While the term “metathesis reaction” might sound complex and specific to advanced chemistry labs, its underlying principle – the exchange of parts between two compounds – is a fundamental concept with surprising relevance across various scientific and technological fields. Often referred to as a double displacement reaction, metathesis involves the swapping of ions or functional groups between two reacting molecules, resulting in the formation of two new compounds. This elegant rearrangement is not just a cornerstone of synthetic organic chemistry but also finds echoes in areas like materials science, biochemistry, and even, in a conceptual sense, in the evolving landscape of drone technology.
The Core Mechanism of Metathesis
At its heart, a metathesis reaction is driven by the formation of a more stable product. This stability can manifest in several ways, including the precipitation of an insoluble solid, the formation of a weakly ionized species (like water), or the generation of a gas. The general equation for a metathesis reaction involving ionic compounds in solution is:
AX + BY → AY + BX
Here, A and B are cations, while X and Y are anions. In this exchange, A partners with Y, and B partners with X. Understanding these basic principles is crucial before exploring its broader applications.
Types of Metathesis Reactions
Metathesis reactions can be broadly classified based on the driving force that leads to the formation of new products. These classifications help chemists predict and control the outcomes of reactions.
Precipitation Reactions
One of the most common types of metathesis is a precipitation reaction. In this scenario, when two soluble ionic compounds are mixed, they combine to form an insoluble solid, known as a precipitate. The driving force here is the low solubility of the newly formed compound, which causes it to separate from the solution. For example, when solutions of silver nitrate (AgNO₃) and sodium chloride (NaCl) are mixed, a white precipitate of silver chloride (AgCl) forms:
AgNO₃(aq) + NaCl(aq) → AgCl(s) + NaNO₃(aq)
Here, silver ions (Ag⁺) and chloride ions (Cl⁻) combine to form the insoluble AgCl, while sodium ions (Na⁺) and nitrate ions (NO₃⁻) remain in solution as sodium nitrate (NaNO₃).
Acid-Base Neutralization Reactions
Another significant category of metathesis is the acid-base neutralization reaction. This occurs when an acid reacts with a base, typically forming a salt and water. The driving force is the formation of the highly stable, weakly ionized molecule, water. A classic example is the reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH):
HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
In this reaction, the hydrogen ions (H⁺) from the acid combine with the hydroxide ions (OH⁻) from the base to form water. The remaining ions, sodium (Na⁺) and chloride (Cl⁻), form the salt sodium chloride.
Gas-Forming Reactions
Metathesis can also lead to the formation of gases. When the products of a double displacement include a gas, its escape from the solution provides the driving force for the reaction. For instance, the reaction between sodium carbonate (Na₂CO₃) and hydrochloric acid (HCl) produces carbon dioxide gas:
Na₂CO₃(aq) + 2HCl(aq) → 2NaCl(aq) + H₂O(l) + CO₂(g)
Here, the initial reaction forms carbonic acid (H₂CO₃), which is unstable and quickly decomposes into water and carbon dioxide gas. The release of CO₂ drives the reaction forward.
Factors Influencing Metathesis Reactions
Several factors can influence the rate and extent of metathesis reactions. Understanding these factors allows for better control and optimization of synthetic processes.
Solubility Rules
Solubility rules are empirical guidelines that predict whether an ionic compound will dissolve in water. These rules are critical for identifying potential precipitates in metathesis reactions. For example, knowing that most nitrates are soluble, but many silver halides are insoluble, allows chemists to predict the formation of AgCl precipitate when silver nitrate and sodium chloride are mixed.
Stoichiometry and Reaction Conditions
The stoichiometry, or the relative amounts of reactants, is vital for ensuring complete reaction and maximizing product yield. Reaction conditions such as temperature, pressure, and solvent can also play a significant role. For example, increased temperature can sometimes overcome unfavorable solubility issues, while specific solvents can enhance or inhibit the formation of certain products.
Metathesis in Organic Chemistry: A Powerful Synthetic Tool
While the basic definition applies broadly, metathesis reactions have taken on a particularly sophisticated role in modern organic chemistry, especially with the advent of highly efficient catalytic systems. These reactions allow for the precise and selective rearrangement of carbon-carbon double bonds, opening up new pathways for synthesizing complex molecules.
