Gamma decay is a fascinating and fundamental process at the heart of nuclear physics, representing one of the primary ways atomic nuclei release excess energy. Unlike alpha and beta decay, which involve the emission of particles and a change in the atomic number or mass, gamma decay is a purely energetic transition. It’s the nuclear equivalent of an electron dropping to a lower energy level in an atom, but on a much grander scale of energy. Understanding gamma decay is not just an academic exercise; it underpins a vast array of technological advancements, from medical imaging to industrial quality control and the detection of radioactive materials.
At its core, gamma decay occurs when an atomic nucleus is in an excited, high-energy state and transitions to a more stable, lower-energy state by emitting a gamma ray photon. These gamma rays are high-energy electromagnetic radiation, identical in nature to X-rays and visible light but occupying the highest energy end of the electromagnetic spectrum. Their unique properties—high energy, no electric charge, and deep penetrative power—make them both valuable in technology and a significant consideration in radiation safety. Delving into the specifics of gamma decay reveals a complex interplay of forces within the nucleus and provides a window into the innovative applications of this powerful phenomenon.

The Fundamental Nature of Gamma Decay
To truly grasp gamma decay, one must first understand the concept of nuclear stability and the various states an atomic nucleus can occupy. Atoms, particularly their nuclei, are not always static entities; they can be profoundly dynamic, undergoing transformations to achieve greater stability.
Unpacking Radioactivity and Nuclear Stability
Radioactivity is the spontaneous process by which unstable atomic nuclei lose energy by emitting radiation. This instability can arise from an imbalance in the number of protons and neutrons, or simply from an excess of energy within the nucleus itself. While alpha decay (emission of a helium nucleus) and beta decay (emission of an electron or positron) alter the composition of the nucleus, changing one element into another, gamma decay primarily deals with energy reallocation within the same nucleus.
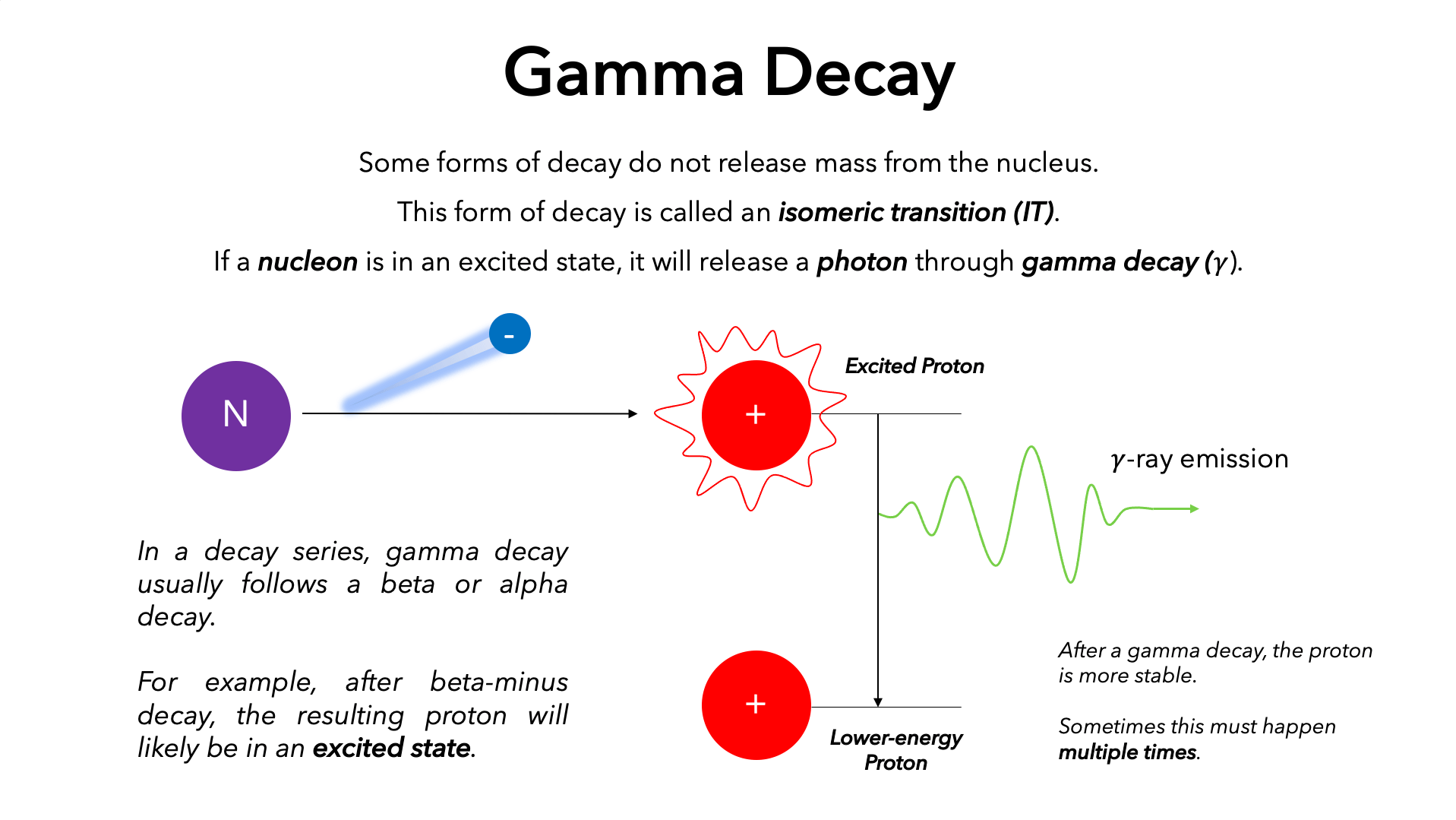
