Alkaline phosphatase (ALP) is an enzyme found in virtually all tissues of the body, with particularly high concentrations in the liver, bone, kidneys, intestines, and placenta. While its physiological roles are diverse, including bone mineralization and fat absorption, its presence and levels in the blood serum serve as a crucial indicator in a wide array of diagnostic applications. From the perspective of Tech & Innovation, understanding alkaline phosphatase in blood is not just about biochemistry; it’s about the sophisticated technologies that enable its measurement, the analytical platforms that interpret its variations, and the potential it holds for advancing predictive diagnostics and personalized medicine.
The technological journey of alkaline phosphatase analysis has evolved significantly, moving from basic colorimetric assays to highly automated, high-throughput systems integrated with advanced data analytics. These advancements are driven by the relentless pursuit of accuracy, speed, and cost-effectiveness in clinical diagnostics, a core tenet of innovation in the health technology sector.
The Technological Underpinnings of ALP Measurement
The accurate quantification of alkaline phosphatase in blood is a testament to the progress in analytical chemistry and laboratory automation. Modern diagnostic laboratories rely on sophisticated instrumentation and precise methodologies to ensure reliable results, which are critical for informed medical decisions.
Enzymatic Assays: The Foundation of Quantification
At the heart of ALP measurement lies enzymatic assay technology. These assays exploit the catalytic activity of ALP to convert a colorless substrate into a colored product. The intensity of the color produced is directly proportional to the concentration of ALP in the sample. Historically, methods involved manual incubation steps and spectrophotometric readings. However, innovation has led to fully automated systems that streamline this process.
Substrate Chemistry and Chromogenic Development
The choice of substrate is paramount. Common substrates like p-nitrophenyl phosphate (pNPP) are hydrolyzed by ALP, releasing p-nitrophenol, which can be quantified spectrophotometrically at a specific wavelength (typically around 405 nm). More advanced substrates might offer enhanced sensitivity or specificity. The development of these substrates, along with the optimized buffer systems and reaction conditions, represents a significant chemical innovation enabling precise enzymatic activity measurement. The integration of these optimized reaction kinetics into automated analyzers minimizes variability and enhances reproducibility.
Spectrophotometry and Colorimetry: From Benchtop to Automation
Spectrophotometry, the measurement of light absorption by a sample, is the cornerstone of colorimetric assays. Modern clinical chemistry analyzers utilize sophisticated spectrophotometers capable of rapid, precise measurements. These instruments often employ diode array detectors, allowing for simultaneous measurement at multiple wavelengths, which can be used for kinetic analysis and interference correction. The miniaturization and high-speed processing capabilities of these devices are direct outcomes of technological advancements in optical engineering and microfluidics.
Automation and High-Throughput Screening
The transformation of ALP measurement from a laboratory-specific procedure to a routine diagnostic test is largely due to automation. High-throughput analyzers, a hallmark of modern clinical laboratories, can process hundreds of samples per hour. These systems integrate sample handling, reagent dispensing, incubation, and measurement into a seamless workflow.
Robotics and Sample Handling
Automated systems employ robotics for sample loading, identification (often via barcode scanning), and aspiration. Precision liquid handling systems, utilizing advanced pumps and sensors, ensure accurate reagent volumes are dispensed, crucial for assay reproducibility. The integration of sample tracking and management software further enhances efficiency and reduces the risk of human error, a critical aspect of quality control in diagnostic technology.
Reagent Systems and Standardization
The development of stable, ready-to-use reagent kits is another area of innovation. These kits, often supplied in pre-filled cartridges or vials, simplify the assay procedure and ensure consistent performance. Standardization of reagents and assay protocols across different platforms is also a key focus, driven by regulatory requirements and the need for inter-laboratory comparability, facilitated by advancements in quality control technologies and reference materials.
