The term “generic medicine” often sparks curiosity and sometimes even skepticism. While the pharmaceutical industry is complex, understanding generic medications is crucial for informed healthcare decisions. At its core, generic medicine refers to a pharmaceutical product that is bioequivalent to a brand-name drug in dosage form, safety, strength, route of administration, quality, performance characteristics, and intended use. This means that a generic medication works in the same way and offers the same therapeutic benefits as its brand-name counterpart. The key distinction lies not in efficacy or safety, but in the proprietary name and, consequently, the price.
The Science Behind Bioequivalence
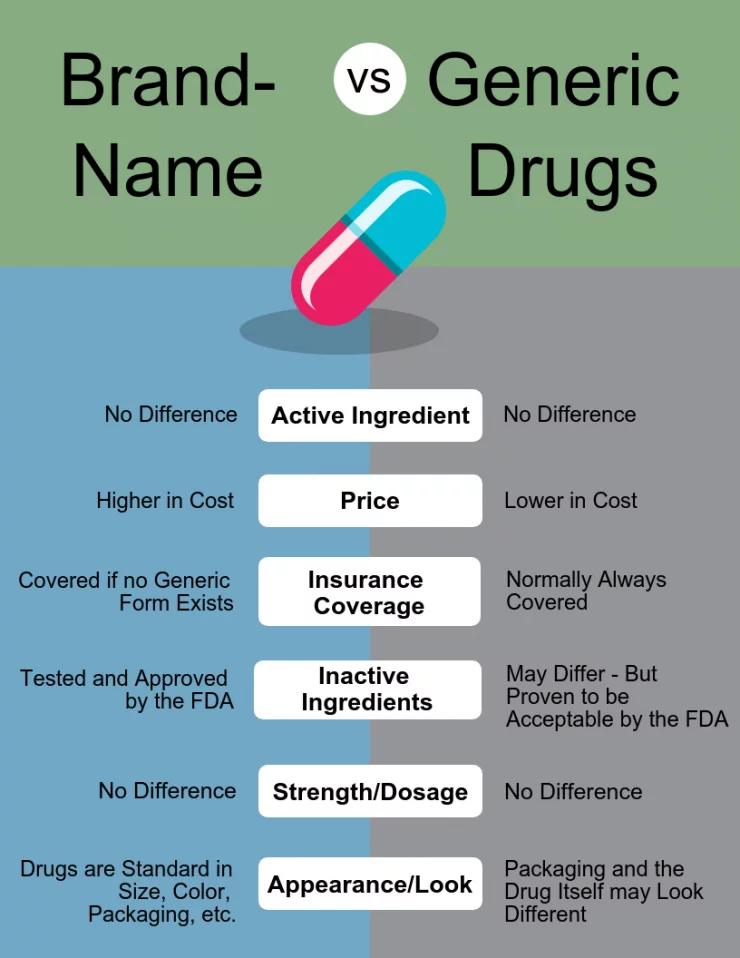
The concept of bioequivalence is the cornerstone of generic drug approval. Regulatory bodies, such as the U.S. Food and Drug Administration (FDA), rigorously assess generic medications to ensure they meet stringent standards before they can be marketed. This process involves demonstrating that the active ingredient in the generic drug is absorbed into the bloodstream at the same rate and to the same extent as the active ingredient in the brand-name drug. This is typically achieved through bioequivalence studies, which compare the pharmacokinetic profiles of the generic and brand-name products in human volunteers.
Active Pharmaceutical Ingredients (APIs)
The active pharmaceutical ingredient (API) is the component in a drug that produces the intended therapeutic effect. In the case of generic medicines, the API must be chemically identical to the API found in the brand-name drug. This means the molecular structure, purity, and characteristics of the API are the same. Generic manufacturers cannot simply substitute a different chemical compound and still claim bioequivalence. The rigorous testing and approval process ensures that the API functions identically within the body.
Dosage Forms and Strength
A generic drug must be available in the same dosage form (e.g., tablet, capsule, liquid, injection) and have the same strength (e.g., milligrams, micrograms) as the brand-name drug. For instance, if a brand-name medication is a 500mg tablet, its generic equivalent must also be a 500mg tablet. Deviations in dosage form or strength would alter the drug’s absorption and effectiveness, thus failing the bioequivalence requirement.
Quality and Manufacturing Standards
Generic drug manufacturers must adhere to the same Current Good Manufacturing Practices (CGMP) as brand-name pharmaceutical companies. These are a set of regulations that ensure the quality, purity, and safety of drugs. CGMP regulations cover all aspects of manufacturing, including facility design, equipment, personnel, raw material sourcing, production processes, and quality control testing. Regulatory agencies conduct regular inspections of manufacturing facilities to ensure compliance with these vital standards.
