High-Performance Liquid Chromatography (HPLC) is a powerful analytical technique used across a vast array of scientific disciplines for the separation, identification, and quantification of components within a liquid sample. At its core, HPLC is a sophisticated form of column chromatography that employs high pressure to force the sample mixture through a stationary phase, which is packed into a column. This high pressure is the key differentiator from traditional liquid chromatography, enabling faster and more efficient separations with greater resolution.
The fundamental principle of HPLC relies on the differential partitioning of sample components between a mobile phase (a liquid solvent or mixture of solvents) and a stationary phase (a solid material packed within the column). As the mobile phase carries the sample through the column, components that have a stronger affinity for the stationary phase will move slower, while those with a weaker affinity will be swept along more quickly by the mobile phase. This difference in migration rates leads to the separation of the mixture into its individual constituents.
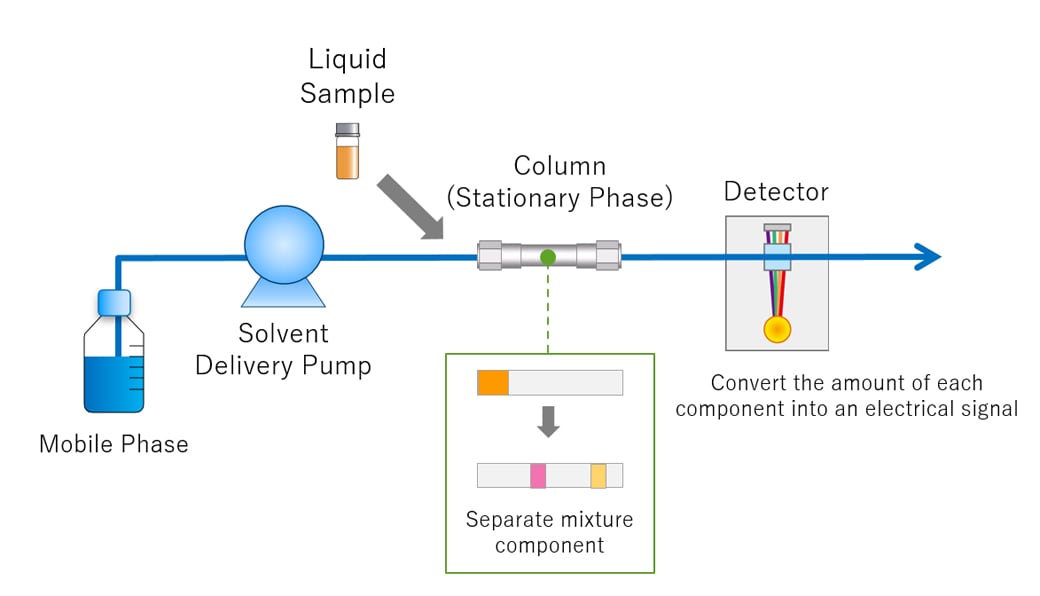
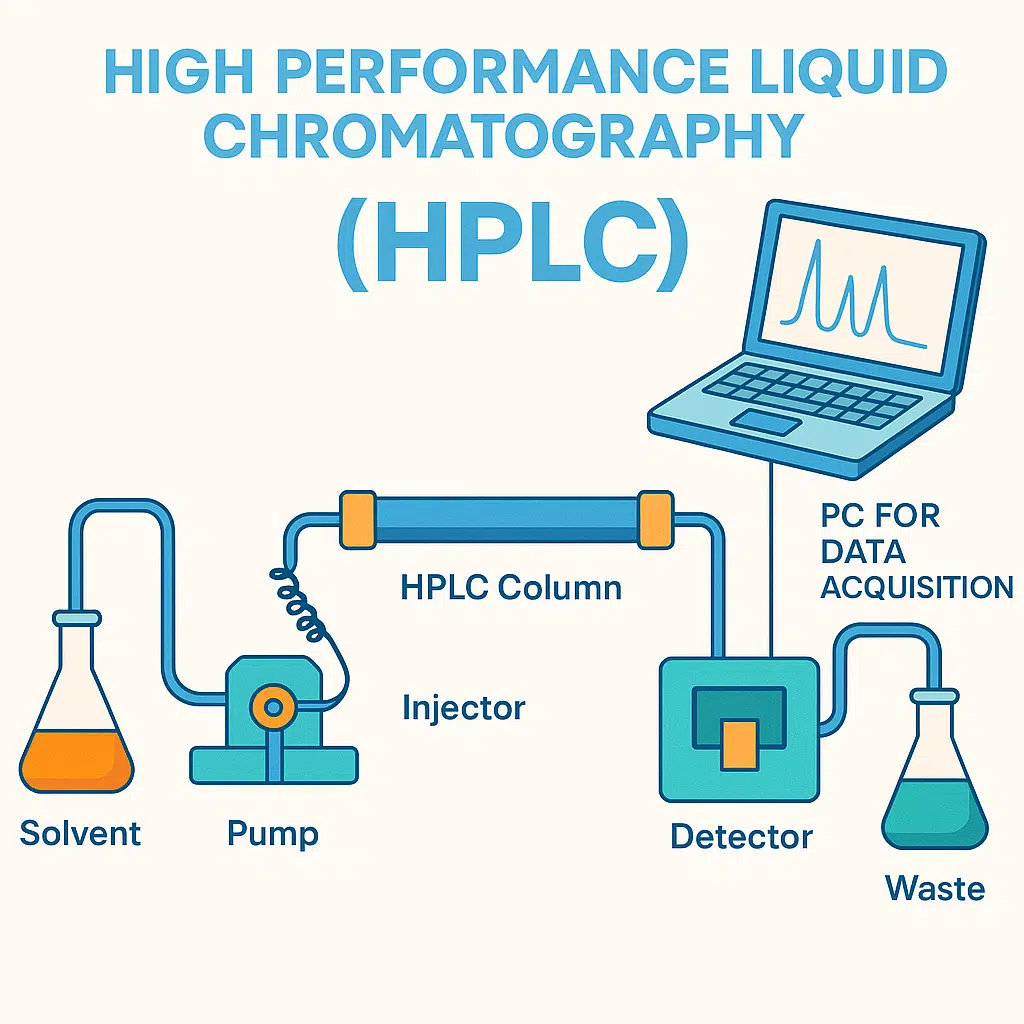
The Core Components of an HPLC System
An HPLC system is a complex assembly of interconnected components, each playing a vital role in the chromatographic process. Understanding these individual parts is crucial to appreciating how HPLC achieves its remarkable analytical capabilities.
The Mobile Phase and Solvent Delivery System
The mobile phase is the liquid vehicle that carries the sample through the HPLC column. Its composition, flow rate, and pressure are meticulously controlled to achieve optimal separation. Common mobile phases include water, organic solvents like methanol and acetonitrile, and various buffer solutions.
The solvent delivery system, often referred to as the pump, is the heart of the HPLC instrument. Its primary function is to deliver the mobile phase to the column at a constant and precise flow rate, typically ranging from 0.1 to 10 mL/min. Modern HPLC systems utilize high-pressure pumps, such as reciprocating or diaphragm pumps, capable of generating pressures up to several hundred bar (or even thousands of bar for UHPLC – Ultra-High-Performance Liquid Chromatography). These pumps must be able to deliver solvents without pulsation, as any variation in flow can significantly impact the separation and the resulting chromatogram.
The Injector (Autosampler or Manual Injector)
The injector is responsible for introducing a precise and reproducible volume of the sample into the mobile phase stream. This introduction must occur at the beginning of the chromatographic run without disrupting the mobile phase flow or introducing air bubbles.
Manual Injectors: These require the analyst to manually introduce the sample via a syringe into a loop. While simpler, they are prone to variations in injection volume and are less suitable for high-throughput analysis.
Autosamplers: These automated systems are designed for high-throughput laboratories. They can store a large number of samples in vials and inject them sequentially into the system with exceptional accuracy and precision. Autosamplers offer programmability for injection volume, flow rates, and other parameters, significantly improving laboratory efficiency and reproducibility.
