The realm of chemistry is a fascinating landscape where the fundamental building blocks of matter are understood, categorized, and manipulated. At the heart of this understanding lies the concept of chemical formulas – concise representations that unlock the secrets of molecular composition. Among the myriad of chemical compounds, calcium chloride (CaCl₂) stands out for its diverse applications and the elegant simplicity of its formula. This article delves into the depths of what the calcium chloride formula signifies, exploring its constituents, the principles behind its representation, and the broader implications of understanding such fundamental chemical notations within the vast spectrum of technological innovation.
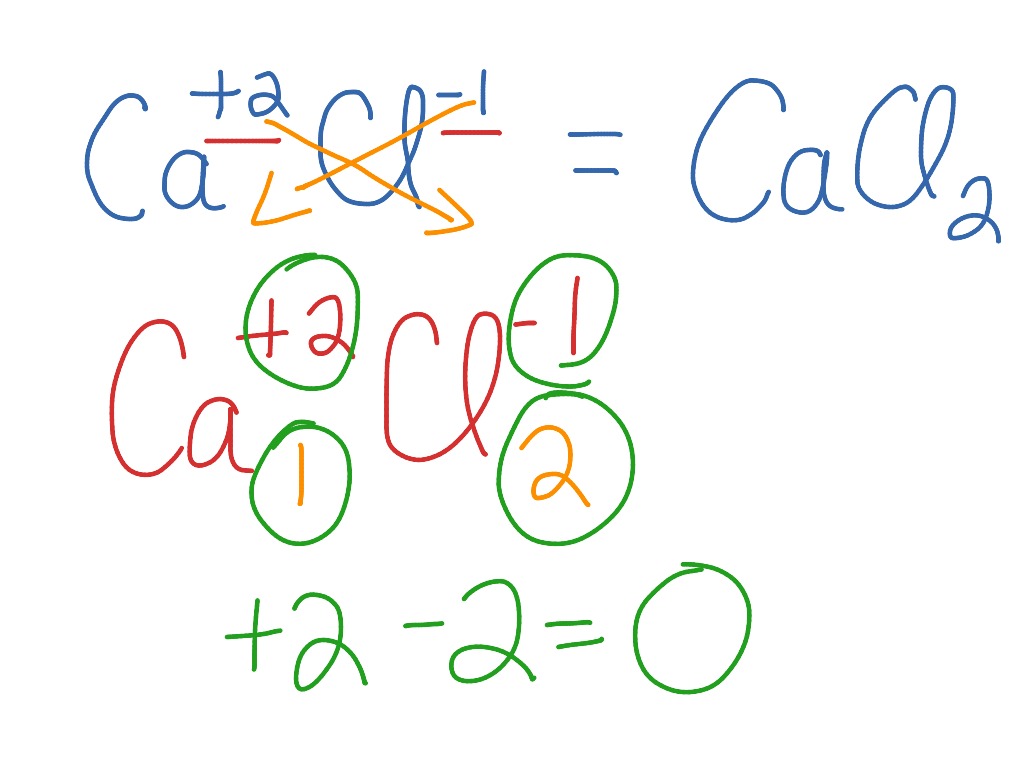
The Anatomy of a Chemical Formula: Deconstructing CaCl₂
A chemical formula is far more than just a string of letters and numbers; it is a precise language that communicates critical information about a compound. When we examine the calcium chloride formula, CaCl₂, we are presented with a clear, unambiguous depiction of its elemental makeup and the stoichiometric ratios of its constituent atoms.
Calcium: The Foundation of the Compound
The first element represented in the formula is Calcium, denoted by the symbol “Ca”. Calcium is an alkaline earth metal, belonging to Group 2 of the periodic table. Its atomic number is 20, indicating that a neutral calcium atom possesses 20 protons and 20 electrons. In its elemental form, calcium is a reactive metal, readily losing its two valence electrons to achieve a stable electron configuration. This tendency to form positively charged ions, specifically a +2 cation (Ca²⁺), is a cornerstone of its chemical behavior and dictates its participation in ionic bonding. The presence of “Ca” in the calcium chloride formula immediately signals the involvement of this essential metal.
