Phosphatases are a critically important class of enzymes that play a fundamental role in cellular regulation across all living organisms. Their primary function is to remove phosphate groups from molecules, a process known as dephosphorylation. This seemingly simple chemical reaction is a cornerstone of cellular signaling, metabolism, and gene expression, making phosphatases indispensable for life. Understanding their diverse roles and mechanisms of action is crucial for deciphering complex biological pathways and for developing targeted therapeutic interventions for a wide range of diseases, including cancer, diabetes, and neurological disorders.
The Catalytic Mechanism and Classification of Phosphatases
The core catalytic activity of a phosphatase involves the hydrolysis of a phosphomonoester bond. This reaction cleaves a phosphate group from a substrate, typically a protein, lipid, or nucleotide, releasing inorganic phosphate (Pi) and a hydroxyl group. The active site of a phosphatase is meticulously designed to bind both the phosphorylated substrate and water, facilitating the nucleophilic attack that breaks the phosphoester linkage. Metal ions, such as magnesium (Mg2+) or manganese (Mn2+), often act as cofactors, coordinating with the phosphate group and stabilizing the transition state, thereby enhancing catalytic efficiency.
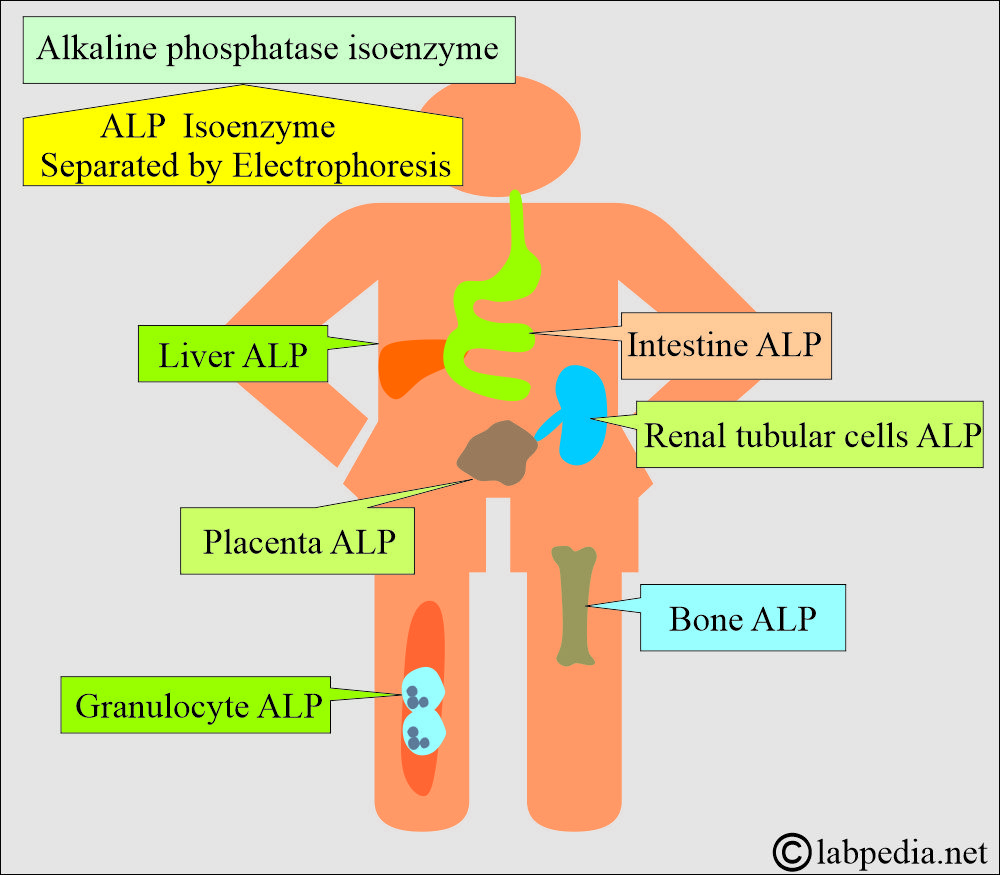
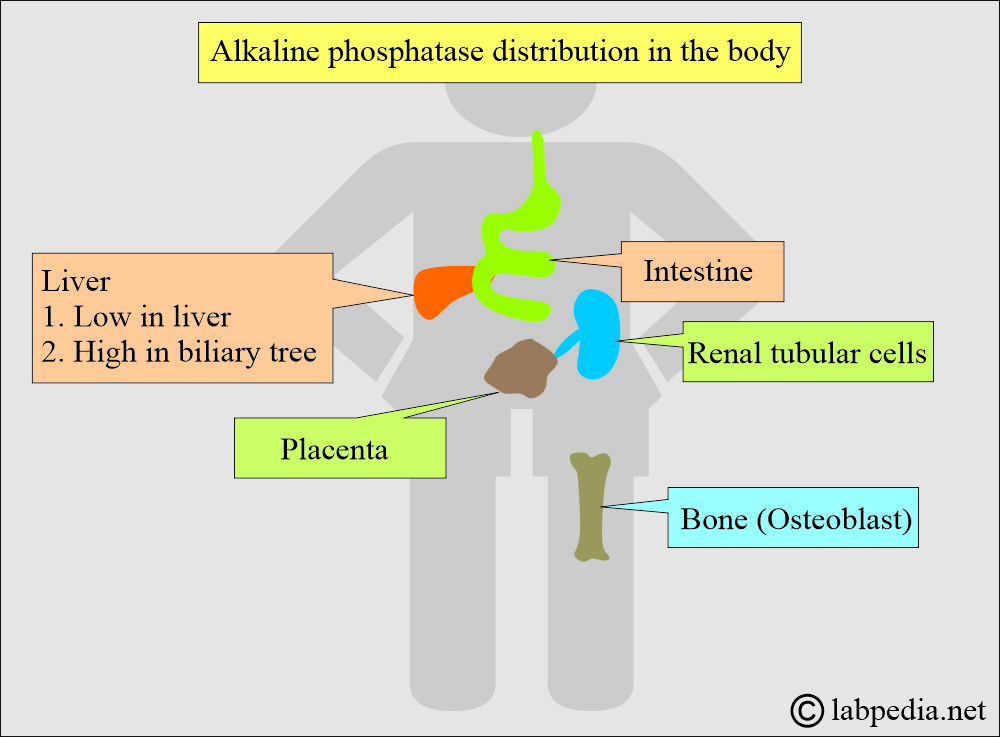
Phosphatases are broadly classified based on the type of substrate they dephosphorylate and the specific amino acid residue in proteins that they target. This classification helps to delineate their diverse functional roles within the cell.
Protein Phosphatases: The Maestros of Phosphorylation Control