The Column
The column is where the actual separation of the sample components takes place. It is a precisely engineered tube, typically made of stainless steel, packed with the stationary phase. The dimensions of the column, the type of stationary phase material, and the particle size of the packing material are critical factors that influence the separation efficiency and resolution.
Stationary Phase: The stationary phase is the material packed within the column that interacts with the sample components. The choice of stationary phase is paramount and depends on the nature of the analytes being separated. Common stationary phases include:
- Silica-based materials: These are the most widely used and can be chemically modified to alter their properties.
- Reversed-phase silica: The most common type, where the silica surface is bonded with hydrophobic organic chains (e.g., C18, C8). Separation is based on the hydrophobicity of the analytes.
- Normal-phase silica: Unmodified or lightly modified silica with polar surface groups. Separation is based on polarity, with polar analytes retained longer.
- Ion-exchange silica: Functionalized with charged groups to separate ionic compounds.
- Chiral stationary phases: Designed to separate enantiomers.
- Polymer-based materials: Offer different selectivity and can be more stable in certain solvent systems.
- Size exclusion chromatography (SEC) packing: Porous particles used to separate molecules based on their size.
The particle size of the stationary phase is crucial. Smaller particles provide a larger surface area for interaction, leading to increased efficiency and resolution, but also require higher pressures to overcome the resistance to flow.
The Detector

The detector is responsible for sensing the separated components as they elute from the column and generating a signal proportional to their concentration. The choice of detector depends on the properties of the analytes and the required sensitivity.
Common HPLC Detectors:
- UV-Visible (UV-Vis) Detector: The most ubiquitous detector, it measures the absorbance of light by the eluting analytes at specific wavelengths. It is suitable for compounds that absorb UV or visible light. Diode Array Detectors (DAD) or Photodiode Array (PDA) detectors are advanced UV-Vis detectors that can acquire full UV-Vis spectra of eluting peaks, allowing for peak purity assessment and identification.
- Refractive Index (RI) Detector: Measures changes in the refractive index of the mobile phase as analytes elute. It is a universal detector, capable of detecting most compounds, but it is less sensitive than UV-Vis detectors and is highly sensitive to temperature fluctuations and mobile phase composition changes.
- Fluorescence Detector: Highly sensitive and selective, it is used for compounds that naturally fluoresce or can be derivatized to become fluorescent.
- Mass Spectrometry (MS) Detector: Provides highly specific and sensitive detection by measuring the mass-to-charge ratio of ionized analytes. HPLC-MS is a powerful hyphenated technique that combines the separation power of HPLC with the identification capabilities of MS.
- Evaporative Light Scattering Detector (ELSD): Detects non-volatile analytes by nebulizing the eluent, evaporating the mobile phase, and then detecting the remaining analyte particles by light scattering.
- Charged Aerosol Detector (CAD): Similar in principle to ELSD but detects charged aerosol particles, offering improved sensitivity for a wider range of analytes.
The Data Acquisition and Processing System (Chromatograph Data System – CDS)
The CDS, typically a computer with specialized software, is responsible for controlling the HPLC instrument, acquiring the signal from the detector, and processing the resulting data. The output from the detector is a chromatogram, a plot of detector response versus time. Each peak in the chromatogram represents a separated component of the sample.
The CDS software allows for:
- Instrument control: Setting flow rates, gradient programs, temperatures, and detector parameters.
- Data acquisition: Recording the detector signal in real-time.
- Chromatogram analysis: Identifying peaks, integrating peak areas, and calculating concentrations.
- Method development and validation: Storing and managing analytical methods.
- Reporting: Generating comprehensive reports of the analytical results.
Principles of Separation in HPLC
The effectiveness of HPLC hinges on the judicious selection of the mobile phase, stationary phase, and operating conditions to achieve optimal separation based on specific physicochemical properties of the analytes.
Modes of HPLC Separation
HPLC employs various separation modes, each suited for different types of compounds and applications:
- Reversed-Phase HPLC (RP-HPLC): The most common mode. It uses a non-polar stationary phase (e.g., C18-bonded silica) and a polar mobile phase (e.g., water/acetonitrile mixture). Separation is based on the hydrophobicity of the analytes, with more hydrophobic compounds retained longer. This mode is widely used for the analysis of a broad range of organic molecules, including pharmaceuticals, peptides, and environmental pollutants.