When a nucleus undergoes alpha or beta decay, it often leaves the daughter nucleus in an excited energy state. Imagine a ball rolling down a hill and momentarily stopping on a ledge before reaching the bottom. The initial decay (alpha or beta) is like the ball rolling to the ledge, and gamma decay is the subsequent drop from that ledge to the bottom of the hill. The nucleus, having shed particles, still holds onto residual excitation energy, analogous to electrons occupying higher orbital shells in an atom. To achieve its lowest possible energy state, known as the ground state, the nucleus sheds this excess energy in the form of a gamma ray photon. This process doesn’t change the number of protons or neutrons, nor does it transform the element. It’s a purification of energy, leading to a more stable, less energetic configuration of the same nucleus. This distinction is crucial for understanding how gamma rays are produced and their role in various technological processes.
The Energetic Emission of Gamma Rays
Gamma rays are photons, massless packets of electromagnetic energy, with wavelengths typically less than 10 picometers (10⁻¹¹ meters) and frequencies exceeding 10¹⁹ hertz. This places them far beyond the visible light spectrum, indicating their immense energy. The energy of a gamma ray emitted during decay is discrete and characteristic of the specific nuclear transition, much like the spectral lines of light emitted by excited atoms. This characteristic energy fingerprint is incredibly valuable for identifying specific radionuclides, a principle widely used in nuclear forensics, environmental monitoring, and medical diagnostics.
The emission of a gamma ray occurs almost instantaneously, typically within 10⁻¹⁰ to 10⁻¹⁴ seconds, after the initial alpha or beta decay that leaves the nucleus in an excited state. However, in some special cases, nuclei can remain in a metastable excited state for a longer period, sometimes seconds, minutes, or even years. These are known as nuclear isomers, and their decay is also through gamma emission, often referred to as isomeric transition. The energy carried by a single gamma ray photon can range from a few kiloelectronvolts (keV) to several megaelectronvolts (MeV), orders of magnitude greater than the energy of visible light photons. This high energy is what grants gamma rays their extraordinary penetrative power, allowing them to pass through significant thicknesses of matter, a property that is both beneficial for certain applications and a challenge for radiation shielding.
