The Invisible Architecture of Oral Health
Dental biofilm, often referred to as plaque, is a complex, structured community of microorganisms, primarily bacteria, encased within a self-produced matrix of extracellular polymeric substances (EPS). This microbial consortium adheres tenaciously to the tooth surface, both above and below the gum line, as well as to dental restorations and prosthetic devices. Far from being a simple aggregation of germs, dental biofilm is a highly organized and dynamic ecosystem that plays a central role in the initiation and progression of common oral diseases such as dental caries (cavities) and periodontal disease (gum disease). Understanding the fundamental nature of dental biofilm is paramount for effective oral hygiene practices, preventative dental care, and the development of novel therapeutic strategies.
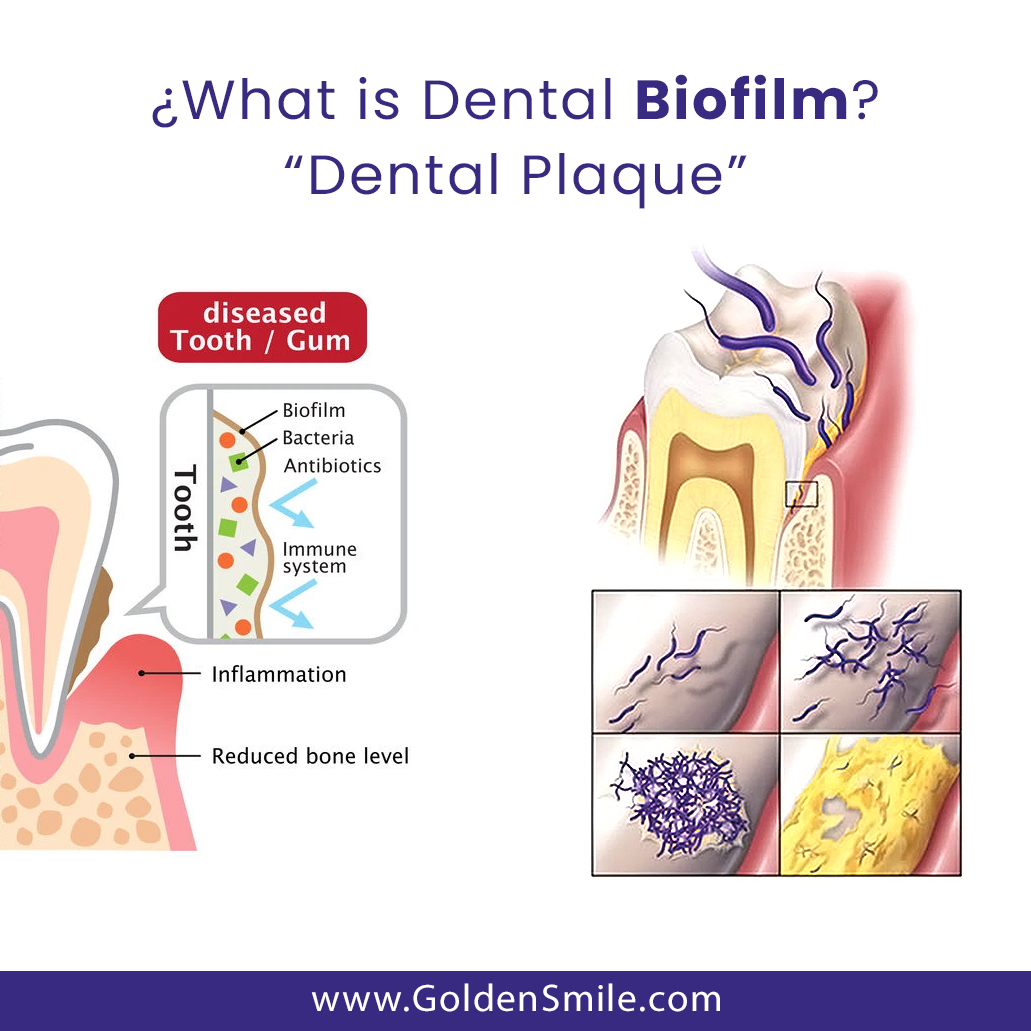
The term “biofilm” itself signifies a departure from the traditional view of bacteria as free-floating planktonic cells. In a biofilm, microorganisms transition into a sessile state, adopting distinct physiological and genetic characteristics that enhance their survival and resilience. This transition is triggered by specific environmental cues and involves intricate communication mechanisms, such as quorum sensing, which allow the bacteria to coordinate their behavior and collectively build their protective matrix. This matrix, predominantly composed of polysaccharides, proteins, lipids, and nucleic acids, acts as a scaffold, providing structural integrity, facilitating nutrient and waste exchange, and shielding the microbial community from external threats like antibiotics and the host immune system.