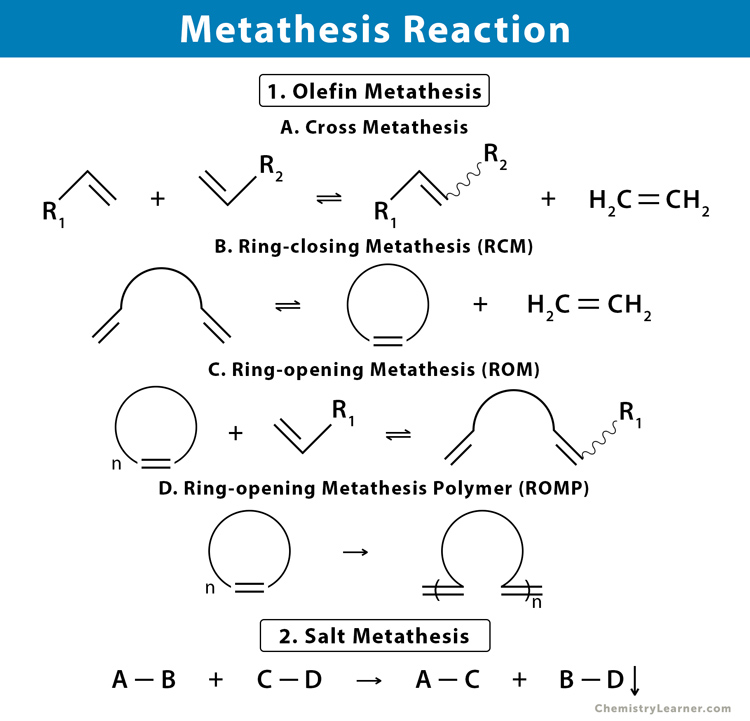
Olefin Metathesis: Revolutionizing Synthesis
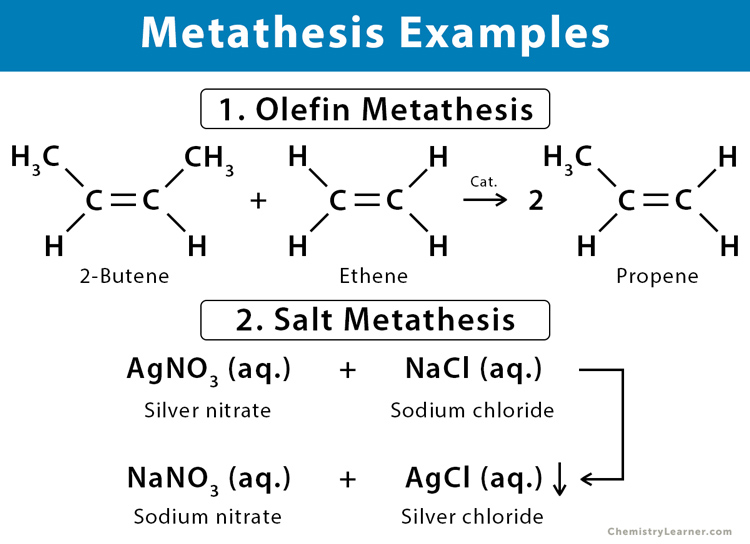
Olefin metathesis, in particular, has been a game-changer. It involves the catalytic redistribution of fragments of alkenes (olefins). The most prominent catalysts for this transformation are based on transition metals, with ruthenium and molybdenum complexes being particularly noteworthy. The general representation of an olefin metathesis reaction is:
R¹-CH=CH-R² + R³-CH=CH-R⁴ ⇌ R¹-CH=CH-R³ + R²-CH=CH-R⁴
This reaction effectively breaks and reforms carbon-carbon double bonds, creating new olefinic structures. The development of well-defined, highly active, and tolerant catalysts has made olefin metathesis an indispensable tool for synthesizing pharmaceuticals, polymers, and fine chemicals.
Grubbs and Schrock Catalysts: The Pioneers
The Nobel Prize in Chemistry in 2005 was awarded to Yves Chauvin, Robert H. Grubbs, and Richard R. Schrock for their contributions to the development of the metathesis method in organic and organometallic chemistry. Grubbs catalysts, often ruthenium-based, are known for their robustness and tolerance to a wide range of functional groups. Schrock catalysts, typically molybdenum or tungsten-based, are highly reactive and efficient for specific applications. These catalysts have dramatically expanded the scope and feasibility of olefin metathesis.