Protein phosphorylation and dephosphorylation are reversible processes that act as molecular switches, controlling the activity, localization, and interaction of countless proteins. Protein phosphatases are the counterpoint to protein kinases, the enzymes that add phosphate groups. Together, they maintain a delicate balance in cellular signaling networks.
Serine/Threonine Phosphatases: These enzymes specifically remove phosphate groups from serine or threonine residues on proteins. This is the most abundant class of protein modification, and therefore, serine/threonine phosphatases are involved in an immense array of cellular processes.
-
Type 1 (PP1) and Type 2A (PP2A) Phosphatases: PP1 and PP2A are considered the major “housekeeping” phosphatases, involved in a vast number of cellular functions, including glycogen metabolism, muscle contraction, cell cycle progression, and neurotransmission. PP2A, in particular, is highly abundant and plays a critical role in dephosphorylating a wide range of substrates. They are often regulated by intricate interactions with regulatory subunits, which confer substrate specificity and modulate their activity. For example, PP1 has dozens of different regulatory subunits, each dictating which proteins it will interact with and dephosphorylate.
-
Other Serine/Threonine Phosphatases: This group includes phosphatases like PP2B (calcineurin), which is calcium-dependent and crucial for immune cell activation and neuronal function, and PP3 (calcineurin-like) phosphatases. The regulation and specific roles of these phosphatases are diverse and continue to be areas of active research.
Tyrosine Phosphatases: These enzymes target phosphorylated tyrosine residues on proteins. Protein tyrosine phosphorylation is a critical event in signal transduction pathways that regulate cell growth, differentiation, and survival. Aberrant tyrosine phosphatase activity is strongly implicated in many cancers, as they can oppose the action of oncogenic tyrosine kinases.
