In the realm of chemistry, a “balanced equation” is a fundamental concept that underpins our understanding of chemical reactions. It’s more than just a representation of reactants transforming into products; it’s a precise statement of the Law of Conservation of Mass, asserting that in any closed system, matter is neither created nor destroyed during a chemical reaction. This principle dictates that the number and type of atoms present before a reaction must precisely equal the number and type of atoms present after the reaction. Therefore, a balanced chemical equation serves as a quantitative blueprint, revealing the exact molar ratios in which substances react and are formed.

The importance of this principle cannot be overstated. Without balanced equations, it would be impossible to predict the amount of product that can be formed from a given amount of reactant, a crucial aspect of chemical synthesis, industrial production, and even understanding biological processes. It allows chemists to perform stoichiometric calculations, enabling them to determine theoretical yields, identify limiting reactants, and optimize reaction conditions for efficiency and safety. In essence, a balanced equation is the silent, yet indispensable, language of chemical change, providing clarity and predictability in a world of molecular transformations.
The Foundation: Understanding Chemical Equations
Before delving into the specifics of balancing, it’s crucial to grasp the basic components of a chemical equation. A chemical equation is a symbolic representation of a chemical reaction, using chemical formulas to denote the reactants and products involved. Reactants are the substances that initiate the reaction and are typically written on the left side of the equation, separated by a plus sign if there are multiple reactants. Products are the new substances formed as a result of the reaction and are written on the right side, also separated by plus signs if there are multiple products. A single arrow (→) separates the reactants from the products, indicating the direction of the reaction.
Atomic Representation and Formulas
At the heart of every chemical equation are the atoms themselves. Chemical formulas, such as H₂O for water or CO₂ for carbon dioxide, represent the specific types and numbers of atoms that constitute a molecule or compound. The subscript numbers in these formulas indicate the number of atoms of each element within a single molecule. For example, in H₂O, there are two hydrogen atoms and one oxygen atom. Understanding these formulas is paramount, as they are the building blocks upon which the entire balancing process is based.
The Law of Conservation of Mass: The Guiding Principle
The Law of Conservation of Mass, first articulated by Antoine Lavoisier, is the bedrock upon which the concept of balanced equations rests. This law states that in any chemical reaction, the total mass of the reactants must equal the total mass of the products. This means that the number of atoms of each element must remain constant throughout the reaction. Atoms are not magically created or destroyed; they are merely rearranged to form new substances. Therefore, a balanced chemical equation is a direct reflection of this fundamental law, ensuring that the atomic inventory is the same on both sides of the reaction arrow.
The Process of Balancing Chemical Equations
Balancing a chemical equation involves adjusting the stoichiometric coefficients, which are numbers placed in front of the chemical formulas. These coefficients represent the relative number of moles (or molecules) of each substance involved in the reaction. The goal is to ensure that the number of atoms of each element is identical on both the reactant and product sides of the equation. This is an iterative process that requires careful attention and systematic application.
Step-by-Step Balancing: A Systematic Approach
The process of balancing typically begins with identifying the elements present in the equation and counting the number of atoms of each element on both sides. A common strategy is to start by balancing elements that appear in only one reactant and one product. Often, oxygen and hydrogen are balanced last, as they tend to appear in multiple compounds. Coefficients are then added incrementally, and the atom counts are re-evaluated after each adjustment. If an element is unbalanced, its coefficient is modified. This process continues until all elements are balanced, meaning the atom count for each element is the same on both sides of the arrow. It’s important to remember that the chemical formulas themselves cannot be altered; only the coefficients can be changed.
Common Pitfalls and Strategies for Overcoming Them
One common pitfall is miscounting atoms, especially in complex molecules or when dealing with polyatomic ions that remain intact during the reaction. It is often helpful to treat polyatomic ions like sulfate (SO₄²⁻) or nitrate (NO₃⁻) as single units if they appear on both sides of the equation without undergoing a chemical change. Another challenge can arise from encountering fractional coefficients. While technically correct, chemical equations are conventionally balanced with the smallest whole number coefficients. Fractional coefficients can be eliminated by multiplying the entire equation by the denominator of the fraction. Patience and practice are key; as one becomes more familiar with balancing, the process becomes more intuitive.
