The acronym “SI” is a ubiquitous and foundational element within the scientific landscape. To the uninitiated, its repeated appearance can be a source of mild confusion, while for those well-versed in scientific disciplines, it represents a fundamental framework for measurement and understanding. In essence, SI stands for the Système International d’Unités, which translates from French to the International System of Units. This globally recognized standard provides a unified and coherent system of measurement that underpins scientific research, technological development, industrial production, and international trade. Its establishment was a direct response to the growing need for consistency and comparability across different nations and scientific fields, moving away from a patchwork of disparate and often incompatible measurement systems.
![]()
The development and adoption of the SI system have been a gradual but crucial process, driven by the inherent limitations of older, non-standardized units. Before the SI, scientists and engineers often relied on a variety of systems, such as the imperial system (with its inches, pounds, and Fahrenheit) or various metric-based systems that, while metric, lacked a fully unified structure. This led to significant challenges in communication, replication of experiments, and the seamless transfer of knowledge and technology across borders. The consequences of miscommunication due to differing units could range from minor inconveniences to catastrophic failures, particularly in fields like engineering and aerospace. The SI system, therefore, represents a triumph of scientific collaboration and a testament to the power of standardization in fostering progress.
The Genesis and Evolution of the SI System
The need for a universal system of measurement became increasingly apparent with the rise of industrialization and the expansion of international scientific exchange in the 18th and 19th centuries. While the metric system, originating in France during the French Revolution, laid much of the groundwork, the SI system as we know it today is a more refined and comprehensive evolution of these early efforts.
Early Foundations: The Metric System’s Precursors
The concept of a decimalized, coherent system of weights and measures gained traction in France following the revolution. The initial goal was to replace the chaotic and often localized measurement practices that existed across the country. Key milestones included the establishment of the meter and the kilogram based on natural phenomena, aiming for universality. The original meter was defined as one ten-millionth of the distance from the North Pole to the equator along the meridian passing through Paris. The kilogram was initially defined as the mass of one cubic decimeter of water at its maximum density. These early definitions, while groundbreaking, were later refined for greater accuracy and stability.
The Birth of the SI: A Unified Framework
The modern SI system was formally established by the 13th General Conference on Weights and Measures (CGPM) in 1971. This conference built upon decades of work by the International Bureau of Weights and Measures (BIPM), an international organization established in 1875 under the Metre Convention. The BIPM is responsible for maintaining the international standards of measurement and ensuring their global uniformity. The SI system is composed of seven base units, each representing a fundamental physical quantity. These base units are the building blocks from which all other derived units are constructed. Their careful selection and precise definition are crucial for the integrity and consistency of the entire system.