-
Protein Tyrosine Phosphatases (PTPs): This large superfamily of enzymes is characterized by a conserved catalytic domain containing a crucial cysteine residue that forms a transient covalent intermediate with the phosphate group during catalysis. PTPs are involved in diverse processes such as immune response, cell adhesion, and neurodevelopment. Their dysregulation can lead to autoimmune diseases and various forms of cancer. For instance, PTP1B is a well-known PTP that plays a significant role in insulin signaling and is a target for diabetes drug development.
-
Dual-Specificity Phosphatases (DSPs): These remarkable enzymes can dephosphorylate serine, threonine, and tyrosine residues. DSPs often act as negative regulators of signaling pathways, rapidly turning off specific signals by dephosphorylating key components. MKP-1 (Mitogen-activated Protein Kinase Phosphatase-1) is a prominent example, playing a crucial role in regulating the activity of MAP kinases, which are central to stress responses and cell proliferation.
Other Types of Phosphatases
Beyond protein phosphatases, other classes of enzymes also possess phosphatase activity, targeting different types of molecules and fulfilling distinct biological roles.
Lipid Phosphatases: These enzymes act on phosphorylated lipids, which are critical components of cell membranes and signaling molecules. For example, PTEN (Phosphatase and TENsin homolog) is a well-known lipid phosphatase that dephosphorylates phosphatidylinositol (3,4,5)-trisphosphate (PIP3), a key second messenger in the PI3K/Akt signaling pathway. PTEN acts as a tumor suppressor, and its inactivation is common in many cancers.
Nucleotide Phosphatases: These phosphatases are involved in the metabolism of nucleotides, the building blocks of DNA and RNA, and also play roles in signaling. For instance, enzymes that hydrolyze cyclic nucleotides like cyclic AMP (cAMP) and cyclic GMP (cGMP) are critical for regulating intracellular second messenger levels.
The Significance of Phosphatase Activity in Cellular Processes
The removal of phosphate groups by phosphatases is not merely a passive deactivation of signaling cascades; it is an active and precisely controlled process that underpins a vast array of cellular functions.
Signal Transduction and Regulation
Phosphorylation events act as a complex language within cells, dictating how signals are transmitted and interpreted. Phosphatases are essential for resetting these signaling pathways, allowing cells to respond to new stimuli and preventing uncontrolled signaling. For example, in response to growth factors, receptor tyrosine kinases become activated and initiate downstream signaling cascades. Tyrosine phosphatases are then required to turn off these signals once the stimulus has subsided, ensuring proper temporal control of cell growth and division. Conversely, dysregulation of phosphatases can lead to persistent signaling, contributing to diseases like cancer.
Cell Cycle Control
The progression through the cell cycle, the series of events leading to cell division, is tightly regulated by cycles of phosphorylation and dephosphorylation. Cyclin-dependent kinases (CDKs) are crucial regulators of the cell cycle, and their activity is controlled by both phosphorylation by other kinases and dephosphorylation by specific phosphatases. For example, the phosphatase Cdc25 plays a critical role in activating CDKs at key checkpoints, ensuring that the cell progresses through different phases of the cell cycle only when appropriate. Errors in phosphatase activity can lead to uncontrolled cell proliferation and the development of tumors.
Metabolism and Energy Homeostasis
Phosphatases are deeply involved in metabolic regulation. For instance, glycogen metabolism, the process of storing and releasing glucose, is controlled by the phosphorylation state of key enzymes. Glycogen phosphorylase and glycogen synthase are activated and inactivated, respectively, by phosphorylation. Phosphatases, particularly PP1, play a crucial role in dephosphorylating and thus activating glycogen synthase and inactivating glycogen phosphorylase, facilitating glucose storage. Similarly, phosphatases are involved in regulating other metabolic pathways, influencing energy production and nutrient utilization.
Gene Expression
While less direct than their roles in signaling, phosphatases can indirectly influence gene expression by regulating transcription factors and chromatin remodeling complexes. Phosphorylation of these proteins can alter their DNA-binding affinity, their ability to interact with other proteins, or their localization within the cell, thereby affecting which genes are turned on or off.
Phosphatases in Disease and Therapeutics
Given their pervasive roles in cellular regulation, it is not surprising that dysregulation of phosphatase activity is implicated in a wide spectrum of human diseases. This makes phosphatases attractive targets for drug development.
