A Bunsen burner is a ubiquitous piece of laboratory equipment, a staple in scientific classrooms and research facilities for its ability to produce a controllable, adjustable flame. At its core, the Bunsen burner’s function is simple: to mix a fuel gas with air in precise proportions to achieve a flame suitable for various heating, sterilization, and analytical purposes. Understanding the mechanics and characteristics of the Bunsen flame is fundamental to its safe and effective use in any scientific endeavor.
The Anatomy of a Bunsen Burner
To comprehend the Bunsen flame, one must first appreciate the design of the burner that produces it. The typical Bunsen burner consists of several key components, each playing a crucial role in the gas-air mixture and subsequent combustion process.
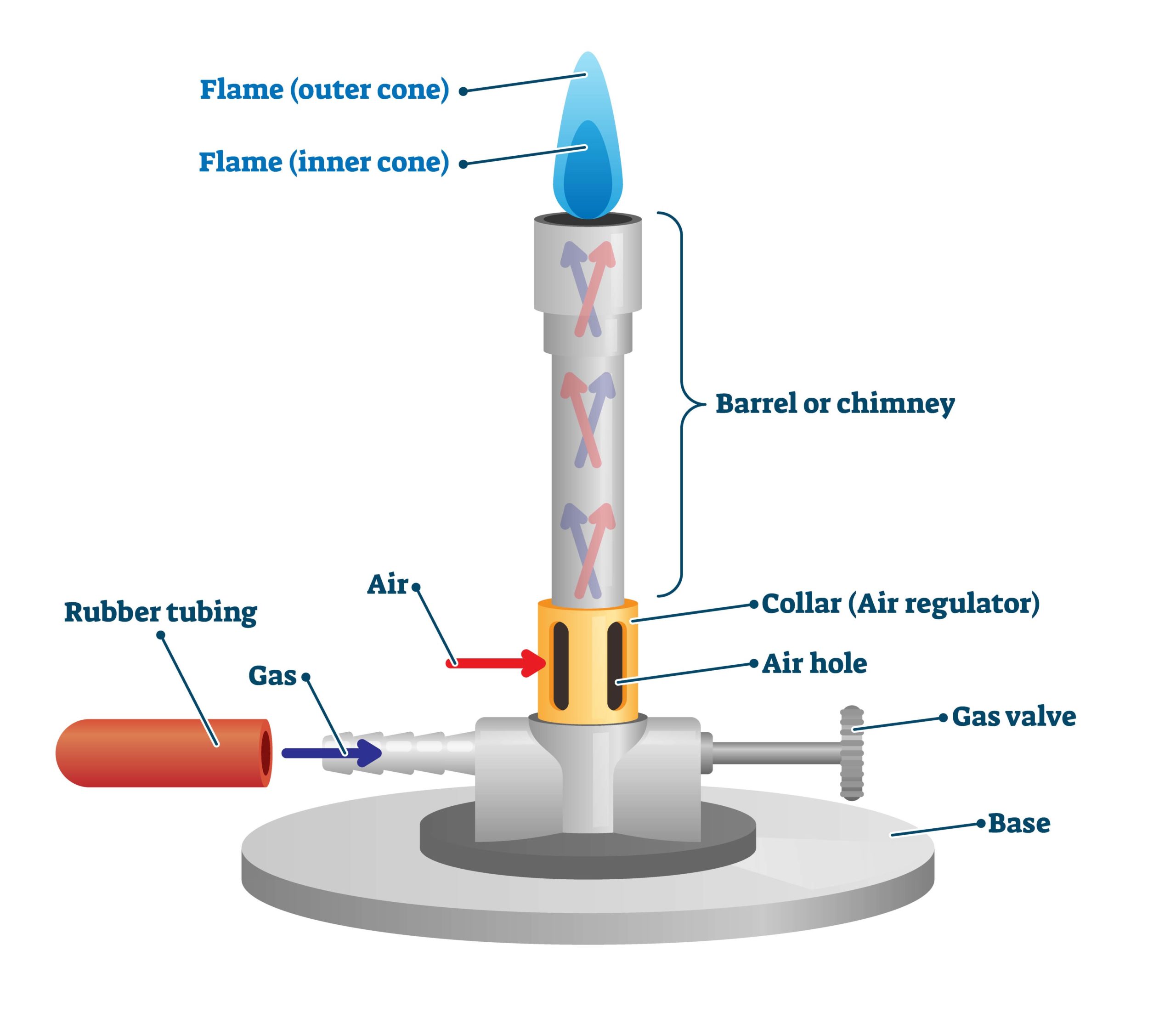
Gas Inlet and Valve
The process begins with the gas supply, usually natural gas (methane) or propane, entering the burner through a gas inlet. This inlet is connected to a laboratory gas line or a portable gas cylinder. Adjacent to the inlet is a needle valve, a control mechanism that regulates the flow rate of the gas. By turning this valve, the user can increase or decrease the amount of fuel gas entering the burner, thereby influencing the intensity of the flame.