The Significance and Application of Balanced Equations
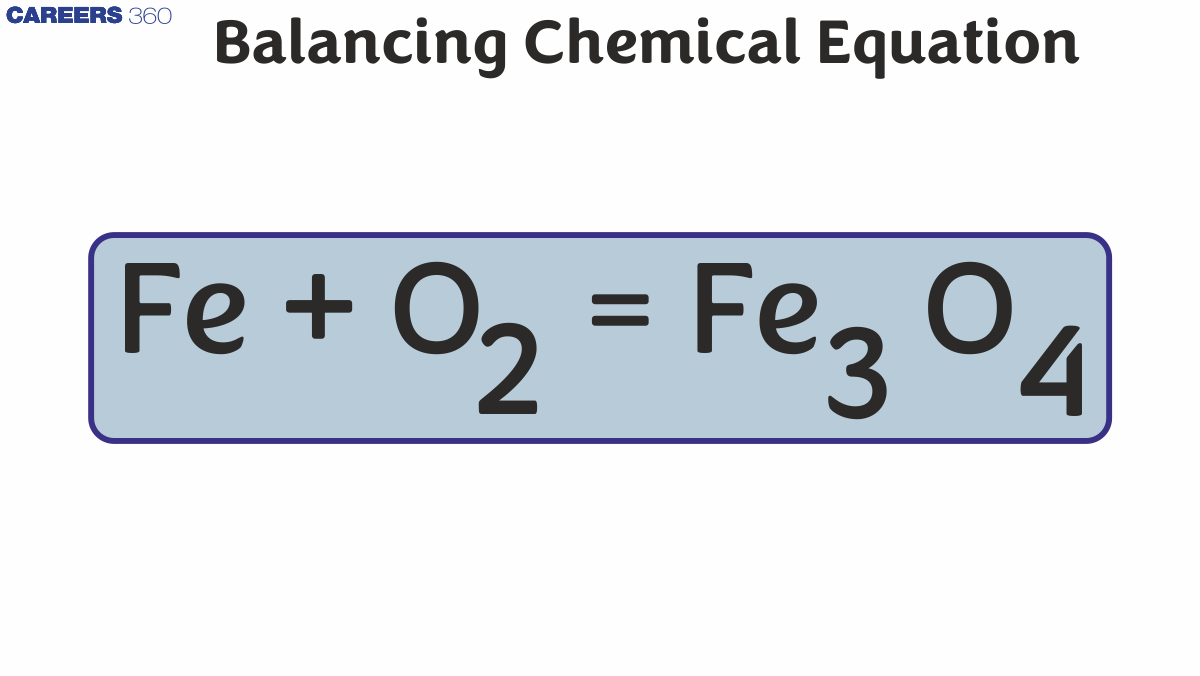
The ability to balance chemical equations unlocks a world of quantitative chemical understanding. It transforms a qualitative description of a reaction into a precise, numerical relationship between the reacting species. This quantitative aspect is the cornerstone of many critical applications in chemistry and related fields.
Stoichiometry: The Quantitative Heart of Chemistry
Stoichiometry, derived from Greek words meaning “measure” and “to measure,” is the branch of chemistry that deals with the quantitative relationships between reactants and products in a chemical reaction. Balanced equations provide the necessary coefficients to perform stoichiometric calculations. For instance, if we have a balanced equation, we can calculate the theoretical yield of a product given a specific amount of reactant, or conversely, determine the amount of reactant needed to produce a desired quantity of product. This is indispensable for chemical synthesis, where precise control over reactant quantities is essential for maximizing product formation and minimizing waste.
Industrial Chemistry and Large-Scale Production
In industrial settings, balanced equations are vital for designing and operating chemical processes efficiently and economically. Chemical engineers use them to determine the optimal amounts of raw materials needed, calculate the energy requirements of a reaction, and predict the volume of byproducts. This allows for the cost-effective production of vast quantities of chemicals, from pharmaceuticals and fertilizers to plastics and fuels. Without accurate stoichiometric calculations based on balanced equations, large-scale chemical manufacturing would be impractical and fraught with inefficiency.
Environmental Science and Analysis
The principles of balanced equations are also critical in environmental science. Understanding how pollutants react in the atmosphere or water requires knowledge of the stoichiometry of those reactions. For example, balancing the equation for the reaction of sulfur dioxide with oxygen and water to form sulfuric acid helps us quantify the amount of acid rain that can be produced. Furthermore, in analytical chemistry, balanced equations are used to determine the concentration of unknown substances through titrations and other quantitative analytical techniques, ensuring the accuracy and reliability of environmental monitoring and assessment.
Types of Chemical Reactions and Their Balancing
While the fundamental principle of balancing remains the same across all chemical reactions, the complexity of balancing can vary depending on the type of reaction. Recognizing the type of reaction can sometimes offer a shortcut or a more intuitive approach to balancing.
Synthesis and Decomposition Reactions
Synthesis reactions involve the combination of two or more simpler substances to form a more complex one (e.g., A + B → AB). Decomposition reactions are the reverse, where a complex substance breaks down into simpler components (e.g., AB → A + B). Balancing these often involves straightforward adjustments of coefficients to match the atoms in the combined or separated entities. For instance, the formation of water from hydrogen and oxygen (2H₂ + O₂ → 2H₂O) is a synthesis reaction that is balanced by ensuring two oxygen atoms and four hydrogen atoms are present on both sides.
Single and Double Displacement Reactions
Single displacement reactions occur when one element replaces another in a compound (e.g., A + BC → AC + B). Double displacement reactions involve the exchange of ions between two compounds (e.g., AB + CD → AD + CB). For these, it’s important to correctly identify the ions or elements being exchanged and then apply the standard balancing procedures. Paying attention to the charges of ions is crucial for correctly writing the formulas of products in displacement reactions.

Combustion Reactions
Combustion reactions, particularly those involving hydrocarbons (compounds containing carbon and hydrogen), are a common type encountered. These typically involve the reaction of a substance with oxygen to produce heat and light, often forming carbon dioxide and water. A general strategy for balancing hydrocarbon combustion is to first balance carbon atoms, then hydrogen atoms, and finally oxygen atoms, as oxygen is usually present as a diatomic molecule (O₂).
By mastering the art of balancing chemical equations, we gain a powerful tool for understanding, predicting, and controlling the myriad chemical transformations that shape our world, from the smallest laboratory experiment to the grandest industrial processes. It is a foundational skill that unlocks a deeper appreciation for the quantitative elegance of chemistry.
