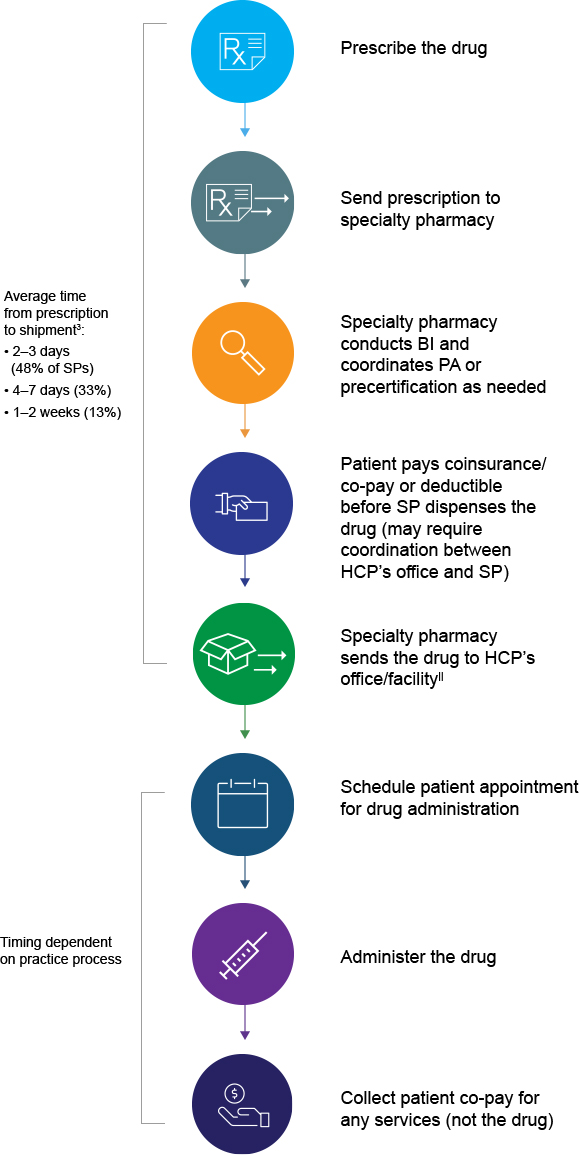
The pharmaceutical landscape is constantly evolving, with a significant portion of innovation and investment focused on a particular class of medications: speciality drugs. These are not your everyday over-the-counter remedies or common prescription pills. Instead, they represent a distinct category of therapeutics characterized by their complexity, high cost, and often, their targeted application to treat rare or chronic, life-altering conditions. Understanding what constitutes a speciality drug is crucial for patients, healthcare providers, insurers, and policymakers alike, as it impacts access, affordability, and the overall direction of medical advancements.
Defining Speciality Drugs: Beyond the Ordinary
At its core, a speciality drug is defined by a confluence of factors that set it apart from traditional pharmaceuticals. These drugs are typically manufactured using complex biological processes, rather than conventional chemical synthesis. This biological origin often means they are large, intricate molecules, frequently derived from living cells or organisms. This manufacturing complexity directly contributes to their higher production costs and, consequently, their elevated price points.
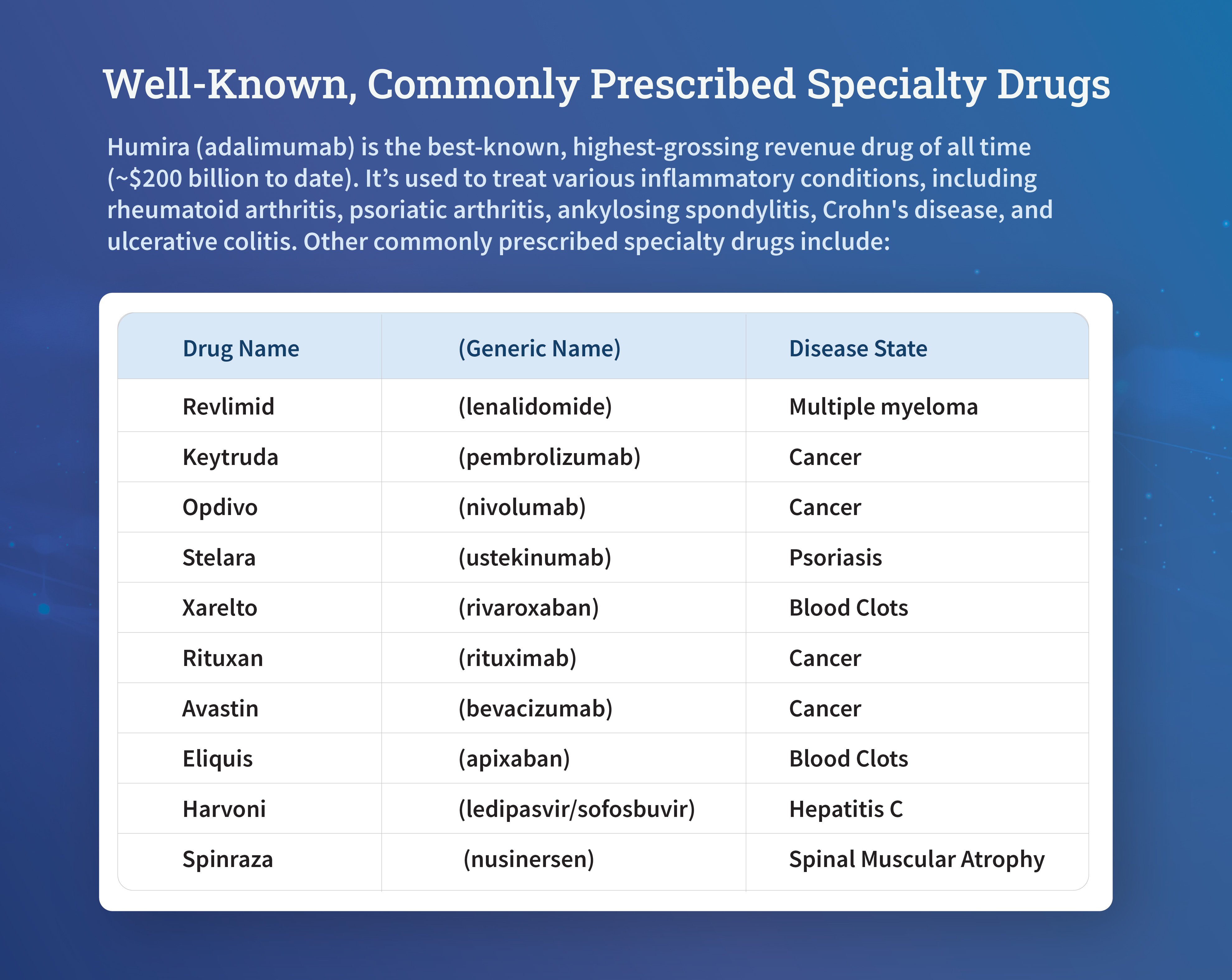
Beyond their manufacturing, speciality drugs are primarily distinguished by the conditions they treat. They are almost exclusively prescribed for serious, chronic, rare, or life-threatening diseases. These conditions often lack readily available or effective conventional treatments, making speciality drugs vital, albeit expensive, options. Examples of diseases commonly treated by speciality drugs include:
- Autoimmune diseases: Conditions like rheumatoid arthritis, Crohn’s disease, multiple sclerosis, and psoriasis, where the immune system mistakenly attacks the body’s own tissues.
- Rare genetic disorders: Diseases such as cystic fibrosis, hemophilia, and Gaucher disease, which affect a small percentage of the population and often require highly specific interventions.
- Cancer: Many advanced or complex forms of cancer are treated with speciality drugs, including targeted therapies and immunotherapies that precisely attack cancer cells or stimulate the body’s own immune response.