Mixing Chamber and Air Holes
Following the valve, the gas travels up through the barrel of the burner. Near the base of the barrel is a collar, often adjustable, which is designed to control the amount of air that mixes with the fuel gas. This collar features openings, known as air holes or vents. As the gas flows upwards, it creates a Venturi effect, drawing ambient air into the barrel through these air holes. The precise size and adjustability of these air holes are paramount to controlling the air-to-gas ratio, which directly dictates the type and temperature of the flame produced. The area where the gas and air first begin to mix is often referred to as the mixing chamber.
Barrel and Chimney Effect
The barrel of the Bunsen burner is essentially a hollow tube, typically made of metal. Its primary function is to guide the mixture of gas and air upwards. As the mixture ascends, it continues to swirl and homogenize. The length and diameter of the barrel are designed to facilitate a stable and consistent flow of the combustible mixture before it reaches the top, where ignition occurs. This upward flow, aided by the heat from the flame above, also contributes to the stable combustion.
Collar and Flame Adjustment
The adjustable collar, as mentioned, is the critical interface between the gas supply and the ambient air. By rotating the collar, the user can selectively open or close the air holes. A fully open collar allows for maximum air intake, leading to a hotter, cleaner flame. Conversely, a partially or fully closed collar restricts air entry, resulting in a cooler, luminous flame. This simple mechanism grants the user significant control over the flame’s characteristics.
Types of Bunsen Flames
The adjustability of the air intake allows for the production of two primary types of Bunsen flames, each with distinct characteristics and applications: the luminous flame and the non-luminous (or blue) flame. The difference lies fundamentally in the completeness of combustion.
The Luminous Flame (Yellow Flame)
When the air holes of a Bunsen burner are closed or significantly restricted, the flame produced is typically a soft, yellow, and luminous one. This type of flame is characterized by incomplete combustion of the fuel gas. The yellow color arises from incandescent particles of unburnt carbon within the flame. These tiny carbon particles become heated to a high temperature, emitting visible light.
Characteristics of the Luminous Flame:
- Temperature: Relatively cool compared to the non-luminous flame, typically around 500-800°C.
- Appearance: Bright yellow, often flickering, and visibly luminous.
- Combustion: Incomplete; produces soot and carbon monoxide due to insufficient oxygen.
- Applications: Generally not ideal for heating in scientific settings due to its low temperature and tendency to deposit soot. However, it can be useful for demonstrating combustion principles or as a temporary light source in specific, low-risk scenarios. Its luminescence also makes it visible, which can be a safety advantage in some situations.
The luminous flame is often the default state when a Bunsen burner is first lit, as the air holes are typically closed. It is important to recognize this flame and adjust the burner to achieve a more efficient and hotter flame when required.
The Non-Luminous Flame (Blue Flame)
By opening the air holes of the Bunsen burner, more ambient air is drawn into the mixing chamber, allowing for a more complete combustion of the fuel gas. This results in a non-luminous, blue flame, which is the preferred flame for most laboratory applications. The blue color is indicative of the chemical reactions occurring within the flame, primarily the combustion of hydrocarbons with sufficient oxygen to produce carbon dioxide and water vapor.
Characteristics of the Non-Luminous Flame:
- Temperature: Significantly hotter than the luminous flame, reaching temperatures of 1000-1500°C, depending on the gas and air mixture.
- Appearance: Primarily blue, with distinct inner and outer cones. It is less visible in bright light than the yellow flame.
- Combustion: More complete; produces fewer byproducts like soot and carbon monoxide.