Distinguishing Gamma Radiation from Other Forms
While all forms of radioactive decay are fundamentally about achieving nuclear stability, the nature of the emitted radiation and its interaction with matter vary significantly, making each type unique in its properties and applications.
Alpha and Beta Decay: A Comparative View
Alpha decay involves the emission of an alpha particle, which consists of two protons and two neutrons (a helium nucleus). This process reduces the atomic number by two and the mass number by four, effectively transforming the parent nucleus into a different, lighter element. Alpha particles are relatively heavy and doubly positively charged, making them highly ionizing but with very low penetrative power; a sheet of paper or the outer layer of skin can typically stop them.
Beta decay, on the other hand, involves the transformation of a neutron into a proton (or vice versa) within the nucleus, accompanied by the emission of an electron (beta-minus decay) or a positron (beta-plus decay) and an antineutrino or neutrino. Beta decay changes the atomic number but keeps the mass number constant, also transforming the element. Beta particles are much lighter and singly charged, giving them greater penetrative power than alpha particles, typically stopped by a few millimeters of aluminum.
Gamma decay stands apart because it involves no change in the nuclear composition (atomic number or mass number). It is purely an energy release. Consequently, a gamma-emitting radionuclide does not change its identity. This fundamental difference means that while alpha and beta sources present primarily internal radiation hazards if ingested or inhaled, gamma sources pose both internal and external hazards due to their deep penetration. This necessitates different approaches to shielding and detection, which are critical considerations in technological designs involving radioactive materials.

Characteristics of Gamma Rays: Penetration and Interaction
The defining characteristic of gamma rays for technological applications is their high energy and resulting penetrative power. Being uncharged, gamma rays do not interact directly with the electrons or nuclei of matter through Coulombic forces, unlike charged alpha and beta particles. Instead, they interact predominantly through three main processes:
- Photoelectric Effect: The gamma ray transfers all its energy to an atomic electron, ejecting it from the atom. This effect is more pronounced with lower energy gamma rays and in materials with high atomic numbers.
- Compton Scattering: The gamma ray interacts with an atomic electron, transferring only part of its energy and deflecting at an angle. This scattered gamma ray has lower energy and can undergo further interactions. This is the dominant interaction for medium-energy gamma rays.
- Pair Production: For very high-energy gamma rays (above 1.022 MeV), the photon can interact with the electric field of a nucleus and convert its energy into an electron-positron pair.
These interaction mechanisms explain why gamma rays can pass through significant thicknesses of material before being absorbed or scattered. Heavy, dense materials like lead or depleted uranium are effective at attenuating gamma radiation because they increase the probability of these interactions. This understanding is paramount in designing shielding for nuclear reactors, medical radiation facilities, and protective equipment for personnel handling radioactive sources, embodying a core principle of radiation technology and safety innovation.
Technological Applications and Innovations of Gamma Rays
The unique properties of gamma rays, particularly their penetrative power and characteristic energy signatures, have led to their widespread adoption and driven innovation across numerous technological sectors.
Medical Imaging and Therapy: Diagnostics and Treatment
In medicine, gamma rays are indispensable tools for both diagnosis and therapy.
- Medical Imaging (SPECT and PET): Single-Photon Emission Computed Tomography (SPECT) and Positron Emission Tomography (PET) are revolutionary diagnostic techniques. In SPECT, a gamma-emitting radionuclide (like Technetium-99m, a pure gamma emitter created through isomeric transition) is introduced into the patient’s body. As it decays, it emits gamma rays, which are detected by external gamma cameras. By mapping the distribution of the radionuclide, doctors can visualize organ function, blood flow, and metabolic activity, identifying tumors, heart disease, and neurological disorders. PET scans, while involving positron emission, also rely on gamma ray detection when the emitted positron annihilates with an electron, producing two gamma rays that are detected to reconstruct detailed metabolic maps. These technologies represent a pinnacle of medical imaging innovation, allowing non-invasive insights into complex biological processes.
