The remarkable capabilities of modern drones—their agility, extended flight times, and sophisticated payloads—are fundamentally dependent on efficient and reliable power sources. At the heart of these power sources, specifically the rechargeable batteries that fuel every drone mission, lies a core electrochemical principle: the galvanic reaction. Understanding this fundamental process is not merely an academic exercise; it provides crucial insights into battery performance, longevity, and the ongoing innovations driving the drone accessory market.
The Electrochemical Foundation of Drone Power
A galvanic reaction, also known as a voltaic cell, is an electrochemical process that generates electrical energy from a spontaneous chemical reaction. Unlike electrolytic cells, which require an external power source to drive a non-spontaneous reaction, galvanic cells create electricity naturally when specific materials are brought into contact, initiating a redox (reduction-oxidation) reaction. This principle is the bedrock upon which all chemical batteries, including the advanced Lithium-ion (Li-ion) and Lithium Polymer (LiPo) batteries powering contemporary drones, are built.
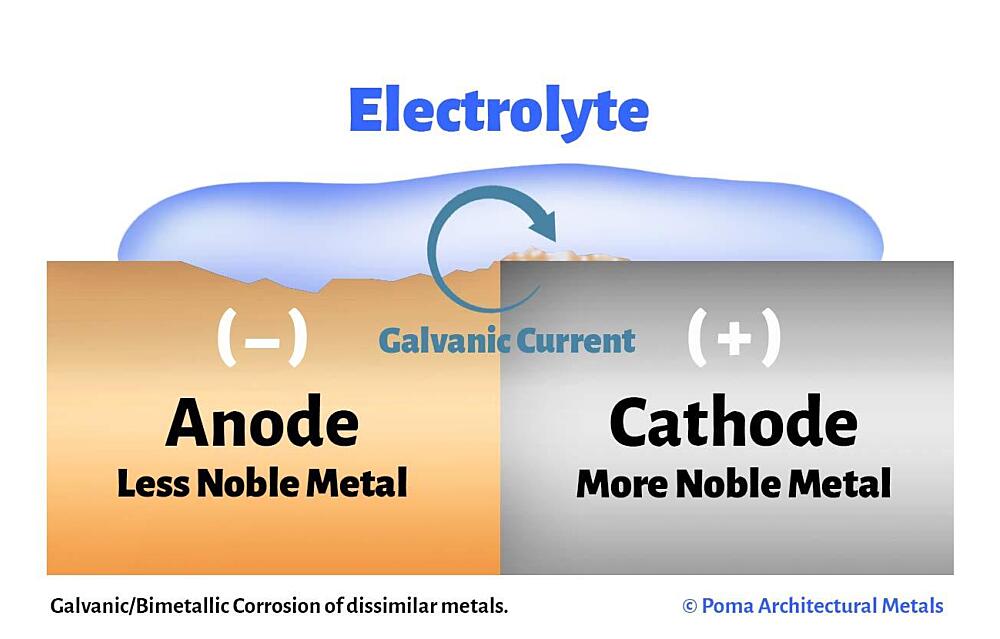
Defining Galvanic Reactions
At its core, a galvanic reaction involves the transfer of electrons between two different chemical species, one of which is oxidized (loses electrons) and the other reduced (gains electrons). This electron transfer, when harnessed in a controlled manner, constitutes an electrical current. The driving force behind this transfer is the difference in electrochemical potential between the two materials. When a circuit is completed, electrons flow from the material with higher electron potential (the anode) to the material with lower electron potential (the cathode), creating the electrical energy that powers a drone’s motors, flight controller, and onboard sensors.
Anode, Cathode, and Electrolyte in Action
Every galvanic cell, whether it’s a simple lemon battery or a high-performance drone battery, consists of three essential components:
- Anode (Negative Electrode): This is where oxidation occurs. The material at the anode readily gives up electrons, becoming positively charged ions that dissolve into the electrolyte or react with it. In a Li-ion battery, the anode is typically made of graphite, which intercalates (stores) lithium ions.
- Cathode (Positive Electrode): This is where reduction occurs. The material at the cathode accepts electrons from the external circuit. In a Li-ion battery, the cathode is usually a lithium metal oxide (e.g., Lithium Cobalt Oxide, Lithium Manganese Oxide, Lithium Nickel Manganese Cobalt Oxide).
- Electrolyte: This is an ionic conductor that facilitates the movement of ions between the anode and cathode, completing the internal circuit. It prevents direct electron flow between the electrodes, forcing electrons to travel through the external circuit to perform work. In drone batteries, the electrolyte is typically a non-aqueous solvent containing lithium salts.
During discharge, lithium ions move from the graphite anode through the electrolyte to the metal oxide cathode, while electrons travel from the anode through the external drone circuit (powering it) to the cathode. During charging, the process is reversed: an external power source forces electrons and lithium ions back to the anode, storing energy for the next flight.
