DDT, an acronym for dichlorodiphenyltrichloroethane, is a synthetic organochlorine insecticide that gained widespread use in the mid-20th century. Its remarkable efficacy in controlling insect populations, particularly disease vectors like mosquitoes, led to its initial embrace as a powerful tool for public health and agricultural pest management. However, the very properties that made DDT so effective also contributed to its eventual downfall and eventual ban in many countries due to its persistence in the environment and harmful effects on wildlife and human health. Understanding the chemistry of DDT is crucial to appreciating its historical significance, its environmental impact, and the lessons learned from its application.

The Chemical Structure and Properties of DDT
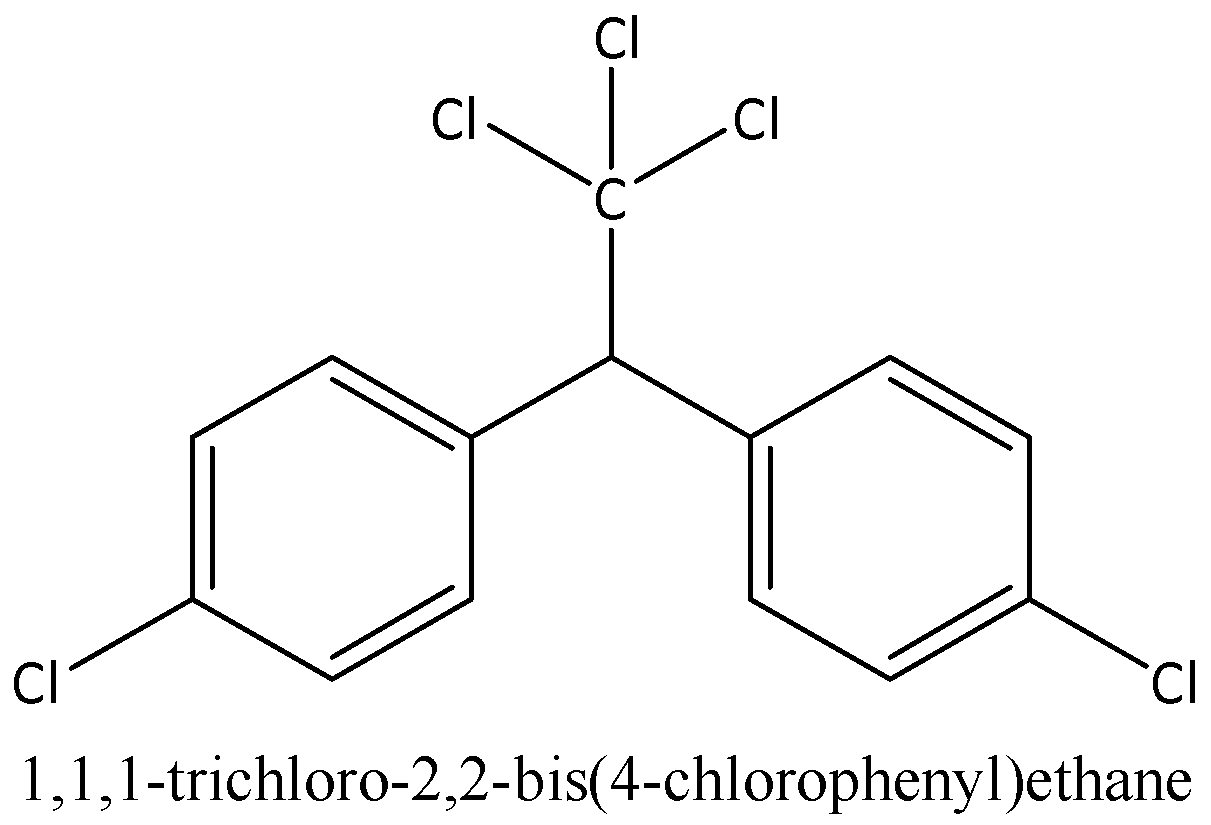
DDT is a complex organic molecule with the chemical formula C₁₄H₉Cl₅. Its structure features two phenyl rings, each substituted with chlorine atoms, and a central ethane backbone. Specifically, it consists of a central carbon atom bonded to a trichloromethyl group (-CCl₃) and a carbon atom that is bonded to two p-chlorophenyl groups. This specific arrangement of atoms confers upon DDT its characteristic lipophilicity (fat-solubility) and its recalcitrance to degradation.