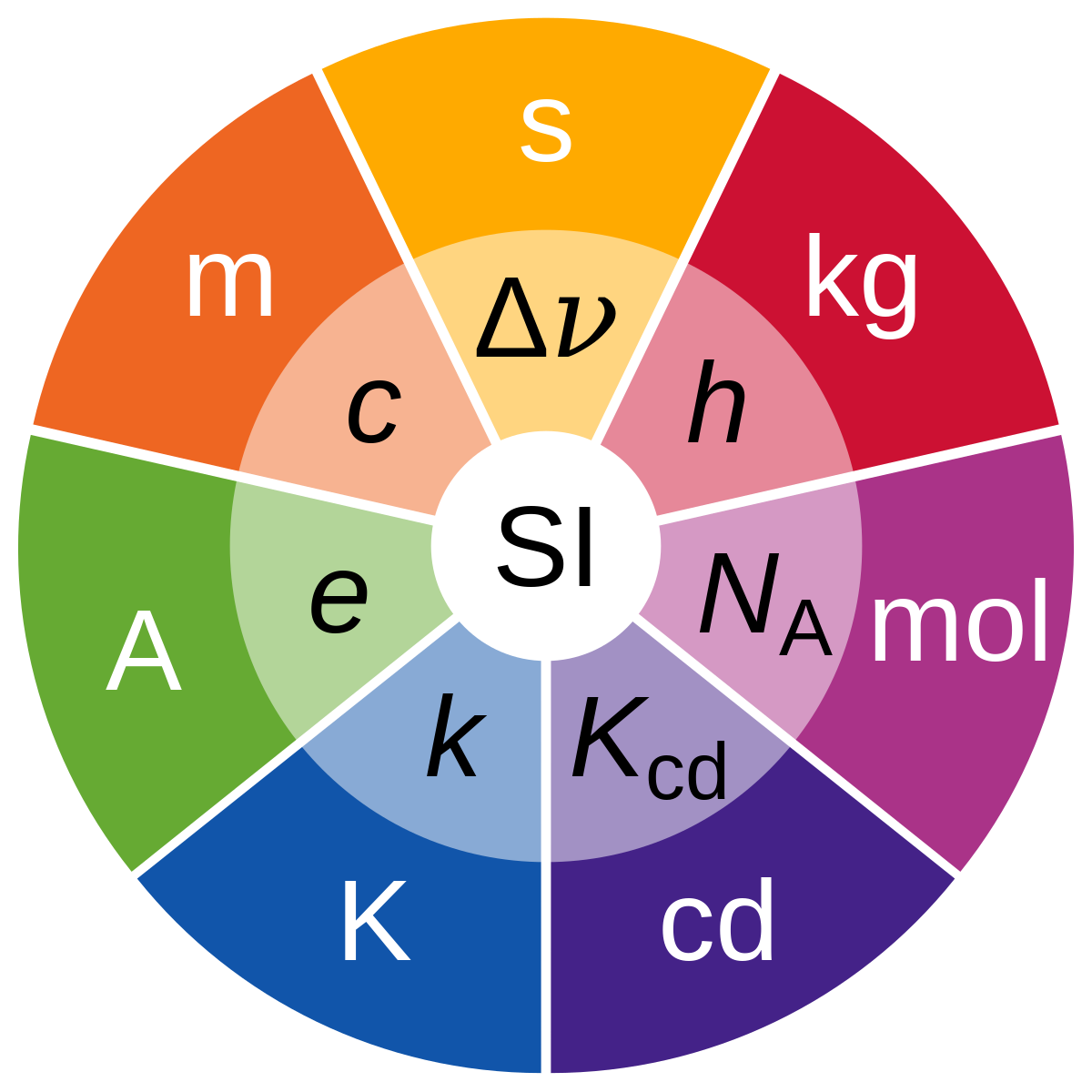
The Seven Pillars of Measurement: SI Base Units
The strength of the SI system lies in its seven fundamental base units. These units are not arbitrarily chosen; they represent the most fundamental physical quantities that can be measured. Each base unit is defined with an extremely high degree of precision and accuracy, ensuring their stability and reproducibility across time and location.
The Kilogram (kg): Mass
The kilogram is the SI base unit of mass. Historically, it was defined by a physical artifact – the International Prototype Kilogram (IPK), a platinum-iridium cylinder kept at the BIPM. However, in 2019, the definition of the kilogram was fundamentally revised. It is now defined based on the Planck constant, a fundamental constant of quantum mechanics. This redefinition ensures that the kilogram’s value is tied to a universal physical constant rather than a physical object that could degrade over time. This change significantly enhances the stability and accessibility of the kilogram’s definition, as it can be realized through experiments that measure the Planck constant.
The Meter (m): Length
The meter is the SI base unit of length. Its definition has also evolved significantly over time. Initially based on Earth’s dimensions, it was later defined by a prototype bar. Today, the meter is defined based on the speed of light in a vacuum. Specifically, it is the length of the path traveled by light in vacuum during a time interval of $1/299,792,458$ of a second. This definition is highly precise and reproducible, relying on fundamental constants of nature and the well-defined duration of the second.
The Second (s): Time
The second is the SI base unit of time. Its definition is based on the cesium atom. One second is defined as the duration of $9,192,631,770$ periods of the radiation corresponding to the transition between the two hyperfine levels of the ground state of the cesium-$133$ atom. This atomic definition provides an incredibly stable and accurate standard for measuring time, essential for everything from GPS systems to fundamental physics research.
The Kelvin (K): Thermodynamic Temperature
The Kelvin is the SI base unit of thermodynamic temperature. It is defined based on the Boltzmann constant. One Kelvin is defined by fixing the numerical value of the Boltzmann constant $k$ to $1.380649 times 10^{-23}$ J/K. This constant provides a direct link between energy and temperature at the microscopic level. This new definition replaced the previous one, which was based on the triple point of water, offering greater accuracy and a more fundamental basis.
The Ampere (A): Electric Current
The Ampere is the SI base unit of electric current. The 2019 redefinition of SI base units also redefined the Ampere. It is now defined by fixing the numerical value of the elementary charge $e$ to $1.602176634 times 10^{-19}$ C, where the coulomb is equal to the ampere-second. This definition directly links the Ampere to the charge of a single electron, providing a more fundamental and stable standard for electric current.
The Mole (mol): Amount of Substance
The mole is the SI base unit of amount of substance. It is defined by fixing the numerical value of the Avogadro constant $N_{text{A}}$ to $6.02214076 times 10^{23}$ mol$^{-1}$. This constant represents the number of elementary entities (such as atoms, molecules, ions, electrons, or other particles) in one mole. This definition ensures that a mole is a fixed number of entities, regardless of the substance.