- Normal-Phase HPLC (NP-HPLC): Employs a polar stationary phase (e.g., silica or cyano-bonded silica) and a non-polar mobile phase (e.g., hexane/ethyl acetate mixture). Separation is based on the polarity of the analytes, with more polar compounds retained longer. This mode is effective for separating isomers and compounds with significant differences in polarity.
- Ion-Exchange Chromatography (IEC): Utilizes a stationary phase with charged functional groups that interact with oppositely charged analytes in the mobile phase. Separation is based on the charge of the analytes. Cation exchange chromatography retains positively charged molecules, while anion exchange chromatography retains negatively charged molecules. This mode is essential for analyzing proteins, nucleic acids, and small ions.
- Size Exclusion Chromatography (SEC): Also known as Gel Permeation Chromatography (GPC) for polymers or Gel Filtration Chromatography (GFC) for biomolecules. This technique separates molecules based on their hydrodynamic volume or size. The stationary phase consists of porous particles with precisely controlled pore sizes. Larger molecules that cannot enter the pores elute first, while smaller molecules that can penetrate the pores are retained longer. SEC is used for determining molecular weight distributions of polymers and for separating proteins and other macromolecules.
- Hydrophilic Interaction Liquid Chromatography (HILIC): A complementary technique to RP-HPLC, HILIC uses a polar stationary phase and a mobile phase with a high organic solvent content and a small amount of aqueous buffer. It is particularly useful for the separation of highly polar or hydrophilic compounds that are poorly retained in RP-HPLC.
Factors Affecting Separation
Several critical factors influence the quality of separation achieved in HPLC:
- Mobile Phase Composition: The ratio of organic solvent to aqueous buffer in the mobile phase dictates the eluting strength and selectivity. Adjusting this ratio can significantly alter retention times and improve resolution.
- Flow Rate: The speed at which the mobile phase is pumped through the column. Higher flow rates generally lead to faster analyses but can reduce resolution. Optimizing flow rate is crucial for balancing speed and separation efficiency.
- Column Temperature: Temperature affects the viscosity of the mobile phase and the kinetics of the interactions between analytes and the stationary phase. Controlled temperature is vital for reproducible results, especially with RI detectors.
- pH of the Mobile Phase: For ionizable compounds, the pH of the mobile phase is critical as it influences the ionization state of both the analyte and potentially the stationary phase.
- Stationary Phase Properties: The chemical nature, particle size, and pore size of the stationary phase are fundamental determinants of selectivity and efficiency.
Applications of HPLC
The versatility and sensitivity of HPLC have made it an indispensable tool in numerous fields:
- Pharmaceutical Industry: Quality control of raw materials and finished drug products, impurity profiling, stability studies, drug discovery and development, and pharmacokinetic studies. HPLC is crucial for ensuring the purity, potency, and safety of medications.
- Environmental Analysis: Monitoring water and air quality, detecting and quantifying pollutants such as pesticides, herbicides, polycyclic aromatic hydrocarbons (PAHs), and heavy metals.
- Food and Beverage Industry: Analyzing nutritional content (vitamins, amino acids, sugars), detecting food additives, contaminants (mycotoxins, pesticides), and authenticating food products.
- Biotechnology and Life Sciences: Analyzing proteins, peptides, nucleic acids, carbohydrates, and metabolites. HPLC is vital in proteomic and metabolomic research, drug discovery, and diagnostic assays.
- Forensic Science: Analyzing biological samples (blood, urine) for drugs of abuse, toxins, and other controlled substances.
- Clinical Diagnostics: Quantifying biomarkers for disease diagnosis and monitoring treatment efficacy.
- Chemical Industry: Quality control of chemicals, process monitoring, and research and development of new materials.
In essence, HPLC chromatography is a cornerstone of modern analytical chemistry, providing robust, sensitive, and reliable methods for unraveling the composition of complex liquid mixtures and ensuring the quality and safety of products across a vast spectrum of industries. Its continued evolution, particularly with the advent of UHPLC and advanced detection techniques, promises even greater analytical power and efficiency in the years to come.