Chlorine: The Halogen Partner
Following calcium, we encounter chlorine, represented by the symbol “Cl”. Chlorine is a halogen, a member of Group 17 of the periodic table, with an atomic number of 17. Like calcium, chlorine is a highly reactive element, but its reactivity stems from its strong tendency to gain a single electron to achieve a stable octet, forming a negatively charged ion, the chloride ion (Cl⁻). This strong electronegativity makes chlorine an excellent candidate for forming ionic bonds with electropositive elements like calcium.
The Subscript: Unveiling Stoichiometry
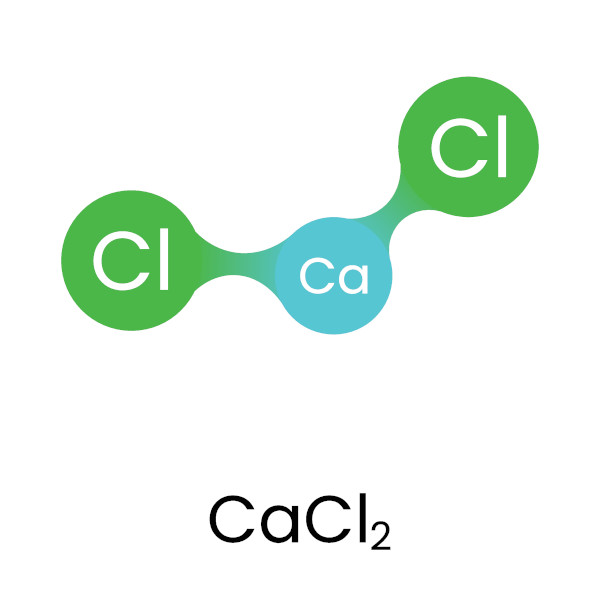
The most crucial element within the calcium chloride formula, in terms of revealing its precise composition, is the subscript. Following the symbol for chlorine (“Cl”), we find the numeral “2”. This subscript is not arbitrary; it represents the number of atoms of that particular element present in one molecule or formula unit of the compound. In the case of CaCl₂, the subscript “2” attached to “Cl” signifies that for every single atom of calcium, there are precisely two atoms of chlorine. This stoichiometric ratio is fundamental to the compound’s existence and its chemical properties. It dictates how calcium and chlorine ions arrange themselves in a crystalline lattice structure in the solid state.
The Ionic Bond: The Glue that Binds
The formation of calcium chloride is a classic example of ionic bonding. Calcium, with its tendency to lose two electrons, and chlorine, with its tendency to gain one electron, engage in a transfer of electrons. To achieve electrical neutrality and stability, one calcium atom donates its two valence electrons, one to each of two separate chlorine atoms. This results in the formation of one calcium cation (Ca²⁺) and two chloride anions (Cl⁻). The electrostatic attraction between the positively charged calcium ion and the negatively charged chloride ions is what holds the compound together, forming the ionic bond. The formula CaCl₂ accurately reflects this 1:2 ratio of ions required for charge balance.
Beyond the Formula: The Significance of Chemical Representation in Technology and Innovation
The ability to concisely and accurately represent chemical compounds through formulas like CaCl₂ is not merely an academic exercise; it forms a bedrock for countless technological advancements and innovations. Understanding chemical formulas empowers scientists and engineers to predict, design, and optimize chemical processes, leading to breakthroughs across diverse fields.
Predictability and Design in Chemical Engineering
In chemical engineering, the understanding of chemical formulas is paramount. When designing a chemical plant to produce or utilize calcium chloride, engineers must meticulously calculate the required quantities of reactants based on their molecular formulas. The CaCl₂ formula informs them that for every mole of calcium they start with, they will require two moles of chlorine (or a chlorine-containing precursor) to achieve a complete reaction. This precise stoichiometric knowledge prevents wastage of raw materials, ensures efficient energy utilization, and guarantees the purity and yield of the final product. Imagine a pharmaceutical company producing a drug that relies on calcium chloride as an excipient; an incorrect understanding of its formula could lead to a product with incorrect dosage or unintended side effects.