The Flow of Electrons and Energy Generation
The consistent flow of electrons from the anode, through the drone’s circuitry, and back to the cathode is what generates the electrical current. The voltage of the battery is determined by the difference in electrochemical potential between the anode and cathode materials. The capacity of the battery (how much energy it can store) is directly related to the amount of reactive material available in the electrodes. For drone operators, understanding this internal dance of electrons and ions highlights why battery chemistry and construction are paramount to achieving desired flight times and power output for demanding maneuvers or heavy payloads.
Galvanic Cells in Drone Batteries: Lithium-Ion and Beyond
The prevalence of Li-ion and LiPo batteries in drones is a testament to their superior energy density and power-to-weight ratio, directly stemming from optimized galvanic reactions. These characteristics are critical for maximizing flight endurance and enabling high-performance operations in a lightweight package.
How Lithium-Ion Batteries Utilize Galvanic Principles
Li-ion batteries leverage the exceptional ability of lithium ions to intercalate into and deintercalate from host materials without significantly altering their crystal structure. This “rocking chair” mechanism allows for thousands of charge and discharge cycles. The specific materials chosen for the anode (graphite) and cathode (various lithium metal oxides) offer a high potential difference, leading to higher voltage per cell (typically 3.7V nominal). The high reactivity of lithium, coupled with its small atomic size, allows for a dense packing of charge carriers, contributing to the high energy density that makes extended drone flights possible.
Understanding Energy Density and Discharge Rates
Energy density, measured in Watt-hours per kilogram (Wh/kg) or Watt-hours per liter (Wh/L), quantifies how much energy a battery can store relative to its weight or volume. Higher energy density means more flight time for the same battery size. The selection of cathode materials, such as NMC (Nickel Manganese Cobalt) or NCA (Nickel Cobalt Aluminum), plays a significant role in achieving these high densities.
Discharge rate, often expressed as a ‘C-rating’ (e.g., 20C, 50C), indicates how quickly a battery can safely deliver its stored energy. A 20C battery can discharge 20 times its capacity in an hour. High discharge rates are crucial for drones, especially racing or heavy-lift models, which demand rapid bursts of power for acceleration, ascent, or carrying heavy cameras. These rates are governed by the kinetics of the galvanic reaction—how quickly ions can move through the electrolyte and react at the electrodes. Batteries designed for high C-ratings typically feature thinner electrodes and more porous separators to facilitate faster ion movement.
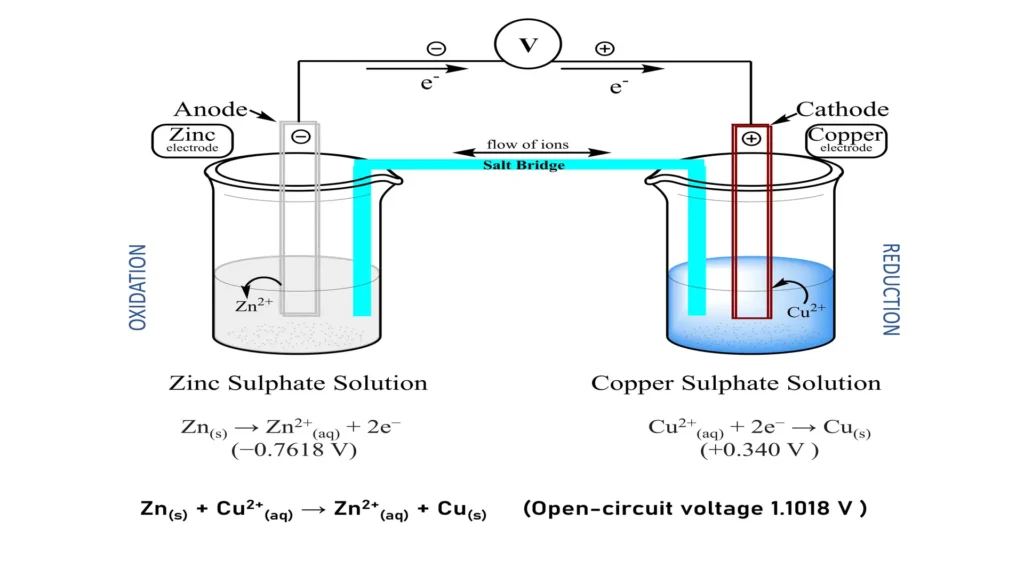
The Role of Materials in Performance
The specific chemical composition and structural engineering of the electrodes and electrolyte profoundly impact a drone battery’s performance. Innovations in materials science are constantly pushing the boundaries:
- Cathode Materials: Different lithium metal oxides offer trade-offs between energy density, power output, cycle life, and safety. For instance, LCO (Lithium Cobalt Oxide) offers high energy density but lower power and safety than NMC or NCA, which balance these attributes.