- Hepatitis C: While new oral antiviral therapies have become more common, the initial treatments for Hepatitis C were considered speciality drugs due to their complex regimens and significant cost.
- HIV/AIDS: Advanced antiretroviral therapies are often classified as speciality drugs.
- Infertility: Certain fertility treatments fall under the speciality drug umbrella.
- Growth hormone deficiencies: Treatments for these conditions are also often categorized as speciality.
The precise definition can vary slightly among different entities, such as pharmacy benefit managers (PBMs), insurance companies, and regulatory bodies. However, the common thread remains the unique therapeutic need, complex nature, and high cost associated with these medications.
The Manufacturing Complexity
The biological nature of many speciality drugs necessitates sophisticated manufacturing processes. Unlike small-molecule drugs synthesized through chemical reactions, speciality drugs are often biologics. These include:
- Monoclonal Antibodies (mAbs): These are laboratory-produced molecules that mimic the immune system’s ability to fight off harmful cells. They are a cornerstone of many modern cancer and autoimmune disease treatments.
- Recombinant Proteins: These are proteins produced in a laboratory using genetically engineered cells. Examples include insulin, growth hormones, and clotting factors.
- Vaccines: While many vaccines are mass-produced and not considered “speciality” in the same vein, certain complex or personalized vaccines, particularly those used in cancer immunotherapy, can fall into this category.
- Cell and Gene Therapies: These are the cutting edge of speciality drugs, involving the modification of a patient’s cells or the introduction of genetic material to treat diseases. These are inherently complex, highly personalized, and astronomically expensive.
These manufacturing processes require sterile environments, highly specialized equipment, and stringent quality control measures. The production yields can be variable, and the shelf life of these complex molecules can be shorter than traditional drugs, further contributing to their cost and handling requirements.
