In the intricate world of chemical reactions, understanding the journey from reactants to products is paramount. While we often focus on the starting materials and the final compounds, a crucial, often transient, player exists in between: the intermediate. Chemical intermediates are not the beginning nor the end of a reaction but rather a species formed during the multi-step process that then reacts further to yield the final product. They are the stepping stones, the brief but essential stages that bridge the gap between initial and final molecular configurations. Grasping the nature and significance of these intermediates is key to comprehending reaction mechanisms, controlling reaction outcomes, and designing new synthetic pathways.
The Elusive Nature of Intermediates
Chemical intermediates are characterized by their fleeting existence. They are typically formed and consumed within a matter of microseconds or even nanoseconds. This ephemeral nature makes them challenging to isolate and study directly. Unlike stable reactants and products, intermediates often possess high reactivity, a direct consequence of their electronic structure. They might have incomplete octets, significant charge separation, or strained molecular geometries, all of which drive them towards further transformation.
Types of Intermediates
The landscape of chemical intermediates is diverse, with several recurring archetypes that chemists frequently encounter. These classifications are based on the electronic and structural features of the transient species.
Carbocations
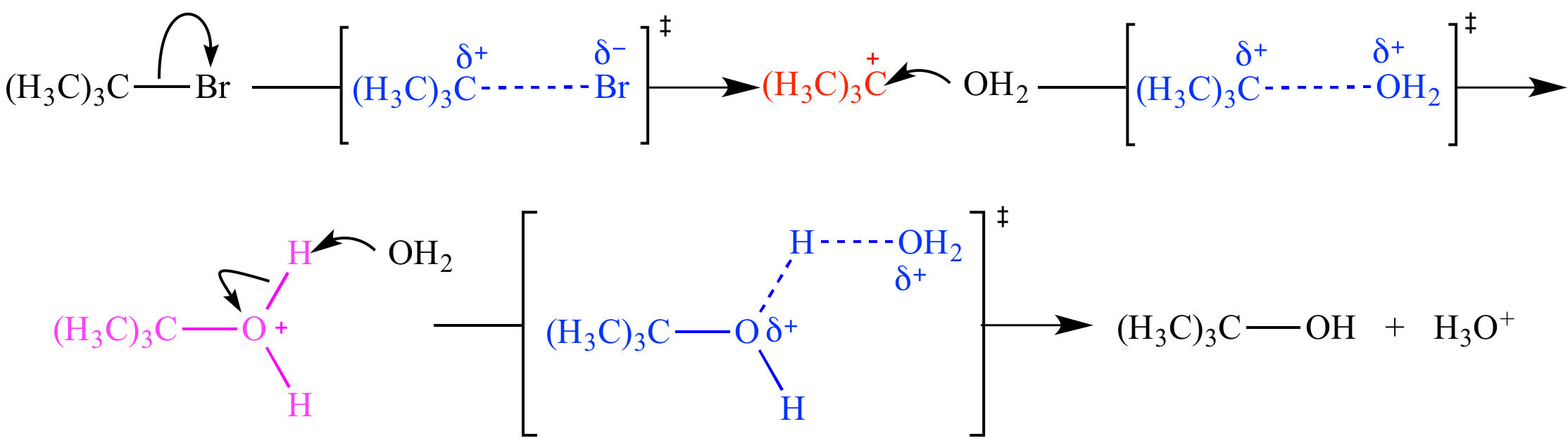
Carbocations are species where a carbon atom bears a positive formal charge. This positively charged carbon typically has only six valence electrons, making it electron-deficient and highly susceptible to attack by nucleophiles (electron-rich species). The stability of carbocations is influenced by several factors, most notably hyperconjugation and the inductive effect of neighboring alkyl groups. Tertiary carbocations (where the positively charged carbon is bonded to three other carbon atoms) are generally more stable than secondary, which are more stable than primary. Resonance stabilization also plays a significant role; for example, allylic and benzylic carbocations are considerably stabilized by the delocalization of the positive charge over multiple atoms.
Carbanions
In stark contrast to carbocations, carbanions feature a carbon atom with a negative formal charge. This carbon atom possesses an extra electron, making the carbanion a potent nucleophile and a strong base. The stability of carbanions is enhanced by electron-withdrawing groups, which can delocalize the negative charge, and by the hybridization of the carbon atom. Carbanions derived from sp-hybridized carbons are more stable than those from sp2, which are more stable than those from sp3, due to the higher electronegativity of sp hybridized orbitals. Organometallic reagents, such as Grignard reagents and organolithium compounds, are common examples of species that can be considered as having significant carbanionic character.
