The Fundamental Principle: Electron Removal and Atomic Stability in Advanced Tech
At the heart of every technological advancement, from the incredibly lightweight frames of racing drones to the sophisticated processors guiding autonomous flight, lies a profound understanding of atomic behavior. Among these foundational principles, ionization energy stands out as a critical determinant of how elements interact, bond, and ultimately perform within complex systems. Simply put, ionization energy (IE) is the minimum energy required to remove one electron from a gaseous atom in its ground state. This energy quantifies how tightly an atom holds onto its outermost electrons, providing crucial insight into an element’s reactivity, its tendency to form ions, and its suitability for various engineering applications.
For instance, an element with a low ionization energy readily loses an electron, often forming a positive ion (cation) and engaging in metallic bonding or electrochemical reactions. Conversely, an element with high ionization energy strongly retains its electrons, making it less likely to form cations and more prone to forming covalent bonds or acting as an electron acceptor. In the realm of drone technology and innovation, these atomic-level propensities translate directly into material properties such as conductivity, strength, ductility, and reactivity, which are paramount for designing durable, efficient, and high-performance components. Understanding the underlying principles of ionization energy allows engineers to make informed choices about materials, optimizing everything from power sources to structural integrity.
Periodic Trends: Predicting Material Behavior for Drone Innovation
The true power of ionization energy lies not just in its definition but in its predictable patterns across the periodic table—its “trend.” These trends enable scientists and engineers to anticipate the chemical and physical properties of elements, informing the design of novel materials and advanced systems.
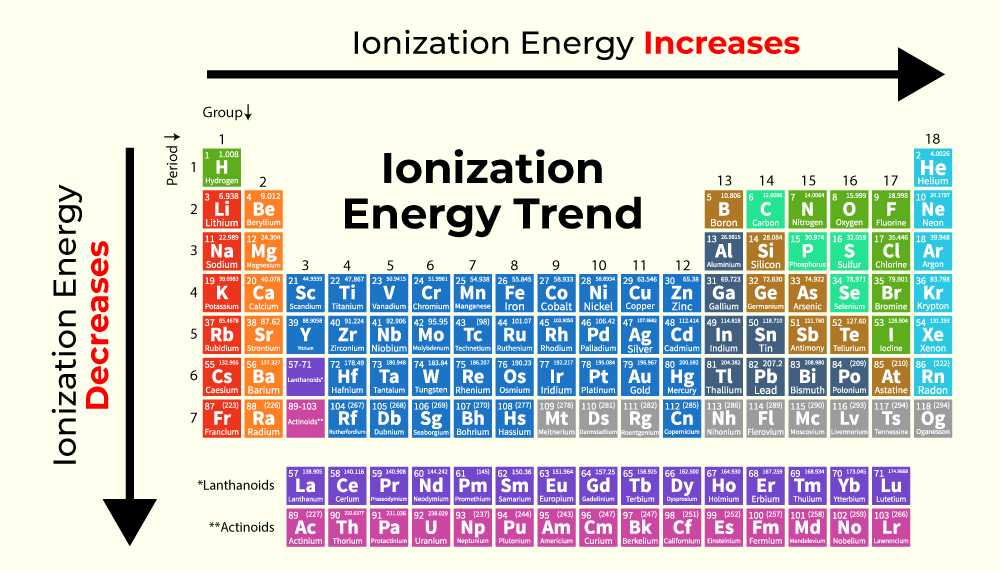
Trends Across a Period (Left to Right)
As one moves from left to right across a period (row) of the periodic table, the ionization energy generally increases. This is primarily because atoms accrue more protons in their nuclei, leading to a stronger positive charge that pulls the electrons closer and more tightly towards the nucleus. Despite the addition of electrons, they occupy the same principal energy level, meaning the shielding effect (where inner electrons block the attraction of the nucleus for outer electrons) does not significantly increase. The result is a greater effective nuclear charge experienced by the valence electrons, requiring more energy to remove them. This trend explains why alkali metals on the far left of the table (like Lithium, crucial for batteries) have very low ionization energies and are highly reactive, while noble gases on the far right (like Neon) possess exceptionally high ionization energies, making them chemically inert. This fundamental difference drives choices for conductors versus insulators, and highly reactive battery components versus stable structural elements.
Trends Down a Group (Top to Bottom)
Conversely, moving down a group (column) in the periodic table generally leads to a decrease in ionization energy. While atoms gain more protons down a group, they also gain additional electron shells. These new shells are progressively further from the nucleus, and the increasing number of inner electrons provides a more significant shielding effect. The combined effect of increased distance and enhanced shielding means the outermost valence electrons experience a weaker attraction to the nucleus. Consequently, less energy is required to remove these electrons. For example, Sodium, below Lithium in the periodic table, has an even lower ionization energy, making it potentially more reactive but also hinting at different electrochemical properties relevant for future energy storage. This trend is vital for understanding the varying conductivity and reactivity within families of elements, from metals used in wiring to the rare earth elements essential for drone motors and sensors.
Factors Influencing Ionization Energy
Beyond the general periodic trends, several other factors fine-tune ionization energy values:
- Atomic Radius: A larger atomic radius implies valence electrons are further from the nucleus, leading to lower IE.
- Nuclear Charge: A greater number of protons (higher nuclear charge) pulls electrons more strongly, increasing IE.
- Shielding Effect: Inner electrons “shield” outer electrons from the full nuclear charge; greater shielding reduces IE.
