engineered pores and channels designed to house osteocytes within “artificial lacunae” and facilitate their connection through printed canaliculi. This controlled microenvironment allows for precise cell seeding and encourages the formation of functional bone tissue. The ability to create personalized, patient-specific bone grafts with tailored microarchitectures holds immense promise for regenerative medicine, addressing issues like large bone defects from trauma, cancer resection, or congenital abnormalities. By integrating living cells within engineered lacunae, these bioprinted constructs aim to not only provide structural support but also to actively participate in bone remodeling and integration with the host tissue.
Smart Materials and Targeted Therapies
The future of lacunae-centric therapies also lies in the development of smart materials and nanotechnology. Smart biomaterials are being engineered to respond to specific physiological cues (e.g., pH changes, enzymatic activity, mechanical stress) within the bone microenvironment, allowing for controlled drug release or activation of regenerative processes. Imagine materials that could detect signs of osteocyte distress within their lacunae and release targeted therapeutic agents to promote cell survival or stimulate repair.
Nanotechnology, specifically, offers unparalleled precision for targeted drug delivery. Nanoparticles can be engineered to specifically home in on bone tissue and even target osteocytes within their lacunae. This capability could lead to highly localized treatments for conditions affecting osteocytes, such as inhibiting apoptosis in osteonecrosis or delivering pro-anabolic factors to stimulate bone formation in osteoporosis, minimizing systemic side effects. Moreover, novel sensing nanoparticles could be developed to monitor the metabolic health of osteocytes within their lacunae in situ, providing real-time feedback on bone health and treatment efficacy. These future innovations represent a convergence of materials science, biology, and advanced engineering, promising a new era of highly effective and personalized bone therapies.

Conclusion
The humble lacunae, microscopic cavities housing the vital osteocytes, are far more than mere anatomical curiosities. They are central to bone’s mechanical integrity, metabolic regulation, and its remarkable ability to adapt and repair. While our fundamental understanding of these structures is rooted in traditional biology, it is the relentless march of Tech & Innovation that has truly illuminated their profound significance.
From the exquisite 3D visualizations provided by micro-CT and synchrotron imaging, allowing us to map their complex architecture, to the predictive power of AI and machine learning that deciphers their role in disease, and the transformative potential of computational models simulating their dynamic functions, technology is fundamentally reshaping bone biology. Looking ahead, the integration of bio-engineering, particularly 3D bioprinting of biomimetic scaffolds with engineered lacunae, and the advent of smart materials and targeted nanotherapies, promises a future where we can not only understand lacunae but actively engineer and manipulate them for unprecedented therapeutic outcomes. The continuous evolution of technology ensures that our insights into “what is lacunae in bone” will forever expand, driving us towards a future of healthier bones and enhanced quality of life.