- Radiotherapy: High-energy gamma rays from sources like Cobalt-60 or medical linear accelerators are used to treat cancer. These gamma rays precisely target and destroy cancerous cells while minimizing damage to surrounding healthy tissue. The ability to precisely focus and control high-energy gamma beams is a testament to sophisticated engineering and continuous innovation in oncology.
- Sterilization: Gamma radiation is also used to sterilize medical equipment, pharmaceuticals, and even food products. Its penetrative power ensures thorough sterilization without the need for heat or chemicals, preserving the integrity of sensitive materials.
Industrial and Environmental Monitoring: Safety and Security
Beyond medicine, gamma rays play a critical role in industrial processes and environmental stewardship.
- Industrial Radiography: Similar to X-rays, gamma rays are used to inspect the integrity of materials in manufacturing, such as welding seams, pipelines, and structural components. By passing gamma rays through an object and capturing the transmitted radiation on film or a digital detector, engineers can detect flaws, cracks, or voids without dismantling the structure. This non-destructive testing (NDT) is vital for ensuring the safety and reliability of critical infrastructure.
- Level and Density Gauges: Gamma sources are incorporated into gauges to measure the fill levels in tanks or the density of materials in pipelines. The attenuation of gamma rays is directly related to the amount of material they pass through, providing precise, real-time measurements crucial for process control in industries ranging from mining to food production.
- Environmental Radiation Monitoring and Security: Gamma-ray detectors are essential for monitoring background radiation levels, detecting nuclear waste, and identifying illicit nuclear materials. Portable gamma spectrometers can quickly identify specific radionuclides based on their unique gamma energy signatures, providing critical information for emergency response, environmental protection, and national security. Innovations in detector technology, including compact and sensitive instruments, enable efficient remote sensing and real-time data acquisition, pushing the boundaries of environmental and security monitoring.
Scientific Research and Material Science: Unlocking New Frontiers
In scientific research, gamma rays are invaluable probes for understanding the fundamental properties of matter and developing new materials.
- Nuclear Spectroscopy: Scientists use gamma-ray detectors to analyze the energy spectra of emitted gamma rays, providing detailed information about the energy levels and structure of atomic nuclei. This research expands our understanding of the strong nuclear force and the fundamental building blocks of the universe.
- Material Modification: Controlled gamma irradiation can alter the properties of materials at a molecular level, leading to the development of new polymers with enhanced strength, heat resistance, or barrier properties. This technique is used in fields from aerospace to electronics.
- Gamma-Ray Astronomy: High-energy gamma rays from space reveal some of the most energetic phenomena in the universe, such as supernovae, black holes, and active galactic nuclei. Ground-based and space-based gamma-ray telescopes are at the forefront of astrophysics, offering a unique perspective on cosmic events that are invisible in other wavelengths. These instruments represent a pinnacle of detector technology and data processing innovation.
The Physics Behind Gamma-Ray Emission
A deeper dive into the physics of gamma decay reveals the intricate quantum mechanics governing nuclear transitions. It’s not merely an arbitrary release of energy but a structured process dictated by nuclear quantum states.
Nuclear Energy Levels and Transitions
Just as electrons in an atom occupy discrete energy levels, protons and neutrons within a nucleus also reside in specific quantized energy states. When an initial radioactive decay (alpha or beta) occurs, the resultant daughter nucleus is often left in one of these higher, excited nuclear energy levels. The nucleus then seeks to transition to a lower, more stable energy state by emitting the excess energy as a gamma ray photon. The energy of the emitted gamma photon precisely matches the energy difference between the initial excited state and the final lower energy state.