- Anode Materials: While graphite is standard, research into silicon-carbon composites and pure silicon anodes promises significantly higher energy densities due to silicon’s ability to store more lithium ions.
- Electrolyte: The electrolyte’s stability, ionic conductivity, and resistance to degradation at various temperatures directly affect battery efficiency, safety, and lifespan. Solid-state electrolytes are a major area of research, aiming to improve safety and energy density.
Optimizing Battery Life and Performance Through Electrochemical Insight
Understanding the galvanic reactions within drone batteries empowers operators to optimize their care and usage, significantly extending lifespan and maintaining peak performance. Mishandling or improper maintenance can accelerate undesirable side reactions that degrade the battery’s internal galvanic cell.
The Impact of Charging and Discharging Cycles
Each complete charge and discharge cycle subjects the battery’s internal galvanic system to stress. During discharge, active material in the electrodes is consumed or undergoes structural changes. During charging, lithium ions are forced back, which can lead to phenomena like lithium plating on the anode if charged too quickly or at low temperatures, forming dendrites that can puncture the separator and cause short circuits—a serious safety concern.
- Partial Cycling: For Li-ion/LiPo batteries, partial discharges followed by charges are often less stressful than deep discharges, which can accelerate capacity loss.
- Charging Rate: Adhering to manufacturer-recommended charging rates (e.g., 1C or 2C) is vital. Overcharging or charging too rapidly can degrade the electrode materials and electrolyte, reducing the number of available sites for the galvanic reaction and shortening lifespan.
Preventing Degradation: Rust, Corrosion, and Internal Resistance
While “rust” in the traditional sense (iron oxidation) isn’t the primary concern for drone batteries, analogous electrochemical degradation processes occur:
- Solid Electrolyte Interphase (SEI) Layer: This protective layer forms on the anode during the first few charge cycles. If it grows excessively or becomes unstable due to improper cycling, it consumes active lithium, leading to capacity fade and increased internal resistance.
- Electrode Degradation: Repeated expansion and contraction of electrode materials during cycling can cause mechanical stress, cracking, and loss of electrical contact, reducing the efficiency of the galvanic reactions.
- Corrosion (Analogous): The electrolyte can slowly react with electrode materials or current collectors over time, especially at high temperatures or improper voltage levels, forming resistive layers that impede ion and electron flow, thus increasing internal resistance and reducing power output.
Increased internal resistance means more energy is converted to heat rather than useful power, leading to shorter flight times and reduced peak performance.
Best Practices for Battery Storage and Maintenance
Proper storage and maintenance are direct applications of electrochemical principles:
- Storage Voltage: Storing LiPo batteries at their recommended “storage voltage” (typically around 3.8V per cell, or 50% charge) minimizes the rate of parasitic side reactions that degrade electrode materials, preserving the galvanic cell’s integrity.
- Temperature Control: Extreme temperatures accelerate all chemical reactions, including the undesirable ones that cause degradation. Storing and operating batteries within recommended temperature ranges is crucial. Charging in cold conditions can be particularly damaging.
- Balancing Cells: Ensuring all cells within a battery pack are at the same voltage prevents individual cells from being overcharged or over-discharged, which could stress their specific galvanic reactions disproportionately.
Future of Drone Batteries: Innovations Driven by Electrochemistry
The future of drone technology is intrinsically linked to advancements in battery technology, all rooted in deeper understanding and manipulation of galvanic reactions. The quest for longer flight times, faster charging, and enhanced safety is fueling extensive research in electrochemistry.
Solid-State Batteries and Enhanced Safety
Solid-state batteries replace the volatile liquid organic electrolyte with a solid material, offering significant safety advantages by eliminating the risk of leakage and fire. From an electrochemical perspective, designing stable, highly conductive solid electrolytes that maintain efficient ion transport at various temperatures is the primary challenge. When perfected, these could revolutionize drone batteries by offering higher energy densities, faster charging capabilities, and improved cycle life, all within a much safer package.
Exploring New Chemistries for Greater Endurance
Researchers are continually exploring alternative chemistries beyond the current Li-ion paradigm. Lithium-sulfur (Li-S) batteries, for example, theoretically offer much higher energy densities than traditional Li-ion cells because sulfur is abundant and lightweight. However, the complex galvanic reactions in Li-S systems, including the formation of polysulfides and significant volume changes, currently limit their cycle life and practical application. Other chemistries, such as lithium-air or sodium-ion batteries, are also being investigated, each presenting unique electrochemical challenges and opportunities for future drone power.