High Cost and Reimbursement Challenges
The most significant and widely discussed characteristic of speciality drugs is their exorbitant cost. A single course of treatment can range from tens of thousands to hundreds of thousands, or even millions, of dollars per year. This high price tag is driven by several factors:

- Research and Development (R&D) Costs: Developing a new drug is an arduous and expensive process. For speciality drugs targeting rare or complex diseases, the patient population is often small, meaning R&D costs must be recouped from a limited number of individuals. The failure rate in drug development is also high, with many promising candidates never making it to market.
- Manufacturing Complexity and Yields: As discussed, the biological manufacturing processes are costly and can have lower yields compared to chemical synthesis.
- Clinical Trial Costs: Conducting rigorous clinical trials to prove efficacy and safety is essential for regulatory approval and involves substantial expenses, especially for niche indications.
- Unmet Medical Needs: Speciality drugs often address conditions with no existing effective treatments, placing a high value on their ability to offer hope and improve quality of life or survival.
These high costs present significant challenges for patients, who may struggle with co-pays and deductibles, and for healthcare systems and insurers who bear the brunt of the expenditure. This has led to various strategies to manage the cost and access of speciality drugs, including:
- Prior Authorization: Insurers often require healthcare providers to obtain prior approval before dispensing a speciality drug, verifying medical necessity and adherence to treatment guidelines.
- Step Therapy: In some cases, patients may be required to try less expensive, established treatments first before a speciality drug will be covered.
- Specialty Pharmacies: Many speciality drugs are dispensed through a limited network of specialty pharmacies. These pharmacies provide additional services beyond dispensing, such as patient education, adherence monitoring, and financial assistance support.
- Patient Assistance Programs (PAPs): Manufacturers often offer PAPs to help eligible patients afford their medications, especially for those who are uninsured or underinsured.
- Negotiation and Rebates: Insurers and PBMs negotiate prices and rebates with manufacturers to reduce the net cost of speciality drugs.
The Role of Speciality Drugs in Modern Medicine
Despite their cost, speciality drugs represent a monumental leap forward in medical treatment. They offer hope and tangible improvements in outcomes for patients suffering from previously intractable diseases.
Advancing Treatment Paradigms
Speciality drugs have revolutionized the treatment of numerous conditions. For instance, in oncology, targeted therapies and immunotherapies have shifted the focus from broad-spectrum chemotherapy to personalized approaches that attack specific molecular pathways or harness the patient’s own immune system. This has led to significantly improved survival rates and reduced side effects for many cancer patients.
Similarly, in autoimmune diseases, biological agents have provided a new arsenal of treatments that can effectively manage inflammation and prevent further tissue damage, allowing patients to maintain a better quality of life and avoid debilitating progression of their illness. For rare genetic disorders, gene and cell therapies are offering the potential for one-time curative treatments, fundamentally altering the prognosis for individuals born with these conditions.
The Future of Pharmaceutical Innovation
The success and demand for speciality drugs have fueled a significant portion of pharmaceutical R&D investment. This focus on complex biologics and advanced therapies is shaping the future of medicine, with ongoing research exploring new applications and novel mechanisms of action. The development of personalized medicine, where treatments are tailored to an individual’s genetic makeup or specific disease characteristics, is intrinsically linked to the advancements in speciality drug development.
Challenges and Ethical Considerations
The profound impact of speciality drugs is undeniable, but their pervasive presence also raises critical ethical and societal questions:
- Access and Equity: The high cost creates a stark divide in access to life-saving treatments. Patients with comprehensive insurance coverage or significant financial resources are far more likely to benefit than those without. This raises concerns about healthcare equity and whether crucial treatments are becoming a luxury.
- Affordability and Sustainability: The escalating cost of speciality drugs poses a significant burden on healthcare budgets. Insurers and governments face increasing pressure to control spending, which can lead to rationing of care or the imposition of stricter access criteria.
- Innovation vs. Affordability Balance: While incentivizing innovation through patent protection and market exclusivity is essential, finding a balance between rewarding R&D and ensuring affordable access to these vital medications remains a significant challenge for policymakers and the industry.
- The “Orphan Drug” Dilemma: For very rare diseases, the patient population is so small that even with high per-patient prices, the overall market may not be large enough to justify the substantial R&D investment without specific incentives, such as those provided by orphan drug legislation.
In conclusion, speciality drugs are a sophisticated and vital component of modern healthcare. They represent the forefront of medical innovation, offering transformative treatments for some of the most challenging diseases. However, their defining characteristics – complexity, targeted application, and high cost – necessitate ongoing dialogue and strategic approaches to ensure that these life-altering therapies are accessible and sustainable for the patients who need them most. As technology advances and our understanding of disease deepens, the realm of speciality drugs is poised to expand, further pushing the boundaries of what is medically possible.