These nuclear energy levels are characterized by specific quantum numbers, including spin and parity. The selection rules governing gamma transitions dictate which transitions are allowed and the probability of their occurrence. For example, a large change in angular momentum between the initial and final states can lead to a longer lifetime for the excited state, resulting in an isomeric transition. The detailed study of these transitions through gamma-ray spectroscopy provides invaluable insights into the internal structure and dynamics of the nucleus, pushing the boundaries of our understanding of fundamental forces and matter.
Isomeric Transitions and Metastable States
A particularly interesting aspect of gamma decay involves nuclear isomers. These are nuclei that exist in an excited energy state for a measurably long period (from nanoseconds to millennia) before decaying via gamma emission to a lower energy state. Such long-lived excited states are referred to as metastable states. The reason for their longevity is often due to large differences in angular momentum between the isomeric state and the ground state. A transition requiring a significant change in angular momentum is “hindered” or less probable, hence the longer half-life.
Technetium-99m (⁹⁹ᵐTc) is a prime example of a nuclear isomer and is arguably the most widely used medical radioisotope. The “m” denotes its metastable state. It decays by isomeric transition, emitting a 140 keV gamma ray with a half-life of 6 hours, transforming into stable Technetium-99. Its pure gamma emission, relatively low energy, and short half-life make it ideal for diagnostic medical imaging, as it minimizes patient radiation dose while providing clear images. The development of technetium-99m generators, which allow hospitals to produce this isotope on-site, is a testament to the innovative application of nuclear physics principles in practical medical technology.
Managing Gamma Radiation: Safety and Detection Technologies
Given the high penetrative power and potential biological impact of gamma rays, understanding their management and detection is paramount in any technological application.
Principles of Radiation Protection
Effective radiation protection relies on three fundamental principles:
- Time: Minimize the duration of exposure to a radiation source.
- Distance: Maximize the distance from the radiation source, as intensity decreases with the square of the distance.
- Shielding: Use appropriate materials to attenuate radiation. For gamma rays, dense materials like lead, concrete, or steel are employed. The thickness of shielding required depends on the energy of the gamma rays and the desired reduction in exposure.
Innovations in radiation safety technology focus on improving these principles, from automated systems that reduce human presence near sources to advanced composite shielding materials and robust regulatory frameworks. Personal dosimeters, which measure an individual’s accumulated radiation dose, are also critical tools for ensuring occupational safety in environments where gamma radiation is present.
Advancements in Gamma-Ray Detection
The ability to accurately detect and characterize gamma rays is foundational to almost all their technological applications. Modern gamma-ray detectors leverage various principles:
- Scintillation Detectors: These detectors use materials (scintillators) that emit flashes of light when struck by gamma rays. The light is then converted into an electrical signal by a photomultiplier tube. Sodium iodide (NaI) crystals are common, though more advanced materials like Lanthanum Bromide (LaBr₃) offer higher energy resolution and faster response times, crucial for demanding applications in security and research.
- Semiconductor Detectors: High-purity germanium (HPGe) detectors provide exceptional energy resolution, allowing for precise identification of specific radionuclides even in complex mixtures. These detectors require cryogenic cooling but are indispensable for scientific research, environmental monitoring, and nuclear forensics where fine spectral detail is required.
- Compact and Portable Detectors: Recent innovations focus on miniaturization and ruggedization of gamma-ray detectors, enabling their deployment in challenging environments, for remote sensing, and in handheld devices for field use. This includes advancements in solid-state detectors that operate at room temperature, simplifying their logistics and expanding their utility in areas such as emergency response and customs inspection.
The ongoing development of more sensitive, selective, and robust gamma-ray detection technologies continues to expand the horizons of scientific discovery, enhance industrial safety, improve medical diagnostics, and bolster global security efforts, truly embodying the spirit of Tech & Innovation. Understanding gamma decay is not just about comprehending a nuclear phenomenon; it’s about harnessing a powerful force of nature for the betterment of society.