- Applications: Ideal for heating substances, boiling water, sterilizing equipment, and performing qualitative and quantitative chemical analyses.
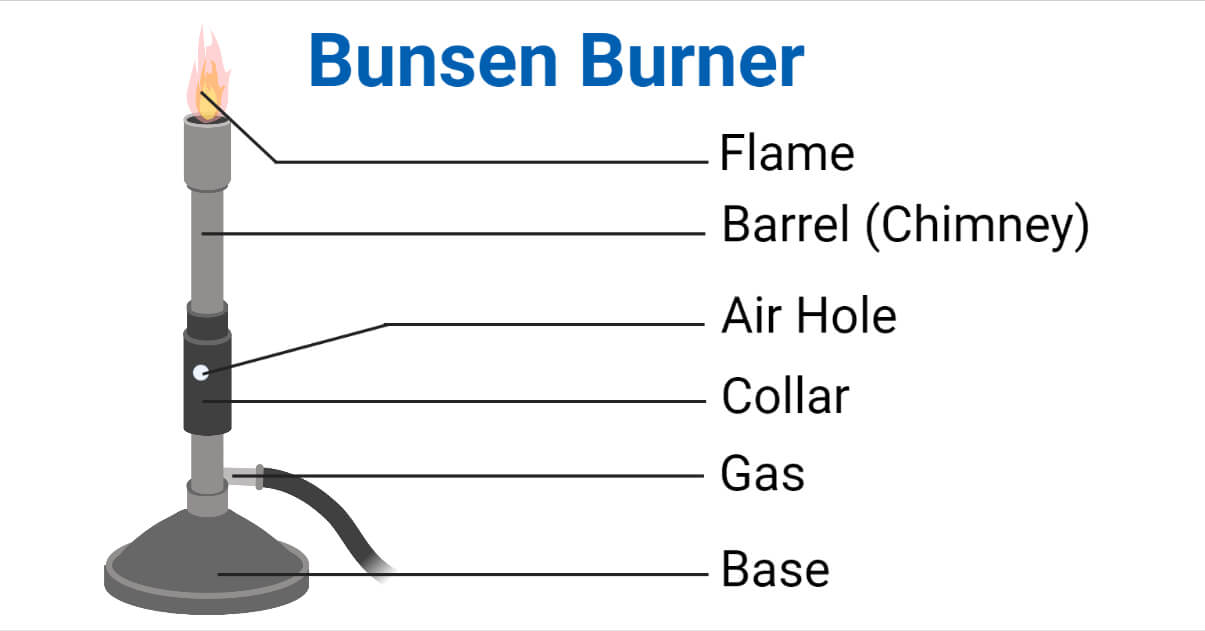
The non-luminous flame is further characterized by its distinct structure, which provides further insight into the combustion process.
The Inner Cone
The inner cone of the non-luminous flame is a pale blue or violet region near the top of the barrel. This is where the initial mixing and partial combustion of the gas and air occurs. It is a region of relatively low temperature and is rich in unburnt fuel gas. Care must be taken not to introduce materials directly into the inner cone, as they might not be heated effectively or could even quench the flame.
The Outer Cone (or Roaring Flame)
Surrounding the inner cone is a larger, darker blue outer cone. This is the hottest part of the Bunsen flame and is where the primary combustion takes place. The outer cone is characterized by a more vigorous and complete reaction between the fuel and oxygen. When the air holes are widely open, the flame can become quite noisy and turbulent, often described as a “roaring” flame. This is the ideal zone for heating and is often referred to as the “perfect flame” for laboratory work.
Controlling the Bunsen Flame
The skillful manipulation of the Bunsen burner’s controls is essential for achieving the desired flame for a given task. This control is primarily exercised through the adjustment of the gas flow rate and the air intake.
Gas Flow Rate Adjustment
The needle valve at the base of the burner controls the supply of fuel gas. A higher gas flow rate will generally lead to a larger flame, assuming adequate air is supplied. Conversely, a lower gas flow rate will produce a smaller flame. It is important to adjust the gas flow to achieve a flame of appropriate size for the heating task without being excessively large or unstable.
Air Hole Adjustment (Collar)
The most critical control for tailoring the flame’s characteristics is the adjustable collar that regulates the air holes.
- Closed Air Holes: Produces a luminous, yellow flame (incomplete combustion). This is often the initial state upon lighting.