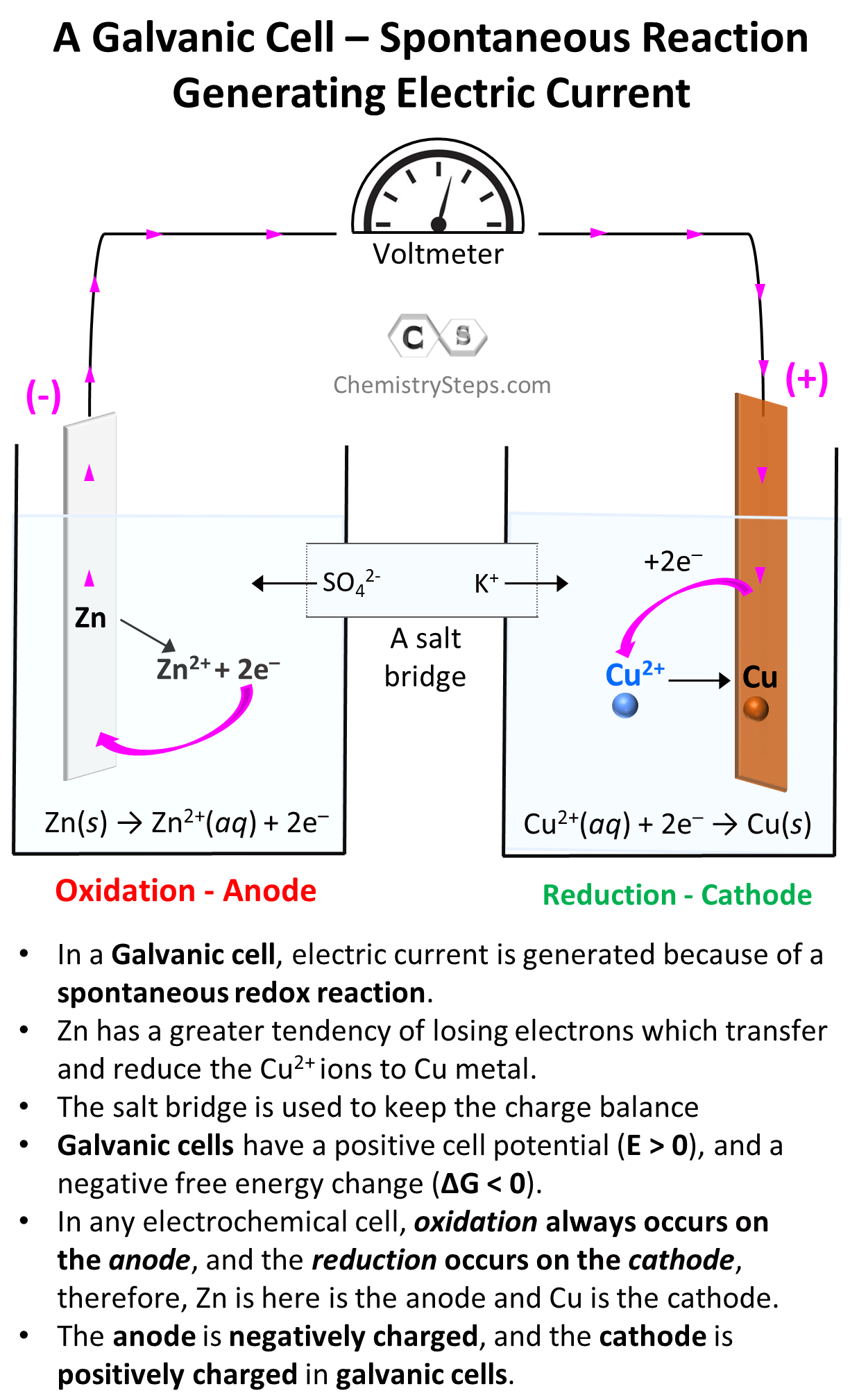
Fast Charging and Thermal Management
The ability to rapidly recharge drone batteries is critical for commercial applications, reducing downtime and increasing operational efficiency. Achieving fast charging without compromising battery lifespan or safety requires meticulous control over the galvanic reactions. This involves developing electrode materials that can rapidly accept and release lithium ions without degradation, and electrolytes that can quickly transport them.
Effective thermal management is also crucial, as rapid charging and discharging generate heat, which can accelerate degradation of the galvanic cell components and even pose safety risks. Integrated cooling systems, advanced battery management systems (BMS), and innovative cell designs are all being developed to manage the heat generated by these powerful electrochemical reactions, ensuring the drone’s power source remains stable and efficient throughout its operational life.
In conclusion, the simple yet profound principle of the galvanic reaction is the unseen engine propelling the entire drone industry forward. From the meticulous design of Li-ion cells to the cutting-edge research into solid-state chemistries, every improvement in drone battery technology is a direct consequence of refining and optimizing these electrochemical processes. For drone enthusiasts and professionals alike, a foundational understanding of galvanic reactions provides not only technical insight but also a deeper appreciation for the sophisticated power accessories that make aerial innovation possible.