- Electron Configuration: Half-filled or fully-filled subshells exhibit extra stability, leading to slightly higher ionization energies than expected due to the energy required to disrupt this stable configuration. This quantum mechanical nuance explains subtle deviations in the general trends but is critical for understanding specific material properties, such as the stability of certain alloys or semiconductor doping characteristics.
Ionization Energy’s Pervasive Influence in Drone Tech & Innovation

The practical implications of ionization energy trends are far-reaching, directly impacting the materials and systems that define modern drone technology. From enhancing structural integrity to pushing the boundaries of power storage, a deep understanding of these atomic properties is a cornerstone of innovation.
Advancing Lightweight Materials: From Aluminum to Composites
The pursuit of lightweight yet robust materials is constant in drone development. Elements with low ionization energies, such as aluminum, are excellent choices for drone frames due to their metallic bonding, which allows for ductility, malleability, and good conductivity while maintaining a reasonable strength-to-weight ratio. Aluminum’s relatively low IE means its valence electrons are delocalized, forming an “electron sea” that provides strong, yet flexible, metallic bonds. However, for even greater strength-to-weight ratios, engineers turn to advanced composites like carbon fiber. Carbon, with a higher ionization energy than metals, forms strong covalent bonds, leading to rigid, incredibly strong materials essential for high-performance drone bodies and propellers. Understanding the IE trends helps in selecting elements for alloys and composite matrices that optimize strength, stiffness, corrosion resistance, and thermal properties – crucial for drones operating in diverse environments. For instance, the atomic structure and electron configuration, influenced by IE, dictate how these materials respond to stress, vibration, and temperature fluctuations during aggressive flight maneuvers.
Revolutionizing Battery Chemistry and Energy Storage
Perhaps nowhere is the impact of ionization energy more evident than in the evolution of drone batteries. Lithium, an alkali metal with an exceptionally low first ionization energy, is the backbone of most high-performance drone batteries. Its eagerness to shed an electron makes it an ideal anode material, providing a strong driving force for electrochemical reactions that generate power. The ability of Lithium ions to readily give up electrons (low IE) and accept them back (forming neutral atoms) is precisely why Li-ion batteries offer high energy density.
Innovations in battery technology often involve exploring new chemistries that balance energy density, power output, safety, and longevity. This includes researching alternative anode materials (e.g., silicon alloys), cathode materials (which often involve transition metals with varying ionization energies and oxidation states), and solid-state electrolytes. The ionization energies of these constituent elements dictate the potential voltage, charge/discharge rates, and cycle life of the battery. For instance, the selection of cathode materials relies on elements that can efficiently accept electrons, often exhibiting higher electron affinities and ionization energies, enabling robust electron transfer mechanisms within the cell. Future advancements in drone battery technology, such as solid-state batteries or metal-air batteries, will continue to rely on a precise understanding of the ionization energy trends of novel elements and compounds to maximize their performance and safety profiles.
Enhancing Semiconductor Performance and Electronic Systems
Drones are intricate flying computers, relying on sophisticated electronic systems for navigation, control, communication, and data processing. The core of these systems lies in semiconductors, materials whose electrical conductivity can be precisely controlled. Silicon, the most common semiconductor, is intrinsically an insulator at room temperature. Its utility arises from “doping”—introducing small impurities of other elements to alter its electrical properties.
The choice of dopants is directly governed by their ionization energies relative to silicon. To create n-type semiconductors (which have an excess of free electrons), elements with slightly lower ionization energies than silicon (e.g., Phosphorus or Arsenic) are used. These donor atoms readily “donate” an extra electron to the silicon lattice, enhancing conductivity. Conversely, for p-type semiconductors (which have an excess of “holes,” or electron vacancies), elements with slightly higher ionization energies (e.g., Boron or Gallium) are introduced. These acceptor atoms readily “accept” an electron from the silicon lattice, creating a hole. This precise manipulation of electron availability, dictated by ionization energy, is fundamental to creating transistors, microprocessors, and sensors that enable AI-powered flight, real-time data processing, and robust communication links in modern drones. As drones become more autonomous and intelligent, the demand for faster, more efficient, and smaller electronic components will only grow, pushing the boundaries of semiconductor design through atomic-level engineering.
Future Materials and Extreme Environments
Looking ahead, understanding ionization energy trends will be pivotal in developing materials for drones designed for extreme environments—be it high altitudes, polar regions, or even extraterrestrial exploration. Materials with specific ionization energy profiles can be engineered for enhanced thermal stability, radiation resistance, or specific electromagnetic properties. For instance, designing robust coatings that prevent corrosion in salty marine environments, or creating components that maintain structural integrity and electronic functionality under intense radiation, relies on selecting elements whose electron configurations and ionization energies lend themselves to these demanding conditions. The pursuit of high-temperature superconductors for more efficient motors or advanced shielding for sensitive electronics will also invariably involve a deep dive into the ionization energy landscape of candidate elements and compounds, pushing the frontier of drone capabilities into previously unimaginable territories.
Conclusion: The Unseen Force Driving Drone Evolution
The seemingly abstract concept of ionization energy trend is, in fact, an indispensable tool for engineers and innovators in the drone industry. It serves as an unseen force, guiding the selection and design of every material, every battery, and every electronic component that contributes to a drone’s performance and capabilities. From the selection of lightweight alloys to the optimization of battery chemistries and the intricate engineering of semiconductor devices, a thorough grasp of how atoms gain or lose electrons—and the energy required for these transformations—is fundamental. As drone technology continues its rapid evolution, pushing the boundaries of autonomy, endurance, and application, the principles of ionization energy will remain at the forefront of scientific inquiry, underpinning the next generation of aerial innovation.
