A flow cytometer is a sophisticated laboratory instrument that analyzes individual cells or particles as they flow in a liquid stream. This powerful technology enables scientists to simultaneously measure multiple physical and chemical characteristics of thousands, even millions, of cells per second. By precisely interrogating each cell, flow cytometry provides a quantitative understanding of cell populations, their properties, and their functions. At its core, the instrument combines fluidics, optics, and electronics to achieve its analytical capabilities.
The Core Components and Principles of Flow Cytometry
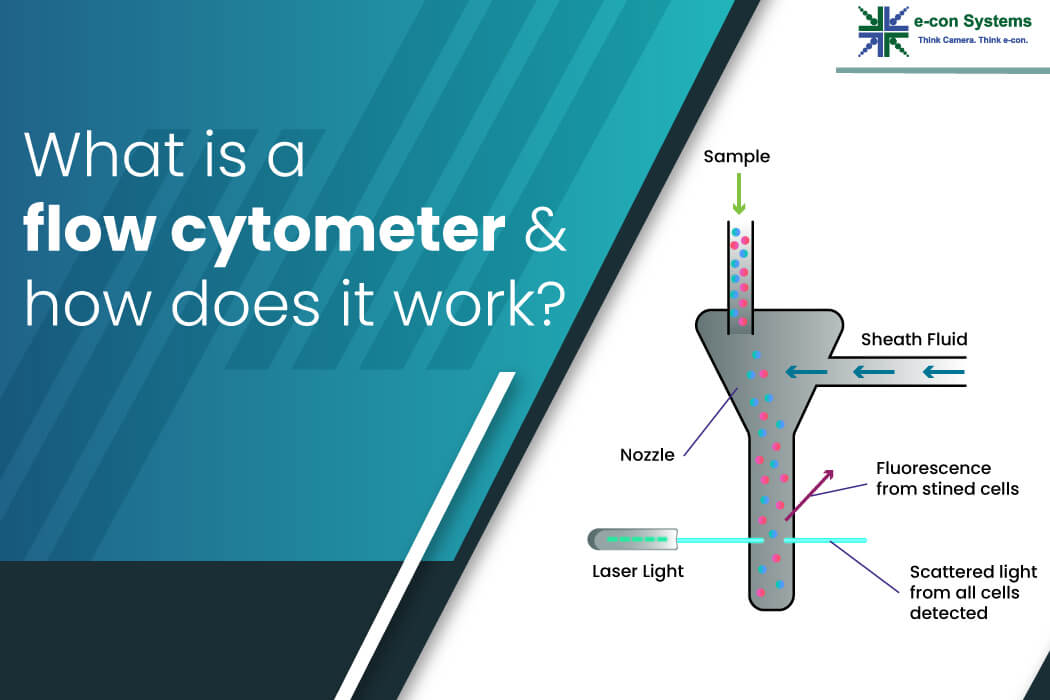
The fundamental operation of a flow cytometer relies on a meticulously orchestrated process involving several key components. The journey begins with sample preparation, where cells are suspended in a fluid medium. This suspension is then introduced into the instrument’s fluidics system, which is designed to deliver a precise and continuous stream of cells.
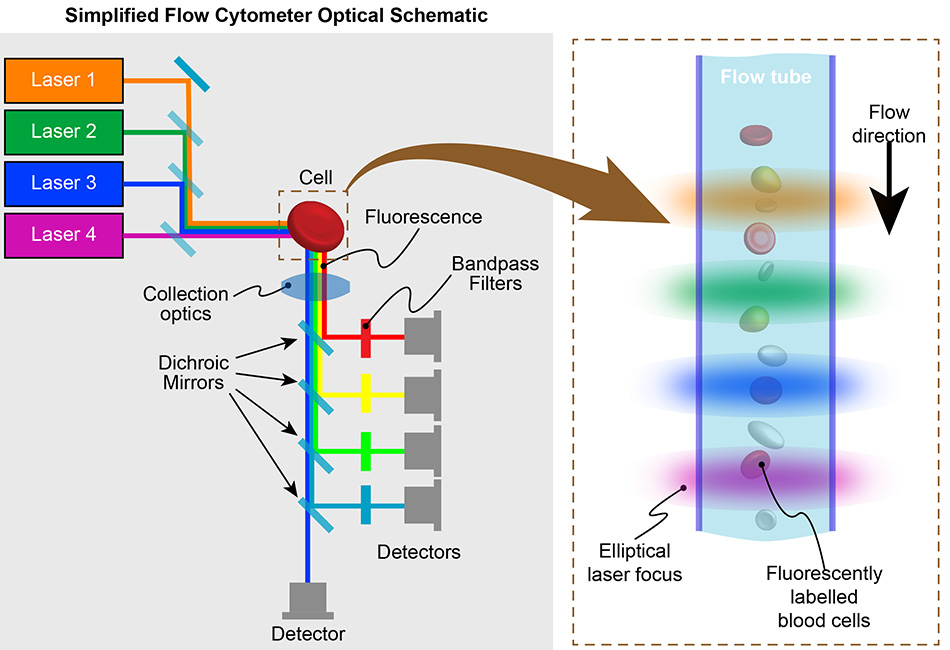
Fluidics System: Sheath Flow and Cell Stream
The heart of the fluidics system is the sheath flow mechanism. A high-pressure stream of a specialized buffer solution, known as the sheath fluid, surrounds the sample stream. This sheath fluid hydrodynamically focuses the sample, forcing the cells into a single file line as they enter the interrogation point. This precise alignment is critical for ensuring that each cell is analyzed individually, preventing doublets or clumps from skewing the data. The sheath fluid also helps to maintain a stable flow rate and pressure, contributing to the instrument’s reproducibility. The sample is introduced through a smaller diameter tube within the sheath flow, ensuring that the cells are confined to the center of the stream.