Types of Olefin Metathesis Reactions
Olefin metathesis encompasses several distinct reaction types, each with its specific applications:
Ring-Closing Metathesis (RCM)
RCM is used to form cyclic molecules from acyclic dienes. This is particularly useful for synthesizing cyclic peptides, macrocycles, and heterocyclic compounds, which are common in natural products and pharmaceuticals.
Ring-Opening Metathesis Polymerization (ROMP)
ROMP is a method for producing polymers with unique properties from cyclic olefins. This technique allows for the synthesis of high-performance polymers with controlled microstructures and functionalities.
Cross Metathesis (CM)
CM involves the reaction between two different alkenes, leading to the redistribution of alkylidene fragments. This reaction is valuable for creating unsymmetrical olefins that might be difficult to synthesize through other methods.
Metathesis in Polymer Chemistry
Beyond olefin metathesis, other forms of metathesis are crucial in polymer synthesis. For example, acyclic diene metathesis polymerization (ADMET) is a process that produces polymers by rearranging acyclic dienes. This method allows for the synthesis of unsaturated polymers with potential applications in advanced materials.
Metathesis in Other Scientific Domains
The concept of exchanging components to form new entities extends beyond traditional chemistry, influencing our understanding of processes in other scientific fields.
Biochemical Metathesis: Enzyme Catalysis
In biochemistry, many enzymatic reactions can be viewed through the lens of metathesis. Enzymes are biological catalysts that facilitate specific chemical transformations within living organisms. For instance, isomerases are a class of enzymes that catalyze the rearrangement of atoms within a molecule, effectively performing an internal metathesis. Similarly, transaminases catalyze the transfer of an amino group from one molecule to another, a clear example of group exchange. These biochemical metathesis reactions are fundamental to metabolism and cellular function.
Materials Science: Designing Novel Structures
The principles of metathesis are also leveraged in materials science for the design and synthesis of novel materials. For example, in the development of advanced ceramics and composites, reactions involving the exchange of ions or molecular fragments can lead to materials with tailored properties, such as enhanced strength, conductivity, or thermal resistance.
Conceptual Parallels in Technology: The “Metathesis” of Drone Systems
While not a direct chemical reaction, the concept of “metathesis” can be applied conceptually to the evolution of complex technological systems, such as those found in drone technology. Consider the development of autonomous flight systems. Initially, drones were largely controlled remotely, with human pilots dictating every maneuver. This was akin to a simple, direct “A controls B” system.
However, as AI and sophisticated sensors have advanced, drones have begun to exhibit autonomous capabilities. An AI algorithm, acting as a new “partner,” now dictates flight paths, navigates obstacles, and even performs complex aerial maneuvers based on programmed objectives and real-time environmental data. This can be conceptually viewed as a form of “metathesis” within the drone’s operational framework. The human pilot (one component) has been partially “displaced” by the AI system (another component), leading to a new operational paradigm.
The Exchange of Control and Data
In this technological analogy, the “reactants” are the drone’s hardware (motors, sensors, flight controller), the environmental conditions, and the mission objectives. The “products” are the executed flight path, the collected data, and the successful completion of the task. The “metathesis” occurs when the locus of control and decision-making shifts. For example, in “AI Follow Mode,” the drone (one entity) exchanges its direct manual control for an autonomous tracking algorithm (the other entity) that dynamically adjusts its position and speed relative to a target.
Innovation Through Component Swapping
Similarly, advancements in sensor technology and processing power have led to the “metathesis” of obstacle avoidance systems. Older systems relied on simpler proximity sensors. Modern systems, incorporating computer vision and lidar, represent a more sophisticated “exchange” of sensing methodologies, leading to vastly improved situational awareness and safer flight. This constant “swapping out” of components and functionalities, driven by innovation, is the technological equivalent of a metathesis reaction, leading to more capable and versatile drone platforms.

Conclusion: A Ubiquitous Principle of Transformation
The metathesis reaction, in its myriad chemical forms, is a fundamental process that underpins countless synthetic pathways and natural phenomena. From the precipitation of salts to the intricate choreography of enzyme activity, the simple act of exchanging partners leads to the creation of new substances and the progression of chemical transformations. Its advanced application in olefin metathesis has revolutionized the synthesis of complex organic molecules, while its conceptual echoes in technological evolution highlight the pervasive nature of change and adaptation. Understanding metathesis, therefore, offers a window into the fundamental principles of how matter rearranges and transforms, a concept that resonates from the smallest molecular interactions to the grandest technological advancements.