The formation of dental biofilm is a multistep process that begins with the conditioning of the tooth surface. Immediately after cleaning, the enamel surface becomes coated with salivary glycoproteins, forming a pellicle. This pellicle serves as an initial attachment site for free-swimming bacteria. Certain species, particularly Gram-positive cocci like Streptococcus mutans and Streptococcus sanguinis, are early colonizers and can adhere to the pellicle through specific adhesins. As the biofilm matures, a succession of microbial species invades and colonizes the developing community. Gram-negative bacteria, such as Porphyromonas gingivalis and Fusobacterium nucleatum, often appear in later stages, contributing to the complexity and pathogenicity of the biofilm. This microbial succession leads to a diverse and intricate consortium, where different species interact synergistically or antagonistically, influencing the overall metabolic activity and virulence of the biofilm.
The Composition and Structure of Dental Biofilm
The extracellular matrix, the defining characteristic of dental biofilm, is a crucial component that dictates its physical properties and protective capabilities. This matrix is not a static entity but is continually remodeled and modified by the resident microorganisms. It is primarily composed of polysaccharides, synthesized by bacteria, which provide a sticky and cohesive framework. These polysaccharides contribute to the adhesion of bacteria to each other and to the tooth surface, as well as to the overall structural integrity of the biofilm. In addition to polysaccharides, the matrix contains a significant amount of proteins, including enzymes, antibodies, and host-derived proteins. These proteins can play roles in nutrient acquisition, interspecies communication, and immune evasion. Lipids are also present, contributing to the hydrophobic nature of the matrix and influencing its permeability. Furthermore, extracellular DNA (eDNA) has emerged as a critical component of the biofilm matrix, contributing to its structural stability, facilitating genetic exchange between bacteria, and enhancing resistance to antimicrobial agents.
Within this matrix, the microorganisms are not randomly distributed but are organized into microcolonies. These microcolonies are embedded within channels and pores that permeate the EPS matrix. These channels are vital for the transport of nutrients, oxygen, and metabolic waste products, ensuring the viability of the microorganisms within the deeper layers of the biofilm. The architecture of these channels can vary depending on the stage of biofilm development and the specific microbial composition. This intricate internal structure allows for a complex interplay of metabolic activities among the different bacterial species, where the byproducts of one species can serve as nutrients for another, fostering a symbiotic relationship that enhances the collective survival and virulence.
The heterogeneity of the biofilm environment is also a significant factor. Microgradients of oxygen, pH, and nutrient availability exist within the biofilm, leading to differential gene expression and physiological states among the bacterial populations. Bacteria located in the outer layers, exposed to higher oxygen levels, may have different metabolic pathways than those in the anaerobic core of the biofilm. This spatial organization and metabolic diversity contribute to the remarkable adaptability and resilience of dental biofilms.
The Pathogenic Potential of Dental Biofilm
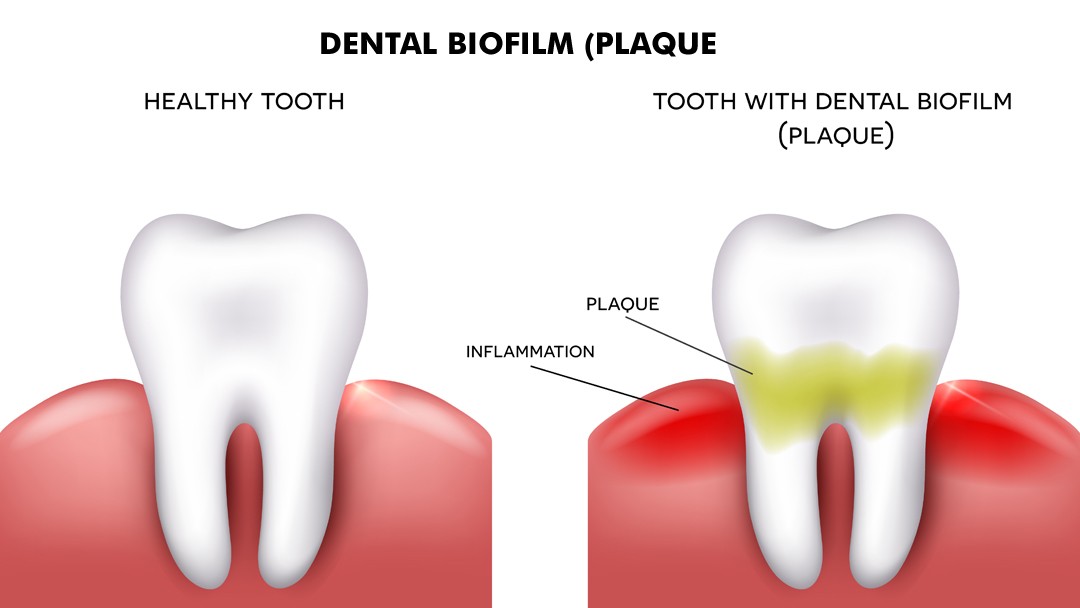
Dental biofilm is the primary etiological factor in the development of the most prevalent oral diseases: dental caries and periodontal disease. The accumulation of metabolically active bacteria within the biofilm leads to the production of acids, enzymes, and toxins that can damage host tissues.