Advanced Analytical Techniques and Isoenzyme Separation
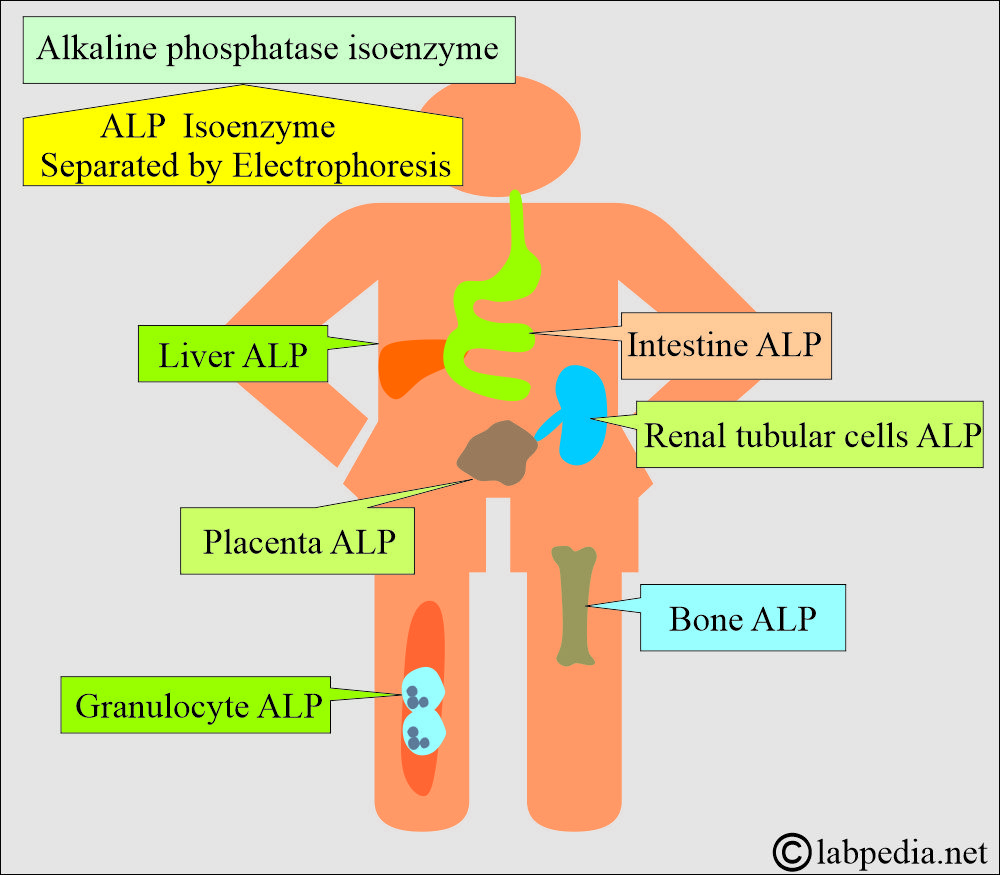
Beyond basic quantification, technological innovations allow for a deeper understanding of ALP by differentiating its various isoenzymes. Different tissues produce distinct forms of ALP, and their relative proportions can provide more specific diagnostic clues.
Isoenzyme Electrophoresis: Visualizing Tissue Origin
Electrophoresis is a powerful separation technique widely employed in clinical diagnostics. For ALP, isoenzyme electrophoresis separates the different forms of the enzyme based on their charge and size as they migrate through a supporting medium (like agarose gel or cellulose acetate) under an electric field.
Gel Electrophoresis and Staining
Traditional gel electrophoresis methods involve running the sample on a gel, followed by incubation with a chromogenic substrate and visualization of distinct bands corresponding to different ALP isoenzymes (e.g., bone, liver, intestinal, placental). Advances in gel matrices and staining techniques have improved resolution and reduced assay time. The development of automated electrophoresis systems further enhances throughput and standardization.

Capillary Electrophoresis: Precision and Speed
Capillary electrophoresis (CE) offers a more modern and efficient alternative to traditional gel electrophoresis. In CE, separation occurs within a narrow fused-silica capillary filled with an electrolyte buffer. The high electric fields and precise control over buffer composition and temperature allow for rapid and highly resolved separations. Detection is typically achieved by UV-Vis spectrophotometry or fluorescence, integrated directly into the capillary system. CE offers significant advantages in terms of speed, reduced reagent consumption, and ease of automation, aligning perfectly with the Tech & Innovation ethos of developing more efficient diagnostic tools.