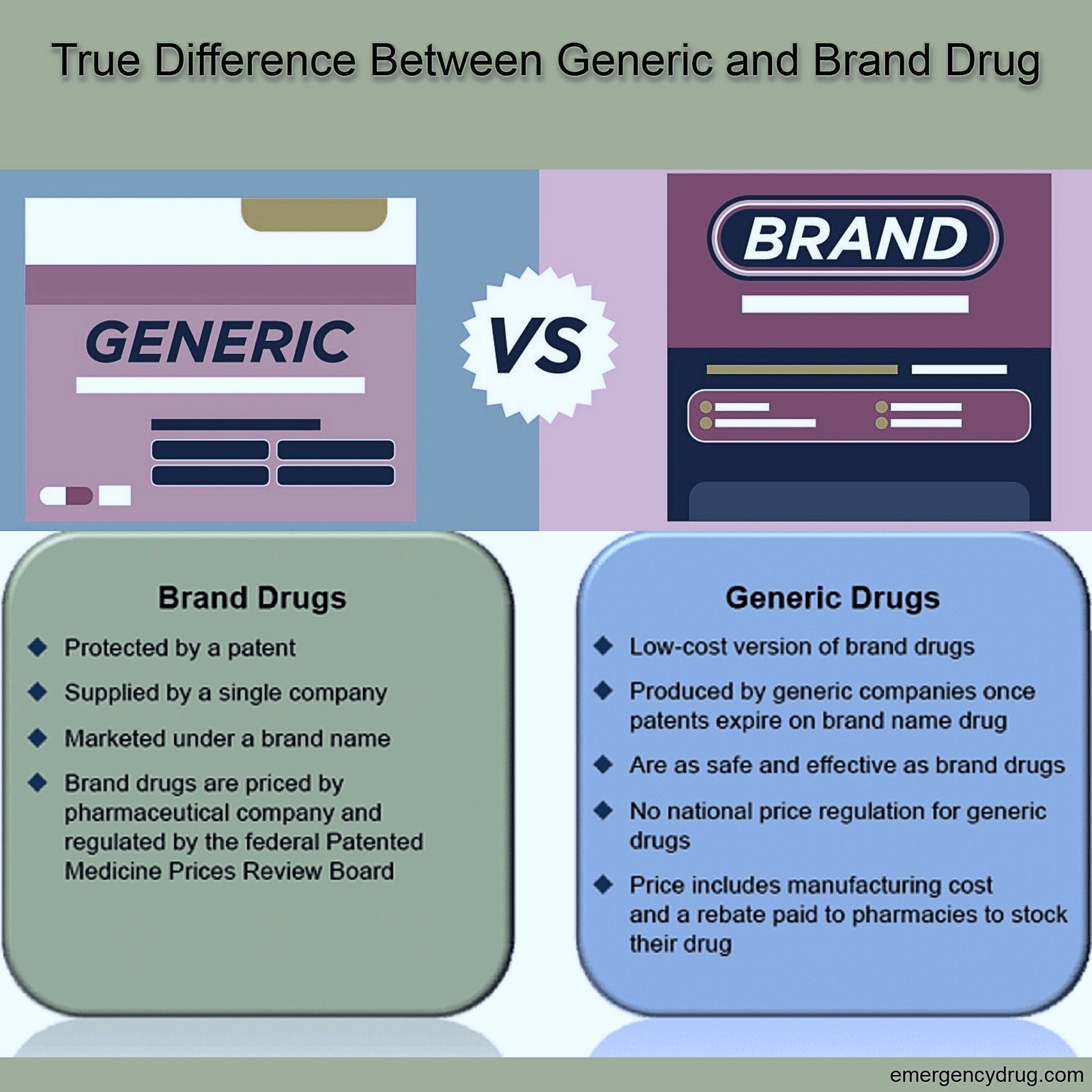
The Role of Patents and Exclusivity
The pharmaceutical industry relies heavily on patent protection to incentivize innovation and research and development. When a pharmaceutical company develops a new drug, it is granted a patent, which provides exclusive rights to market that drug for a specified period. During this patent-protected period, only the brand-name manufacturer can sell the drug.
Patent Expiration and Generic Entry
Once the patent on a brand-name drug expires, other pharmaceutical companies are permitted to develop and market generic versions of that medication. This process is often facilitated by specific legal provisions, such as the Hatch-Waxman Act in the United States, which provides a framework for the approval of generic drugs and aims to balance the incentives for innovation with the public’s need for affordable medications.