Dental Caries
Dental caries, commonly known as tooth decay, is initiated when acidogenic bacteria within the biofilm metabolize dietary carbohydrates, such as sugars, to produce organic acids, primarily lactic acid. The EPS matrix of the biofilm acts as a diffusion barrier, concentrating these acids against the tooth surface. The enamel, which is highly mineralized and composed of hydroxyapatite, is susceptible to demineralization in an acidic environment. When the rate of demineralization exceeds the rate of remineralization, the tooth structure begins to break down, leading to the formation of a carious lesion. Early colonization by acid-tolerant bacteria like Streptococcus mutans is crucial in this process. As the lesion progresses, the biofilm can penetrate deeper into the dentin, recruiting other cariogenic bacteria and accelerating tissue destruction. The acidic environment also alters the composition of the biofilm, favoring the growth of more acid-tolerant species and perpetuating the cycle of demineralization.
Periodontal Disease
Periodontal disease, a spectrum of inflammatory conditions affecting the gums and supporting bone structures of the teeth, is also directly linked to the presence and pathogenicity of dental biofilm. Biofilm accumulation, particularly subgingivally (below the gum line), triggers an inflammatory response from the host’s immune system. Gram-negative anaerobic bacteria, such as Porphyromonas gingivalis, Tannerella forsythia, and Prevotella intermedia, are key pathogens in periodontitis. These bacteria produce a range of virulence factors, including lipopolysaccharides (LPS), proteases, and endotoxins, which directly damage host tissues and stimulate the release of pro-inflammatory cytokines. The host’s immune response, while intended to clear the infection, can inadvertently lead to the destruction of periodontal tissues, including the gums, periodontal ligament, and alveolar bone. The unique architecture of subgingival biofilms, often characterized by a mixed flora with a significant proportion of Gram-negative anaerobes, creates an environment conducive to chronic inflammation and tissue destruction. The EPS matrix provides protection against host defenses and antibiotics, making these biofilms particularly challenging to eradicate.

Disruption and Prevention of Dental Biofilm
Given its central role in oral disease, the effective disruption and prevention of dental biofilm are cornerstones of modern dental care. Mechanical disruption, through brushing and flossing, remains the most effective method for removing supragingival (above the gum line) biofilm. These actions physically dislodge the microbial colonies and their matrix from the tooth surfaces. However, the complex and tenacious nature of biofilm, particularly its ability to adhere to microscopic irregularities on the tooth surface and within interdental spaces, means that complete removal through mechanical means alone can be challenging.
Chemical agents, such as antimicrobial mouthwashes containing ingredients like chlorhexidine, essential oils, or cetylpyridinium chloride, can play a supplementary role in reducing bacterial load and inhibiting biofilm formation. However, these agents are generally considered adjuncts to mechanical cleaning and are not typically sufficient on their own for effective biofilm control.
Professional dental cleanings, performed by dental hygienists and dentists, are essential for removing tenacious biofilm and calculus (hardened plaque) that cannot be eliminated by daily home care. These procedures involve scaling and root planing, which mechanically remove deposits from tooth surfaces, both above and below the gum line.
Emerging strategies for biofilm control are exploring novel approaches to target the structural components of the biofilm matrix or to disrupt microbial communication. These include the use of enzymes to degrade the EPS, bacteriophages that specifically target pathogenic bacteria within the biofilm, and agents that interfere with quorum sensing mechanisms. Research into understanding the molecular mechanisms of biofilm formation and the complex interspecies interactions within these communities continues to pave the way for more targeted and effective preventative and therapeutic interventions. Ultimately, a comprehensive approach that combines diligent daily oral hygiene with regular professional dental care, underpinned by a thorough understanding of dental biofilm, is crucial for maintaining lifelong oral health.