Immunological Assays: Targeted Detection
Immunological techniques, such as enzyme-linked immunosorbent assays (ELISAs) and chemiluminescent immunoassays (CLIAs), offer high specificity and sensitivity for detecting and quantifying ALP and its isoenzymes. These methods utilize antibodies that are specifically designed to bind to particular ALP isoenzymes.
Monoclonal Antibodies and Assay Design
The development of highly specific monoclonal antibodies against human ALP isoenzymes has been a significant biotechnological innovation. These antibodies enable the design of immunoassays that can selectively target and quantify specific ALP forms, even in the presence of other proteins.
Chemiluminescence and Fluorescence Detection
Modern immunoassays frequently employ chemiluminescent or fluorescent labels. Chemiluminescence, the emission of light from a chemical reaction, offers extremely high sensitivity, allowing for the detection of very low concentrations of analytes. Fluorescence detection also provides excellent sensitivity and a wide dynamic range. The integration of these detection technologies into automated immunoassay platforms has revolutionized diagnostic testing, offering faster turnaround times and improved diagnostic accuracy.
Data Analytics and the Future of ALP Diagnostics
The wealth of data generated by ALP testing, when combined with other clinical information, is increasingly being leveraged through advanced data analytics and artificial intelligence, marking the cutting edge of Tech & Innovation in healthcare.
Predictive Modeling and Risk Stratification
Elevated ALP levels are associated with various conditions, including liver disease, bone disorders (like Paget’s disease and osteoporosis), rickets, and certain cancers. By analyzing ALP levels in conjunction with patient demographics, other laboratory markers, and imaging data, sophisticated algorithms can be developed to predict the risk of developing these conditions. This moves diagnostics from reactive identification to proactive risk stratification.
Machine Learning in Diagnostic Interpretation
Machine learning algorithms can be trained on vast datasets to identify subtle patterns in ALP levels and their correlations with disease progression or treatment response that might not be apparent to human observers. This can lead to earlier and more accurate diagnoses, especially in complex or early-stage diseases. For example, AI could be trained to identify specific ALP isoenzyme profiles that are highly indicative of early-stage bone metastasis in cancer patients.
Personalized Medicine and Treatment Monitoring
The precise measurement and interpretation of ALP, particularly its isoenzymes, can guide personalized treatment strategies. For instance, understanding the bone-specific component of elevated ALP can inform the selection of therapies for osteoporosis. Furthermore, monitoring changes in ALP levels over time, analyzed through sophisticated data platforms, can provide real-time feedback on the efficacy of treatment, allowing for timely adjustments and optimizing patient outcomes. This data-driven approach to healthcare is a prime example of Tech & Innovation driving personalized medicine.
Integration with Digital Health Platforms
The future of ALP diagnostics lies in its seamless integration into broader digital health ecosystems. This includes electronic health records (EHRs), patient portals, and wearable technology.
Real-time Monitoring and Alerts
As diagnostic technologies become more portable and accessible, there is potential for real-time monitoring of relevant biomarkers, including ALP, in specific patient populations. Data from these devices can be transmitted wirelessly and analyzed by cloud-based platforms, triggering alerts for healthcare providers in case of significant deviations from normal ranges, facilitating timely interventions.
Big Data and Population Health Management
Aggregated ALP data from large patient populations, when analyzed through advanced data analytics, can provide valuable insights into population health trends, disease prevalence, and the effectiveness of public health interventions. This big data approach, enabled by the underlying technological infrastructure, is crucial for improving public health strategies and resource allocation.
In conclusion, alkaline phosphatase in blood is far more than a simple biochemical marker. From a Tech & Innovation perspective, it represents a vital component in a dynamic and evolving landscape of diagnostic technology. The continuous advancements in assay methodologies, automation, separation techniques, and data analytics are transforming our ability to measure, interpret, and utilize ALP levels for more accurate diagnoses, personalized treatments, and ultimately, better health outcomes. The journey from basic enzymatic assays to sophisticated AI-driven predictive models underscores the profound impact of technological innovation on the field of clinical diagnostics.