Optics System: Excitation and Detection
As the single-file stream of cells passes through the interrogation point, it encounters one or more lasers. These lasers act as excitation sources, illuminating the cells. The wavelengths of the lasers are carefully chosen to excite fluorescent molecules, either naturally present within the cells or introduced as fluorescent labels (fluorochromes) attached to specific cellular components. When a fluorochrome absorbs the laser light, it is excited to a higher energy state. As it returns to its ground state, it emits light at a longer wavelength. This emitted fluorescence, along with scattered light from the cell, is collected by the optics system.
The optics system is a complex arrangement of lenses, mirrors, and filters. Lenses focus the laser light onto the cells and collect the emitted light. Dichroic mirrors and emission filters are crucial for separating the different wavelengths of light. Dichroic mirrors reflect light of certain wavelengths while allowing others to pass through, enabling the separation of fluorescence from different fluorochromes. Emission filters are designed to block unwanted wavelengths and allow only the specific fluorescence emission of a particular fluorochrome to reach its designated detector.
Detectors: Quantifying Cell Properties
The separated light signals are then directed to an array of detectors. Photomultiplier tubes (PMTs) and photodiodes are the most common types of detectors used in flow cytometry. These detectors convert the light signals into electrical pulses. The intensity of each electrical pulse is proportional to the intensity of the light signal received.
Two primary types of light scattering are measured: forward scatter (FSC) and side scatter (SSC). Forward scatter is related to the size of the cell, as light is scattered in a forward direction when it encounters a particle. Side scatter, on the other hand, is related to the internal complexity or granularity of the cell. Cells with more internal structures will scatter more light at a 90-degree angle.
In addition to scattered light, the fluorescence emitted by the fluorochromes is also detected. Each detector channel in a flow cytometer is typically associated with a specific fluorochrome or light scattering measurement. The intensity of the fluorescence detected in each channel provides information about the abundance of specific molecules within or on the surface of the cell.
Electronics and Software: Data Acquisition and Analysis
The electrical pulses generated by the detectors are sent to an electronics system that amplifies, digitizes, and records the data. This data acquisition system translates the physical and chemical properties of each individual cell into a set of numerical values, often referred to as a “parameter” for each cell. For a sample containing thousands of cells, the instrument will generate millions of data points.
The raw data collected by the flow cytometer is then processed and analyzed using specialized software. This software allows researchers to visualize the data, typically in the form of scatter plots or histograms. Scatter plots, also known as dot plots, display the relationship between two parameters for individual cells. Histograms display the distribution of a single parameter across a population of cells.
Through a process called gating, researchers can identify and isolate specific cell populations of interest within the larger sample. For example, a gate can be drawn around a population of cells that exhibit a particular combination of forward and side scatter, indicating a specific cell type. Further analysis can then be performed on these gated populations, allowing for the quantification of cell numbers, the measurement of protein expression levels, or the assessment of other cellular characteristics.
Applications of Flow Cytometry in Science and Medicine
The versatility and quantitative power of flow cytometry have made it an indispensable tool across a vast spectrum of scientific disciplines and clinical applications. Its ability to analyze individual cells in high throughput has revolutionized research in immunology, cancer biology, infectious diseases, and beyond.
Immunology: Characterizing Immune Cells
One of the most prominent applications of flow cytometry is in immunology. Immune cells, such as lymphocytes (T cells, B cells, NK cells), monocytes, and granulocytes, express a diverse array of surface and intracellular proteins that define their lineage, activation state, and function. Flow cytometry, particularly when employing multicolor fluorescence (using multiple fluorochromes simultaneously), allows for the precise identification and enumeration of these different immune cell subsets.
Researchers can use flow cytometry to:
- Quantify immune cell populations: Determine the relative and absolute numbers of different immune cells in blood, tissue, or other biological samples. This is crucial for monitoring immune status in health and disease.
- Assess immune cell activation: Detect the expression of activation markers on immune cells, providing insights into their responsiveness to stimuli.
- Analyze immune cell subsets: Differentiate between various subtypes of T cells (e.g., helper T cells, cytotoxic T cells, regulatory T cells) based on the expression of specific cell surface markers like CD4 and CD8.
- Study immune responses to pathogens and vaccines: Monitor changes in immune cell populations and functions following infection or vaccination.