Free Radicals
Free radicals are characterized by the presence of an unpaired electron on an atom, most commonly carbon. This unpaired electron makes them highly reactive, as they are driven to pair up with another electron to achieve a stable configuration. Free radical reactions often proceed via chain mechanisms involving initiation, propagation, and termination steps. The stability of free radicals is influenced by similar factors to carbocations, including hyperconjugation and resonance. Tertiary radicals are generally more stable than secondary and primary radicals.
Carbenes
Carbenes are neutral species containing a divalent carbon atom, meaning the carbon atom is bonded to only two other atoms and has two non-bonding valence electrons. These non-bonding electrons can exist as a lone pair (singlet carbene) or as a single electron in two different orbitals (triplet carbene). Carbenes are highly reactive electrophiles, often participating in insertion reactions into sigma bonds or addition reactions to pi bonds. Examples include methylene (CH2) and dichlorocarbene (:CCl2).
Reaction Mechanisms and Intermediates
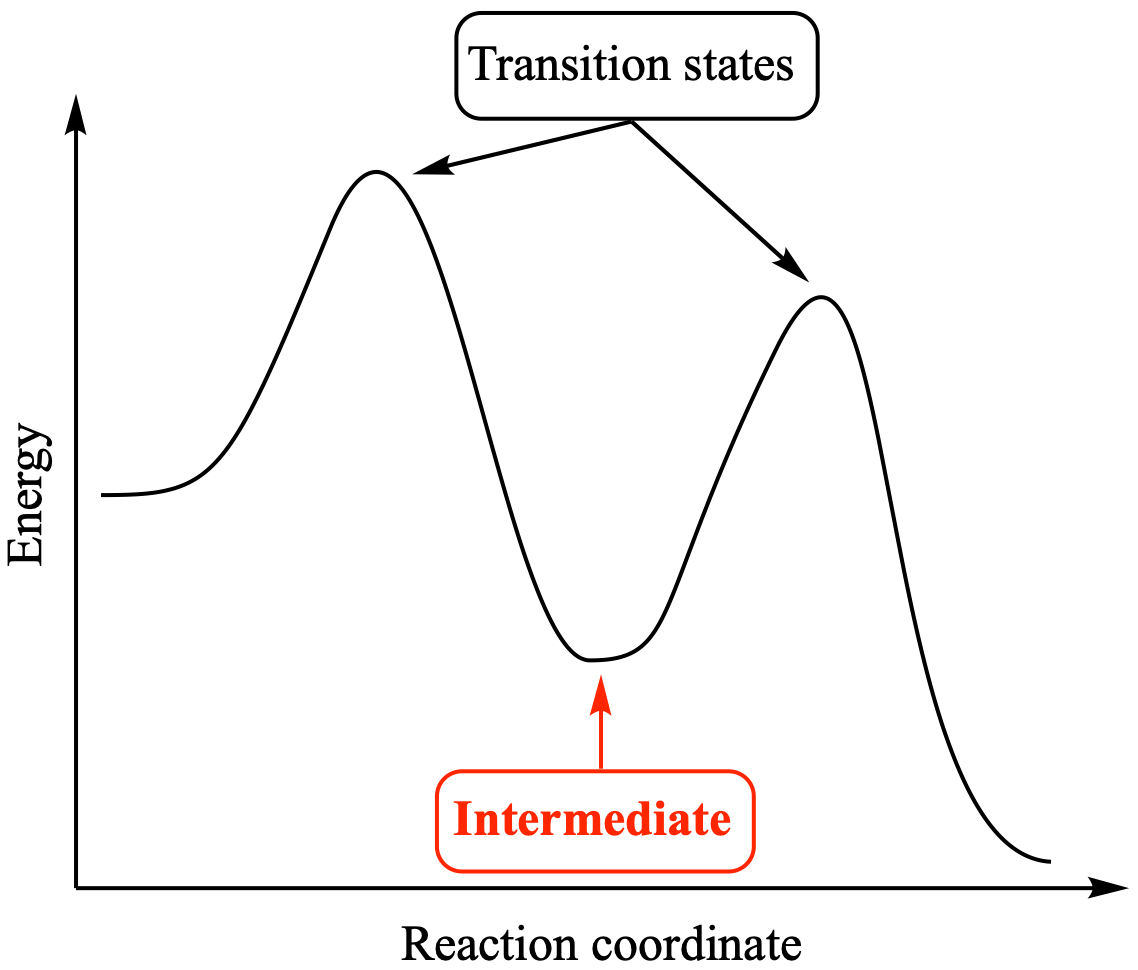
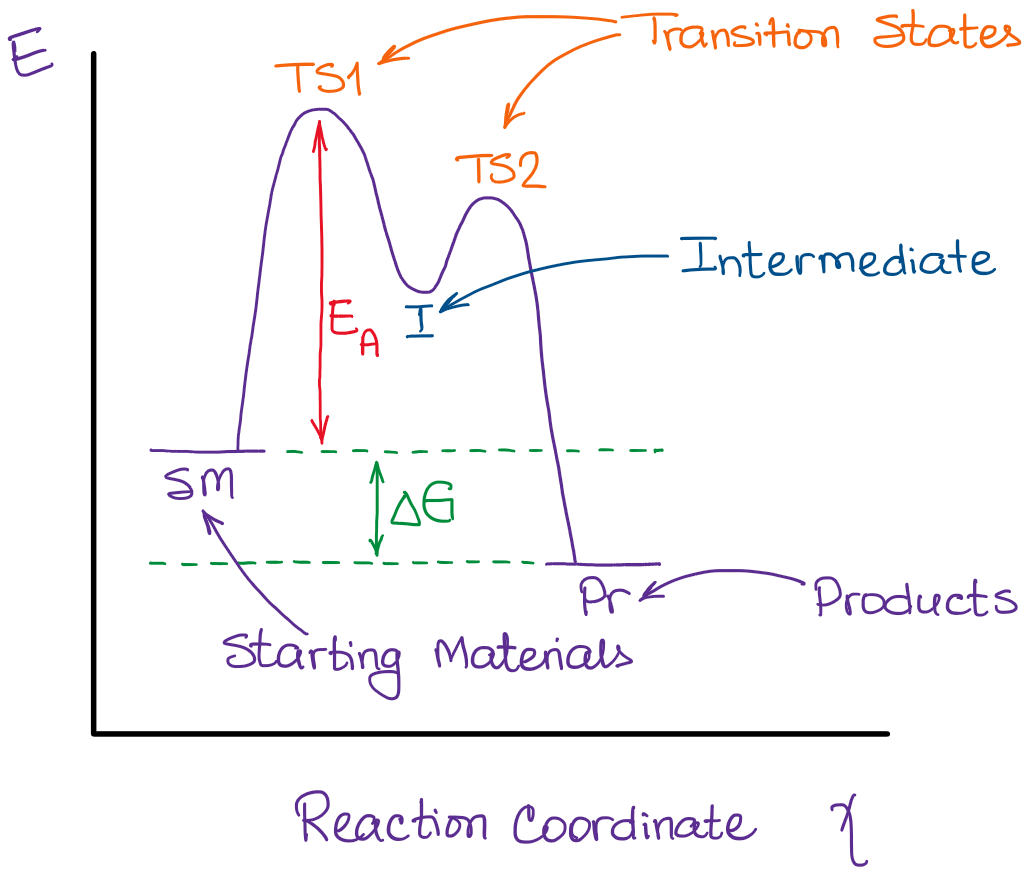
The concept of intermediates is central to understanding reaction mechanisms, which are detailed step-by-step descriptions of how a chemical reaction occurs. By identifying the intermediates formed, chemists can piece together the pathway by which reactants transform into products. This often involves drawing energy diagrams (potential energy surfaces) that illustrate the relative energies of reactants, intermediates, transition states, and products. Intermediates appear as local minima on these energy diagrams, representing transiently stable species.
Stepwise vs. Concerted Reactions