Market Competition and Price Reduction
The entry of generic competitors into the market following patent expiration leads to significant price reductions. This increased competition drives down the cost of medications, making them more accessible to a wider population. Brand-name drug manufacturers often price their products higher to recoup the substantial investments made in research, development, clinical trials, and marketing. Generic manufacturers, on the other hand, do not incur these initial research and development costs, allowing them to offer their products at substantially lower prices.
Benefits of Generic Medicines
The widespread availability and affordability of generic medicines offer numerous advantages to patients, healthcare systems, and the economy.
Affordability and Accessibility
The most significant benefit of generic medicines is their affordability. By offering chemically identical alternatives at a fraction of the brand-name price, generics make essential medications accessible to more people. This is particularly important for individuals with chronic conditions who require long-term medication or for those who face financial constraints in managing their healthcare costs. Increased access to treatment can lead to better health outcomes and reduced healthcare disparities.
Cost Savings for Healthcare Systems
Generic medications play a vital role in controlling healthcare expenditures. Governments and insurance providers can significantly reduce their drug spending by promoting the use of generics. This cost savings can then be reinvested in other areas of healthcare, such as research, public health initiatives, or expanding access to care for underserved populations. The widespread adoption of generics has been a key factor in managing rising healthcare costs globally.
Patient Choice and Empowerment
The availability of generic options empowers patients to make more informed choices about their healthcare. They can discuss treatment options with their healthcare providers, considering both efficacy and cost. This increased agency can lead to greater adherence to treatment regimens, as patients are more likely to continue taking medications that are financially sustainable for them.
Addressing Common Misconceptions
Despite the scientific and regulatory assurances, some misconceptions about generic medicines persist. It is important to address these to foster greater trust and understanding.
“Generic means lower quality.”
This is a common, yet unfounded, misconception. As previously discussed, generic medicines undergo rigorous testing and must meet the same quality, safety, and efficacy standards as brand-name drugs. The active ingredient is identical, and the manufacturing processes are subject to the same strict regulations. The lower price is a result of reduced research and development costs and market competition, not compromised quality.
“Generics are just as expensive as brand-names.”
This is generally untrue. While some specialized or newly off-patent generics might initially be priced higher, the competitive nature of the generic drug market typically leads to significant price reductions over time. The vast majority of generic medications are considerably less expensive than their brand-name counterparts, often by 80-85% or more.
“Generics contain different active ingredients.”
This is incorrect. For a drug to be considered a generic equivalent, it must contain the exact same active pharmaceutical ingredient (API) as the brand-name drug. While the inactive ingredients (fillers, binders, colors, flavorings) may differ, these do not affect the drug’s therapeutic action, safety, or efficacy. The inactive ingredients are also regulated and must be safe.
“My doctor won’t prescribe generics.”
Most healthcare professionals are well-versed in the science and benefits of generic medications. In many countries, prescribing guidelines and insurance formularies actively encourage or mandate the use of generics whenever a suitable option is available. Patients should feel comfortable discussing generic options with their doctors.
The Future of Generic Medicines
The landscape of generic medicine is continually evolving. As new brand-name drugs come off patent, opportunities arise for the development of new generic alternatives. Furthermore, advancements in pharmaceutical technology may lead to the development of more complex generic formulations, such as biosimilars for biologic drugs, which offer similar therapeutic benefits to their reference products at a lower cost.
Biosimilars: The Next Frontier
Biosimilars are a class of generic drugs derived from biological sources, such as living cells. Unlike small-molecule generics (which are chemically synthesized), biologics are complex molecules produced in living systems. Developing biosimilars is a more intricate process, and they are not considered exact copies but rather “highly similar” to their reference biologic product. Regulatory pathways for biosimilars are designed to demonstrate analytical, clinical, and pharmacokinetic similarity, ensuring comparable safety and efficacy. The increasing availability of biosimilars holds significant promise for reducing the cost of biologic therapies, which are often expensive and used to treat conditions like cancer, autoimmune diseases, and diabetes.
Regulatory Evolution and Global Harmonization
Regulatory agencies worldwide are continuously refining their approval processes for generic and biosimilar drugs to ensure patient safety while facilitating timely market entry. There is also a growing trend towards global harmonization of regulatory standards, which can streamline the approval process for manufacturers seeking to market their products in multiple countries. This can lead to wider availability of affordable medicines on a global scale.
In conclusion, generic medicine represents a critical component of a sustainable and equitable healthcare system. By providing safe, effective, and affordable alternatives to brand-name drugs, generics enhance access to essential treatments, reduce healthcare costs, and empower patients. Understanding the science behind bioequivalence, the role of patents, and the benefits of generics is essential for navigating the pharmaceutical landscape and making informed healthcare decisions.