Cancer Biology: Diagnosis, Prognosis, and Treatment Monitoring
Flow cytometry plays a critical role in the diagnosis, prognosis, and monitoring of various cancers, particularly hematological malignancies like leukemia and lymphoma. Certain cancers are characterized by the aberrant expression of specific cell surface antigens. By analyzing these antigens on cells obtained from bone marrow or blood, clinicians can:
- Diagnose and classify leukemia and lymphoma: Identify and characterize cancerous cells based on their unique immunophenotype. This helps in determining the specific type and subtype of cancer, which is essential for appropriate treatment.
- Monitor treatment response: Track the reduction of cancer cells in the patient’s body during and after therapy. A decrease in the abnormal cell population is indicative of treatment efficacy.
- Detect minimal residual disease (MRD): Identify very small numbers of remaining cancer cells after treatment, which can be a predictor of relapse. Flow cytometry’s sensitivity allows for the detection of cancer cells at much lower levels than conventional methods.
- Assess prognosis: Certain immunophenotypic features can be associated with different patient outcomes, aiding in risk stratification and treatment planning.
Infectious Diseases: Pathogen Detection and Host Response
Flow cytometry is also instrumental in studying infectious diseases. It can be used to:
- Detect and quantify infected cells: Identify cells that are infected by viruses or bacteria by looking for pathogen-specific antigens or by observing changes in host cell markers.
- Analyze the host immune response to infection: Monitor the activation and proliferation of immune cells that are fighting the infection. This can involve tracking the changes in specific immune cell subsets or assessing the production of cytokines by these cells.
- Assess the efficacy of antimicrobial therapies: Evaluate how treatments affect pathogen burden and the host’s immune response.
Other Applications
Beyond these major areas, flow cytometry finds applications in:
- Cell sorting: More advanced flow cytometers are equipped with cell sorters. This capability allows for the physical separation of specific cell populations from a mixed sample based on their measured properties. Sorted cells can then be used for downstream applications like cell culture, genomic analysis, or further experimentation.
- Stem cell research: Characterizing and isolating different types of stem cells based on their unique surface markers.
- Drug discovery and development: Screening for the effects of new drug candidates on cell viability, proliferation, and other cellular functions.
- Environmental monitoring: Analyzing microbial populations in water or soil samples.
- Forensic science: Identifying and characterizing biological materials.
Advances and Future Directions in Flow Cytometry
The field of flow cytometry is continuously evolving, with ongoing technological advancements pushing the boundaries of its capabilities. These innovations aim to increase sensitivity, expand multiplexing capacity, improve automation, and integrate with other analytical platforms.
Spectral Flow Cytometry: Overcoming Fluorescence Overlap
One of the most significant recent advancements is spectral flow cytometry. Traditional flow cytometers use optical filters to separate fluorescence emissions, which can lead to spectral overlap between fluorochromes, making it challenging to distinguish between them, especially in multicolor experiments. Spectral flow cytometry, in contrast, measures the entire emission spectrum of each fluorochrome. This allows for the deconvolution of complex fluorescence signals, enabling researchers to use a much larger number of fluorochromes simultaneously (often 40 or more) with greater specificity. This increased multiplexing capacity provides a more comprehensive view of cellular phenotypes and functions.
High-Throughput and Automation
The demand for higher throughput and increased automation in flow cytometry is driven by the need to analyze large sample volumes, particularly in clinical settings and large-scale research projects. Advances in robotics, liquid handling systems, and software have led to highly automated platforms that can process samples with minimal manual intervention. This not only increases efficiency but also improves reproducibility and reduces the risk of human error.
Imaging Flow Cytometry: Combining Flow and Microscopy
Imaging flow cytometry represents a powerful fusion of flow cytometry and microscopy. In addition to collecting fluorescence and scatter data for each cell, imaging flow cytometers capture images of individual cells as they pass through the interrogation point. This provides a wealth of morphological information that is not available with traditional flow cytometry. Researchers can analyze cell shape, texture, internal structures, and subcellular localization of fluorescent signals, adding a new dimension to cellular analysis.
Integration with Other Technologies
The integration of flow cytometry with other omics technologies, such as genomics, transcriptomics, and proteomics, is opening up new avenues for discovery. By combining flow cytometric phenotyping with molecular profiling of sorted cell populations, researchers can gain a deeper understanding of the complex biological processes that govern cell behavior and disease. Single-cell multi-omics approaches, which can analyze multiple molecular layers from individual cells, are increasingly being powered by flow cytometry’s ability to isolate specific cell types.

Artificial Intelligence and Machine Learning
The vast amounts of data generated by modern flow cytometers present challenges for manual analysis. Artificial intelligence (AI) and machine learning algorithms are being developed to automate and enhance data analysis. These tools can help identify complex cell populations, detect subtle patterns that might be missed by human operators, and predict outcomes based on cellular characteristics. AI is also being applied to optimize experimental design and instrument performance.
The future of flow cytometry is bright, promising even greater insights into cell biology, disease mechanisms, and therapeutic interventions. As technology continues to advance, flow cytometry will undoubtedly remain at the forefront of biological research and clinical diagnostics.