Cancer
Many phosphatases, particularly tyrosine phosphatases, act as tumor suppressors by antagonizing the activity of oncogenic kinases. Inactivation or reduced expression of these phosphatases can lead to uncontrolled cell growth, survival, and metastasis. Conversely, some phosphatases can promote cancer by dephosphorylating tumor suppressor proteins or activating oncogenic pathways. Targeting phosphatases in cancer therapy is a complex but promising area, with efforts focused on inhibiting phosphatases that promote tumor growth or restoring the activity of tumor-suppressive phosphatases.
Diabetes and Metabolic Disorders
As mentioned earlier, phosphatases play critical roles in insulin signaling and glucose metabolism. PTP1B, for example, is a negative regulator of insulin signaling, and its inhibition has been explored as a strategy for treating type 2 diabetes. Disruptions in phosphatases involved in lipid metabolism also contribute to metabolic syndrome and obesity.
Neurological Disorders
Phosphatases are essential for proper neuronal function, synaptic plasticity, and neurodevelopment. Calcineurin (PP2B), a calcium-dependent phosphatase, is vital for learning and memory and plays a role in excitotoxicity. Dysregulation of phosphatases has been linked to neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease, as well as psychiatric disorders like depression and schizophrenia.
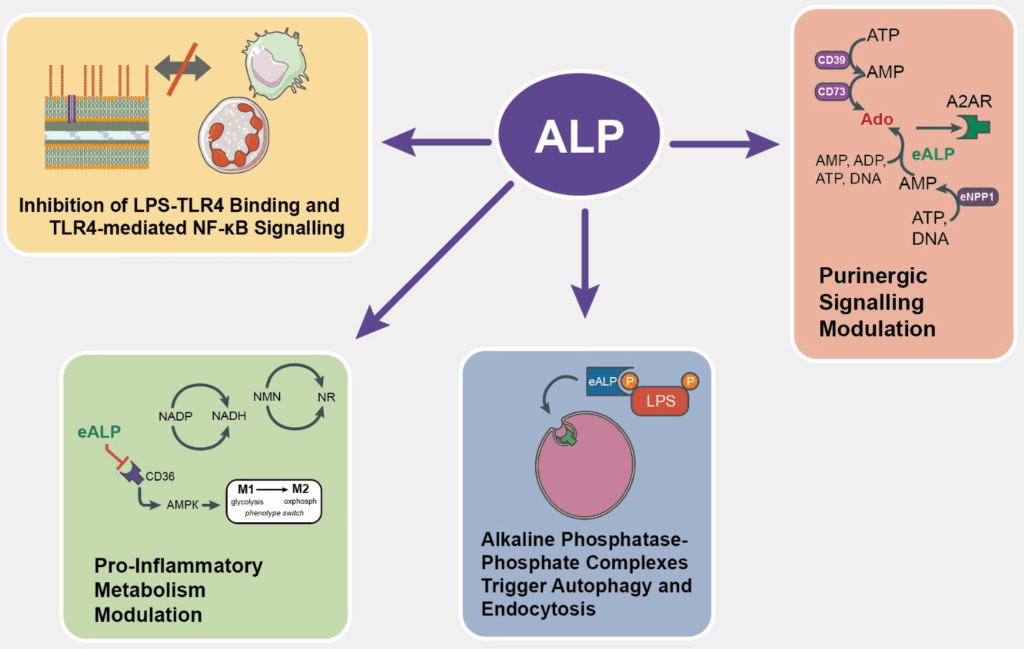
Autoimmune Diseases
The intricate signaling pathways that govern immune cell activation and function are heavily reliant on precise phosphorylation and dephosphorylation events. Phosphatases like calcineurin are critical regulators of T-cell activation, and drugs that inhibit calcineurin are used as immunosuppressants to prevent organ transplant rejection and treat autoimmune diseases. However, the complex roles of phosphatases mean that targeting them requires careful consideration to avoid unintended consequences.
In conclusion, phosphatases are far more than just enzymes that remove phosphate groups. They are indispensable regulators of cellular life, orchestrating complex signaling networks, controlling vital processes like cell division and metabolism, and profoundly impacting human health. Continued research into the diverse world of phosphatases promises to unlock new therapeutic strategies for a multitude of diseases, highlighting their central importance in the landscape of molecular biology and medicine.
