Mad Cow Disease, scientifically known as Bovine Spongiform Encephalopathy (BSE), is a devastating neurodegenerative disease that affects cattle. While the primary concern is its impact on livestock, the potential for it to transmit to humans and cause a similarly fatal prion disease has been a significant public health issue. The human form of this disease is called Variant Creutzfeldt-Jakob Disease (vCJD). Understanding how BSE affects cattle and how it can cross the species barrier to humans is crucial for appreciating the public health measures implemented to prevent its spread. This article will delve into the nature of prion diseases, the pathological processes of BSE in cattle, and the mechanisms by which it can affect human health.

Understanding Prion Diseases: The Rogue Proteins
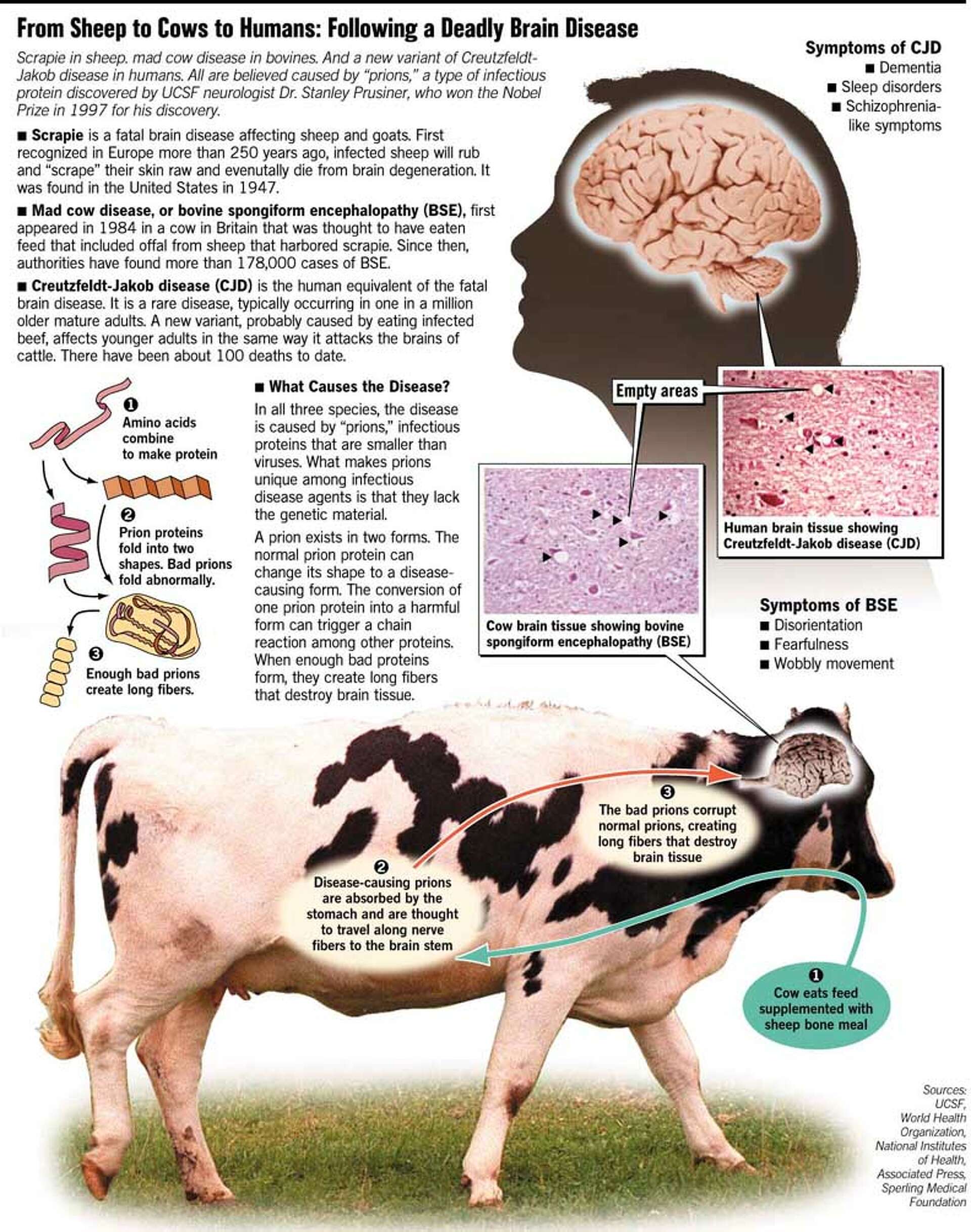
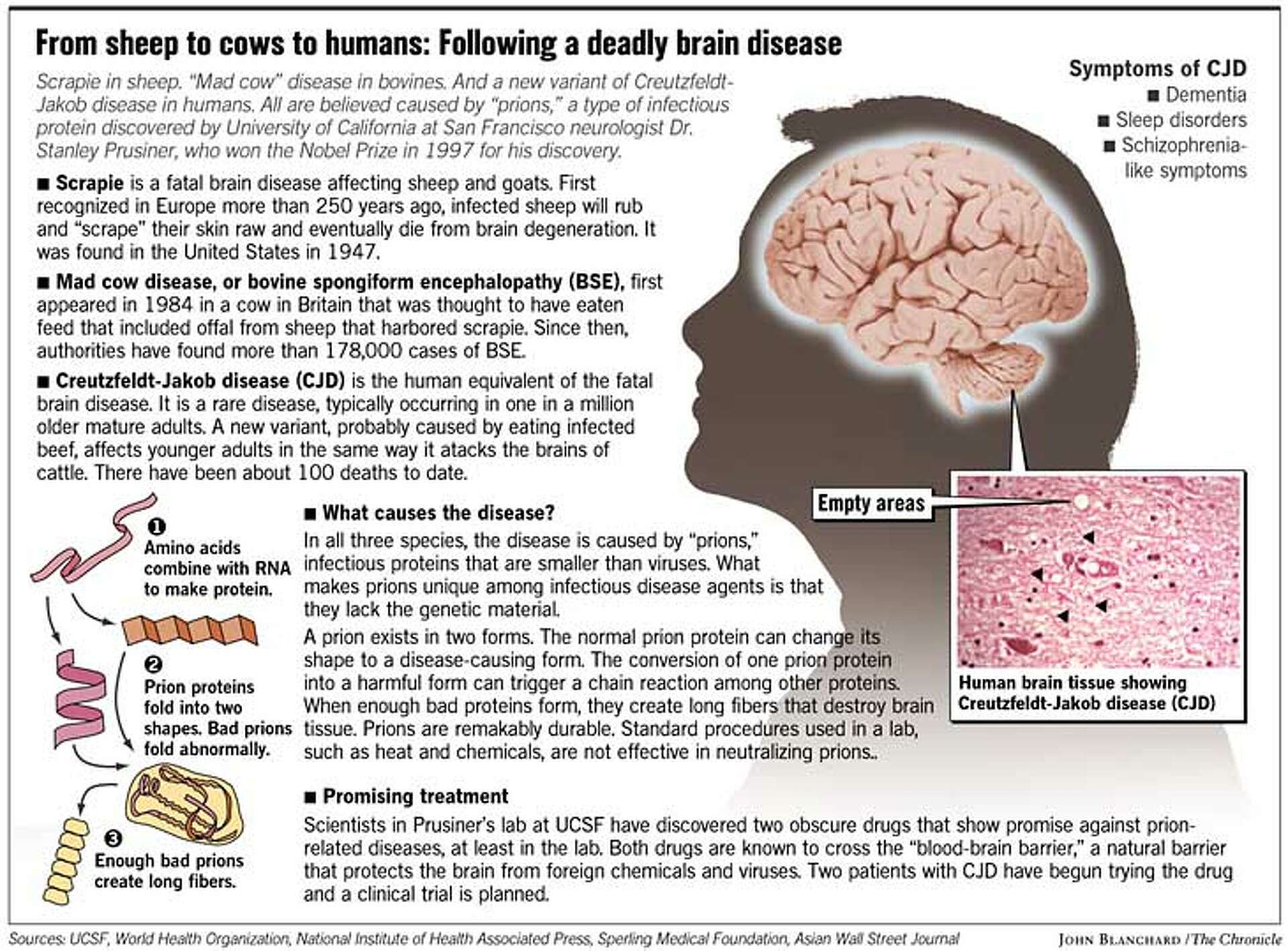
The root of Mad Cow Disease and its human equivalent lies in a class of naturally occurring proteins called prions. These are not viruses or bacteria, but rather misfolded proteins that can induce normal, healthy prions in the brain to also misfold. This chain reaction is the hallmark of prion diseases, leading to a progressive and irreversible destruction of brain tissue.
The Normal Prion Protein (PrPC)
In its healthy form, the prion protein, denoted as PrPC, is found in abundance in the brain and other tissues of mammals. Its exact physiological function is still a subject of ongoing research, but it is believed to play roles in cellular signaling, neuronal development, and protection against oxidative stress. PrPC is a soluble protein, and its three-dimensional structure is critical for its normal function.
The Misfolded Prion Protein (PrPSc)
The danger arises when PrPC misfolds into an abnormal, infectious form known as PrPSc. This misfolded prion protein is characterized by a different three-dimensional structure, making it resistant to degradation by enzymes that normally break down cellular waste. The key feature of PrPSc is its ability to interact with normal PrPC molecules and induce them to also misfold into the pathogenic form.
The Cascade of Neurodegeneration