Material Science and Compound Development
The field of material science heavily relies on the understanding of chemical formulas to develop new materials with specific properties. Calcium chloride itself exhibits a range of useful properties, such as its hygroscopic nature (ability to absorb moisture) and its ability to lower the freezing point of water. These properties, directly linked to its chemical structure as represented by CaCl₂, make it invaluable in applications like de-icing roads, dust control, and as a drying agent in laboratories. When scientists aim to create novel materials, they begin by understanding the elemental composition and bonding characteristics, all encapsulated within chemical formulas. For instance, developing a new type of biodegradable plastic might involve intricate combinations of elements and functional groups, each precisely defined by its chemical formula.
Research and Development of Novel Applications
The continuous pursuit of innovation often involves repurposing existing compounds or discovering entirely new applications for them. A deep understanding of the calcium chloride formula allows researchers to explore its reactivity and potential interactions with other substances. For example, understanding that CaCl₂ is an ionic compound with a significant charge density on its constituent ions helps in predicting its solubility in various solvents and its potential to participate in different types of chemical reactions. This knowledge can lead to novel uses in areas like advanced battery technologies, where ionic conductivity is crucial, or in the development of novel catalysts for industrial processes. The ability to manipulate and understand chemical reactions at a fundamental level, guided by accurate chemical formulas, fuels the engine of technological progress.
Ensuring Safety and Regulatory Compliance
In industries ranging from food production to manufacturing, adherence to safety standards and regulatory requirements is non-negotiable. Chemical formulas play a critical role in this aspect. For instance, when calcium chloride is used as a food additive (e.g., as a firming agent in canned vegetables or tofu), regulatory bodies require precise labeling, often including the chemical name and formula. This ensures that consumers and manufacturers are aware of the exact chemical composition of the product and can assess any potential risks or allergenic properties. The unambiguous nature of chemical formulas prevents misinterpretation and contributes to a safer and more transparent industrial landscape.
The Broader Implications: Chemical Formulas as a Universal Language
The calcium chloride formula, CaCl₂, is but one example within an intricate and universal language of chemistry. This language allows scientists and engineers worldwide to communicate complex ideas about matter and its transformations with clarity and precision. The fundamental principles behind its construction – element symbols, subscripts for stoichiometry, and implicit understanding of bonding – are applicable to virtually every chemical compound known.
Periodic Table as the Rosetta Stone
The periodic table of elements serves as the ultimate reference for understanding chemical symbols. Each element’s position on the table provides insights into its properties, reactivity, and typical bonding behavior. When encountering an element symbol within a formula, such as “Ca” or “Cl,” one can immediately access a wealth of information about that element from its corresponding position on the periodic table. This interconnectedness makes chemical formulas a powerful tool for deciphering the composition of even unknown substances.
Unlocking the Secrets of Complex Molecules
While CaCl₂ is relatively simple, the principles of chemical formulas extend to incredibly complex molecules, such as DNA, proteins, and pharmaceuticals. For these larger structures, more elaborate notations, like structural formulas and condensed formulas, are employed. However, the foundational concept of representing atoms and their ratios remains the same. For example, understanding the formula for glucose (C₆H₁₂O₆) reveals the exact number of carbon, hydrogen, and oxygen atoms that make up this vital sugar molecule, crucial for understanding its role in biological processes and its potential uses in bio-based technologies.
The Foundation of Scientific Literacy
A basic understanding of chemical formulas is fundamental to scientific literacy in the modern world. From reading ingredient labels on food packaging to comprehending environmental reports about pollutants, the ability to interpret chemical information is increasingly important. By demystifying formulas like CaCl₂, we empower individuals to engage more critically with scientific information and to appreciate the chemical underpinnings of the technologies that shape our lives.

Conclusion: The Enduring Power of Chemical Notation
The seemingly simple query, “what is calcium chloride formula,” opens a gateway to understanding a fundamental aspect of chemistry that underpins significant technological advancements. CaCl₂ is more than just a set of symbols; it is a precise blueprint of a compound, revealing its elemental constituents and their precise ratios. This concise representation is a testament to the power of scientific notation, enabling predictability, design, and innovation across a vast array of industries. From the development of new materials to ensuring the safety of consumer products, the clarity and universality of chemical formulas like that of calcium chloride remain indispensable tools in the ongoing quest for knowledge and technological progress. The ongoing exploration and application of these fundamental chemical principles continue to drive innovation, promising even more exciting breakthroughs in the future.