- Partially Open Air Holes: Creates a flame that transitions from yellow to orange and then to a fainter blue. The temperature increases as more air is introduced.
- Fully Open Air Holes: Results in the hottest, non-luminous blue flame. This is typically the desired flame for most heating and sterilization purposes.
Striking a Bunsen Flame
The proper procedure for lighting a Bunsen burner involves safety and achieving the desired flame efficiently.
- Close Air Holes: Ensure the air holes are initially closed or nearly closed.
- Turn on Gas: Open the gas valve slowly to allow gas to flow through the burner.
- Ignite: Use a match or a striker to ignite the gas at the top of the barrel. A yellow flame will likely appear.
- Adjust Air: Once the flame is stable, slowly open the air holes by rotating the collar until the desired blue flame is achieved. If the flame “strikes back” (burns down into the barrel), the air holes are too open, or the gas flow is too low. In such cases, turn off the gas immediately, let the burner cool, and relight, adjusting the air holes more cautiously.
Safety Considerations for Bunsen Flames
The Bunsen burner, while an indispensable tool, involves open flames and hot surfaces, necessitating strict adherence to safety protocols.
Flammability and Ventilation
The fuel gas used by Bunsen burners is flammable. It is crucial to ensure that the laboratory is well-ventilated to prevent the accumulation of gas in the event of a leak. Flammable materials, such as paper, solvents, and volatile chemicals, should be kept at a safe distance from the open flame.
Heat and Burns
The Bunsen flame, especially the non-luminous blue flame, reaches very high temperatures. Direct contact with the flame or hot objects heated by the flame can cause severe burns. Always handle hot glassware or equipment with appropriate heat-resistant tongs or gloves. Allow heated items to cool before touching them.
Soot and Carbon Monoxide
Incomplete combustion, as seen in the luminous yellow flame, produces soot and carbon monoxide. Soot can accumulate on glassware, making it difficult to see through, and can be a fire hazard. Carbon monoxide is a colorless, odorless, and poisonous gas. Proper ventilation is essential to prevent its buildup.
Flame “Striking Back”
As mentioned earlier, if the air holes are too widely open or the gas flow is too low, the flame can burn down into the barrel of the burner. This is known as “striking back” and can cause the burner to overheat, potentially damaging it and creating a safety hazard. If striking back occurs, immediately turn off the gas supply, allow the burner to cool completely, and then relight with appropriate adjustments to the air intake and gas flow.
Proper Extinguishing
To extinguish a Bunsen burner, simply turn off the gas supply at the needle valve. Ensure the flame is completely out before leaving the area.
Applications of the Bunsen Flame in Science
The versatility of the Bunsen burner’s adjustable flame makes it indispensable across a wide range of scientific disciplines.
Heating and Sterilization
The primary use of the Bunsen burner is for heating substances. The non-luminous blue flame provides sufficient heat to boil water, melt solids, and drive chemical reactions that require elevated temperatures. It is also used for sterilizing equipment, such as inoculating loops in microbiology, by passing them through the hottest part of the flame.
Glassblowing and Manipulation
In some scientific contexts, particularly in chemistry labs that require custom glassware, a Bunsen burner can be used for rudimentary glass manipulation. The controlled heat allows for the softening and bending of glass tubes to create specific shapes or connections.
Qualitative Analysis
Bunsen flames are used in flame tests for qualitative chemical analysis. Certain metal ions, when introduced into the flame, emit characteristic colors. For example, sodium produces a bright yellow flame, potassium a lilac flame, and copper a green flame. Observing these colors helps identify the presence of specific elements in a sample.
Demonstrations and Education
In educational settings, the Bunsen burner is a fundamental tool for demonstrating principles of combustion, heat transfer, and chemical reactions. The visible difference between the luminous and non-luminous flames provides a tangible lesson in the importance of complete versus incomplete combustion.
In conclusion, the Bunsen flame is more than just a simple fire; it is a carefully controlled scientific tool whose properties are dictated by the precise interplay of fuel gas and air. Understanding its anatomy, the types of flames it can produce, and the methods for controlling it is crucial for its safe and effective utilization in a vast array of scientific applications. Mastering the Bunsen burner is a foundational skill for anyone venturing into a laboratory environment.