Synthesis of DDT
The synthesis of DDT is a relatively straightforward electrophilic aromatic substitution reaction. It is typically produced by the reaction of chloral (trichloroacetaldehyde, CCl₃CHO) with chlorobenzene (C₆H₅Cl) in the presence of a strong acid catalyst, such as concentrated sulfuric acid. The reaction proceeds in two steps. First, chloral reacts with sulfuric acid to form a carbocation intermediate. This carbocation then acts as an electrophile, attacking the electron-rich phenyl rings of chlorobenzene. The strong electron-withdrawing nature of the chlorine atoms on the chlorobenzene influences the position of substitution, favoring the para positions relative to the point of attachment to the ethane backbone.
The overall reaction can be represented as:
2 C₆H₅Cl + CCl₃CHO $xrightarrow{H2SO4}$ (ClC₆H₄)₂CHCCl₃ + H₂O
The product of this reaction is a mixture of isomers, with p,p’ -DDT being the most abundant and insecticidally active isomer (approximately 75-80% of the mixture). Other isomers include o,p’ -DDT and o,o’ -DDT, which have lower insecticidal activity. The commercial production of DDT focused on maximizing the yield of the p,p’ isomer.
Physical and Chemical Characteristics
DDT is a white, crystalline solid that is virtually insoluble in water but readily soluble in organic solvents such as fats, oils, and many organic solvents like kerosene, acetone, and benzene. This low water solubility and high fat solubility are key to understanding its environmental behavior.
Its chemical stability is another defining characteristic. DDT is resistant to hydrolysis, oxidation, and photolysis, meaning it does not readily break down in water, air, or through exposure to sunlight. This persistence allows it to remain in the environment for extended periods, accumulating in soil, water, and living organisms. The C-Cl bonds within the molecule are strong and require significant energy to break, contributing to its recalcitrance.
The Mechanism of Insecticidal Action
DDT’s effectiveness as an insecticide lies in its ability to disrupt the normal functioning of insect nervous systems. It acts as a potent neurotoxin, primarily by interfering with the voltage-gated sodium channels in the neurons of insects.
Action on Sodium Channels
In a healthy neuron, sodium channels are responsible for the rapid influx of sodium ions into the cell, which generates the electrical impulse (action potential) that allows nerve cells to communicate. These channels open and close in a precisely regulated sequence. DDT binds to the sodium channel and prevents it from closing properly after it has opened. This prolonged opening leads to a persistent influx of sodium ions, causing repetitive and uncontrolled firing of the neuron.
This continuous neuronal excitation results in tremors, uncoordinated movements, paralysis, and ultimately, the death of the insect. The mechanism is thought to involve DDT binding to a specific site on the sodium channel protein, altering its conformation and hindering its inactivation gate from closing. While this disruption is lethal to insects, its effect on mammals is less pronounced, primarily due to differences in sodium channel structure and the metabolic pathways available for detoxification.
Resistance Development
A significant consequence of DDT’s widespread and prolonged use was the evolution of resistance in insect populations. Insects exposed to DDT could develop genetic mutations that conferred resistance. These mutations often involved changes in the genes coding for the sodium channel protein, altering the binding site for DDT, or increasing the insect’s ability to metabolize and detoxify the insecticide.
Detoxification Mechanisms
In insects that can metabolize DDT, the primary detoxification pathway involves enzymatic dehydrochlorination, where a molecule of hydrogen chloride (HCl) is removed from the DDT molecule, converting it into the less toxic compound DDE (dichlorodiphenyldichloroethylene). This metabolic process is often mediated by enzymes such as mixed-function oxidases. However, DDE itself is still persistent and bioaccumulative.
Environmental Persistence and Bioaccumulation
The very stability that made DDT an effective insecticide also became its most significant environmental drawback. Its resistance to degradation meant that once released into the environment, it persisted for years, even decades. This persistence, coupled with its lipophilic nature, led to a phenomenon known as bioaccumulation and biomagnification.
Bioaccumulation
Bioaccumulation is the process by which an organism accumulates a substance at a rate faster than it can be eliminated or metabolized. Because DDT is fat-soluble, it is readily absorbed by organisms and stored in their fatty tissues. As an organism is repeatedly exposed to DDT, the concentration of the chemical within its body increases over time.
Biomagnification
Biomagnification occurs when the concentration of a substance, such as DDT, increases as it moves up through successive trophic levels in a food chain. At the bottom of the food chain, organisms like algae or small invertebrates might absorb DDT from the environment. When these organisms are consumed by herbivores, the DDT is transferred, and since the DDT is not metabolized or excreted, it accumulates in the herbivore’s tissues. This process continues at each higher trophic level, with top predators accumulating the highest concentrations of DDT.