Reactions can be broadly classified as either stepwise or concerted. In a concerted reaction, all bond breaking and bond forming occur in a single step, passing through a single transition state. There are no intermediates. In contrast, a stepwise reaction involves the formation and consumption of one or more intermediates. Each step in a stepwise reaction has its own transition state.
The Role of Intermediates in Kinetics
The rate law of a chemical reaction, which describes how the reaction rate depends on the concentrations of reactants, can often be elucidated by understanding the role of intermediates. In many multi-step reactions, the overall rate is limited by the slowest step, known as the rate-determining step. If an intermediate is formed rapidly and then reacts slowly in the rate-determining step, its concentration will be low and relatively constant, allowing for approximations (like the steady-state approximation) to be used in deriving the rate law. Conversely, if an intermediate is involved in the rate-determining step, its formation or consumption directly influences the overall reaction rate.
Detection and Characterization of Intermediates
The transient nature of intermediates poses significant challenges to their direct observation. However, chemists have developed a sophisticated arsenal of techniques to detect and characterize these elusive species.
Spectroscopic Techniques
Spectroscopy plays a pivotal role in identifying intermediates. Various spectroscopic methods can probe the electronic and vibrational structure of short-lived species.
Nuclear Magnetic Resonance (NMR) Spectroscopy
While conventional NMR spectroscopy typically requires stable molecules, specialized techniques, such as low-temperature NMR and fast-acquisition NMR, can capture the spectra of transient intermediates. By rapidly quenching a reaction at very low temperatures or by employing rapid pulsing sequences, it is sometimes possible to detect and characterize intermediates that exist for even short periods.