The Candela (cd): Luminous Intensity
The Candela is the SI base unit of luminous intensity, which measures the power emitted by a light source in a particular direction. It is defined by fixing the numerical value of the luminous efficacy of monochromatic radiation of frequency $540 times 10^{12}$ Hz to $683$ lm/W. This definition directly relates luminous intensity to the physical quantity of radiant flux, providing a more robust and scientifically grounded standard.
Derived Units and Their Significance in Science and Technology
Beyond the seven base units, the SI system encompasses a vast array of derived units. These units are formed by combining the base units through mathematical relationships, allowing for the measurement of virtually any physical quantity encountered in science and technology. The clarity and consistency of these derived units are crucial for accurate scientific communication and practical applications.
Force, Energy, and Power: Interconnected Concepts
Derived units like the Newton (N) for force, the Joule (J) for energy, and the Watt (W) for power exemplify the interconnectedness of physical concepts. A Newton is defined as the force required to accelerate a mass of one kilogram at a rate of one meter per second squared ($1 , text{N} = 1 , text{kg} cdot text{m/s}^2$). A Joule is the energy transferred when a force of one Newton is applied over a distance of one meter ($1 , text{J} = 1 , text{N} cdot text{m}$). A Watt is the rate at which energy is transferred or converted, defined as one Joule per second ($1 , text{W} = 1 , text{J/s}$). These relationships are not merely abstract definitions; they reflect fundamental laws of physics, such as Newton’s laws of motion and the principle of conservation of energy.
Electrical and Magnetic Quantities: The Language of Electromagnetism
The SI system provides standardized units for a wide range of electrical and magnetic phenomena. The Volt (V) for electric potential difference, the Ohm ($Omega$) for electrical resistance, the Farad (F) for capacitance, and the Tesla (T) for magnetic flux density are all crucial for understanding and engineering electrical circuits and devices. For instance, Ohm’s Law, a fundamental principle in electrical engineering, states that the voltage across a resistor is directly proportional to the current flowing through it and the resistance of the resistor ($V = I cdot R$), all expressed in SI units.
Pressure, Volume, and Density: Describing Matter and Its Properties
Units for pressure, volume, and density are essential for fields ranging from fluid mechanics to chemistry. The Pascal (Pa) is the SI unit of pressure, defined as one Newton per square meter ($1 , text{Pa} = 1 , text{N/m}^2$). The cubic meter (m$^3$) is the SI unit of volume. Density, the ratio of mass to volume, is expressed in kilograms per cubic meter (kg/m$^3$). Understanding these units allows scientists to accurately quantify and predict the behavior of substances under various conditions, from the atmospheric pressure of a planet to the density of a novel material.
The Global Impact and Future of SI
The widespread adoption and consistent application of the SI system have had a profound and overwhelmingly positive impact on global scientific progress, technological innovation, and international commerce. Its continued evolution reflects the ongoing advancements in scientific understanding and measurement capabilities.
Facilitating International Collaboration and Trade
The SI system acts as a universal language of measurement, removing barriers that once hindered scientific collaboration and international trade. When scientists from different countries share research findings, they can be confident that their measurements are directly comparable. Similarly, in manufacturing and trade, standardized units ensure that products meet specified requirements, preventing costly disputes and misunderstandings. This global consistency is indispensable for complex international projects, such as the development of new pharmaceuticals, the construction of global infrastructure, and the exploration of space.
Driving Technological Advancement
The precision and coherence of the SI system are fundamental to technological innovation. From the microchips that power our digital devices to the advanced sensors used in autonomous vehicles, the design and manufacturing processes rely heavily on accurate measurements. The ability to precisely define and measure quantities like length, time, and electric current at extremely small scales or over vast distances is what enables the development of increasingly sophisticated technologies. As scientific understanding deepens, the SI system continues to adapt, incorporating new definitions that are based on fundamental constants, ensuring its relevance for future technological leaps.
Ongoing Refinement and the Future of Measurement
The SI system is not static. The 2019 redefinition of the base units is a prime example of its dynamic nature. As scientific understanding progresses and measurement techniques become more sophisticated, the definitions of the base units are periodically reviewed and refined to ensure they remain as accurate and stable as possible. Future developments may involve further integration of quantum phenomena or the exploration of new fundamental constants to underpin our measurement standards. The ongoing evolution of the SI system underscores its commitment to providing a robust and future-proof framework for measurement, essential for continued scientific discovery and technological progress across the globe.