For example, fish living in DDT-contaminated water would accumulate DDT in their tissues. Birds of prey that consume these fish would then ingest a much higher concentration of DDT.
Ecological Impacts
The ecological consequences of DDT’s persistence and biomagnification were profound and devastating for many wildlife populations.
Impact on Birds of Prey
One of the most well-documented and significant impacts of DDT was on bird populations, particularly birds of prey like eagles, ospreys, and peregrine falcons. DDT and its metabolite DDE interfered with calcium metabolism in these birds, leading to the production of eggshells that were abnormally thin and fragile. These thin eggshells would break during incubation, resulting in reproductive failure and drastic population declines for many species. The iconic image of the bald eagle teetering on the brink of extinction due to DDT contamination became a potent symbol of environmental degradation.
Effects on Aquatic Life
Aquatic ecosystems also suffered significantly. DDT could enter waterways through agricultural runoff, industrial discharge, and atmospheric deposition. Fish and other aquatic organisms absorbed DDT from the water and sediment, leading to bioaccumulation within these populations. This contamination could affect the health and reproductive success of fish, amphibians, and invertebrates, disrupting the delicate balance of aquatic food webs.
Broader Ecosystem Effects
Beyond specific species, the widespread presence of DDT in the environment had broader ecosystem effects. It could alter insect populations, impacting pollination and pest control dynamics. Its persistence meant that even after DDT use ceased, contaminated sites continued to pose a risk to wildlife for many years.
Human Health Concerns and Regulatory Actions
While DDT was initially lauded for its role in combating vector-borne diseases like malaria and typhus, concerns about its potential long-term effects on human health began to emerge.
Exposure Pathways
Humans are exposed to DDT primarily through the consumption of contaminated food, particularly fatty foods like meat and dairy products, which can accumulate DDT from the environment. Exposure can also occur through occupational handling of the insecticide and through contaminated drinking water.
Health Effects
Studies have investigated potential links between DDT exposure and various human health issues. While direct causality is often complex to establish due to multifactorial influences, some research has suggested associations between DDT exposure and increased risks of certain cancers, reproductive problems, developmental issues in children, and endocrine disruption. The lipophilic nature of DDT means it can accumulate in human adipose tissue and can be passed from mother to child through breast milk.
Regulatory Bans and International Agreements
The mounting evidence of DDT’s environmental persistence, bioaccumulation, and potential health risks led to increasing calls for its regulation. In the United States, Rachel Carson’s seminal 1962 book “Silent Spring” played a pivotal role in raising public awareness about the dangers of pesticides, including DDT. This public outcry and scientific evidence prompted the U.S. Environmental Protection Agency (EPA) to ban most uses of DDT in 1972.
Many other countries followed suit, implementing bans or severe restrictions on DDT production and use. However, DDT remains on the World Health Organization’s (WHO) list of essential medicines and is still used in some limited public health programs, particularly in malaria-endemic regions, where its benefits in disease prevention are weighed against its environmental risks. The Stockholm Convention on Persistent Organic Pollutants (POPs) aims to eliminate or restrict the production and use of POPs, including DDT, although it allows for specific exemptions for disease vector control.
Legacy and Continued Relevance
The story of DDT is a powerful case study in the complex relationship between chemistry, human innovation, and environmental stewardship. Its rise and fall highlight the importance of understanding the long-term consequences of chemical use and the need for rigorous scientific assessment and precautionary principles in introducing new substances into the environment.
Lessons Learned
The DDT experience underscored the critical need for:
- Comprehensive Environmental Risk Assessment: Evaluating not just immediate efficacy but also persistence, bioaccumulation potential, and toxicity across ecosystems.
- Integrated Pest Management (IPM): Developing strategies that combine biological, cultural, and chemical methods to minimize reliance on single, broad-spectrum pesticides.
- Sustainable Chemistry: Designing chemicals and processes that are inherently less hazardous and more degradable.
- Global Cooperation: Addressing transboundary environmental issues requires international collaboration and agreements.
Ongoing Research and Monitoring
While DDT use has been drastically curtailed, its legacy persists. Environmental monitoring continues to detect DDT and its metabolites in various ecosystems worldwide. Research also continues to investigate its long-term health effects and to develop more effective and environmentally benign alternatives for pest and disease control. The chemistry of DDT, its environmental fate, and its biological interactions remain subjects of scientific inquiry, offering valuable insights into the enduring challenges of managing chemicals in a complex world.