Infrared (IR) and Raman Spectroscopy
IR and Raman spectroscopy are sensitive to the vibrational modes of molecules. Changes in vibrational frequencies can indicate the formation of new functional groups or alterations in bond strengths associated with an intermediate. Time-resolved IR spectroscopy, in particular, allows for the observation of spectral changes occurring within microseconds to milliseconds, making it invaluable for studying fast reactions.
Electron Paramagnetic Resonance (EPR) Spectroscopy
EPR spectroscopy is specifically designed to detect species with unpaired electrons, making it the technique of choice for identifying and characterizing free radicals. The characteristic spectral patterns in EPR provide information about the radical’s structure and environment.
Ultraviolet-Visible (UV-Vis) Spectroscopy
UV-Vis spectroscopy probes electronic transitions in molecules. Many intermediates, particularly those with conjugated pi systems or significant charge separation, absorb light in the UV-Vis region. Time-resolved UV-Vis spectroscopy can track the appearance and disappearance of these absorbing species during a reaction.
Trapping and Spectrographic Analysis
One common strategy to confirm the existence of an intermediate is through “trapping.” This involves introducing a reagent that reacts rapidly and selectively with the suspected intermediate. If the trapping reagent forms a stable product that can be identified and quantified, it provides strong evidence for the intermediate’s existence and its chemical reactivity. For example, if a reaction is suspected to form a carbene, adding an alkene can lead to cyclopropane formation, trapping the carbene.
Computational Chemistry
In conjunction with experimental techniques, computational chemistry has become an indispensable tool for studying intermediates. Quantum mechanical calculations can predict the structure, stability, and spectroscopic properties of proposed intermediates, often guiding experimental efforts. By calculating reaction pathways and energy profiles, computational methods can help confirm whether a proposed intermediate is energetically plausible and consistent with observed kinetics. These calculations can also provide detailed insights into the electronic distribution and bonding within the intermediate.
The Significance of Intermediates in Synthesis and Industry
Understanding and controlling chemical intermediates is not merely an academic exercise; it has profound implications for synthetic chemistry and industrial processes.
Optimizing Reaction Yields and Selectivity
By elucidating the reaction mechanism and identifying the intermediates, chemists can manipulate reaction conditions to favor the desired pathway and minimize side reactions. This can involve adjusting temperature, pressure, solvent, catalyst, or reactant concentrations. For instance, if a particular intermediate is prone to decomposition or leads to unwanted byproducts, altering conditions to either prevent its formation or ensure its rapid conversion to the desired product becomes a key optimization strategy. Selectivity, the ability to form one product over others, is often dictated by the relative stabilities and reactivities of different possible intermediates.
Designing Novel Catalysts
Many catalytic processes rely on the formation and facile interconversion of catalytic intermediates. Understanding the nature of these intermediates allows for the rational design of more efficient and selective catalysts. For example, in organometallic catalysis, the catalytic cycle involves a series of intermediates where the metal center undergoes various transformations. By studying these intermediates, researchers can engineer ligands and metal precursors to stabilize desired intermediates and promote specific catalytic steps.
Industrial Chemical Synthesis
Intermediates are cornerstones of industrial chemical production. Many large-scale syntheses involve multiple steps, and specific compounds are isolated and purified as intermediates before being carried forward to the next stage. For example, in the petrochemical industry, a vast array of intermediates are generated and transformed through processes like cracking, reforming, and polymerization to produce fuels, plastics, and other essential materials. The efficient synthesis and handling of these intermediates are critical for economic viability and environmental sustainability.
Pharmaceutical and Fine Chemical Manufacturing
In the pharmaceutical industry, the synthesis of complex drug molecules often involves numerous steps, with specific intermediates being crucial building blocks. The ability to synthesize these intermediates cleanly and efficiently is paramount to producing safe and effective medications. Similarly, in the fine chemical industry, where specialized chemicals are produced in smaller quantities but with high purity, the control of intermediates is essential for achieving the desired product specifications.
Conclusion: The Unsung Heroes of Chemical Transformation
Chemical intermediates, though often unseen and transient, are fundamental to the art and science of chemistry. They are the pivotal species that dictate the course of a reaction, bridge the gap between reactants and products, and offer chemists profound insights into the molecular choreography of chemical change. Through a combination of advanced spectroscopic techniques, ingenious trapping strategies, and powerful computational tools, chemists continue to unravel the mysteries of these elusive entities. The ability to detect, characterize, and control intermediates empowers us to design more efficient syntheses, develop innovative catalysts, and drive progress in countless fields, from material science to medicine. They are, in essence, the unsung heroes of chemical transformation, silently orchestrating the creation of the molecular world around us.