Once PrPSc begins to accumulate, it forms aggregates, often referred to as amyloid plaques or fibrils, within the brain. These aggregates are toxic to neurons, disrupting their function and eventually leading to their death. As more and more neurons are destroyed, the brain tissue begins to develop characteristic “spongiform” changes – small holes or vacuoles – giving rise to the term “spongiform encephalopathy.” This progressive neuronal loss results in the severe neurological symptoms observed in both BSE and vCJD. The infectious nature of the PrPSc molecule, its ability to recruit and convert normal prions, is what makes these diseases so insidious and challenging to combat. There is no known cure or effective treatment for prion diseases, and they are invariably fatal.
Bovine Spongiform Encephalopathy (BSE): The Cattle Connection
BSE emerged as a significant animal health concern in the late 1980s, primarily in the United Kingdom. The disease was linked to the practice of feeding cattle meat and bone meal derived from infected animals, a practice that inadvertently created a pathway for the prions to enter the food chain.
The Origin and Spread of BSE
The exact origin of the first BSE case remains somewhat elusive, but it is widely believed that the prions were introduced into the cattle population through contaminated animal feed. Specifically, the practice of recycling animal carcasses into protein-rich feed for cattle, which are herbivores, provided a perfect environment for the prion to propagate. When infected animal by-products were processed into meat and bone meal, and then fed to other cattle, the prions could accumulate and spread within the herd. The incubation period for BSE in cattle is typically long, often spanning several years, meaning that infected animals could be present in the food supply for a considerable time before symptoms became apparent.
Pathological Changes in Infected Cattle
Infected cattle develop a progressive neurological disorder characterized by a range of symptoms. These can include changes in temperament, such as nervousness or aggression, difficulties with coordination and movement, particularly affecting the hind limbs, and a general decline in milk production and body condition. As the disease progresses, cattle may stumble, have trouble standing, and exhibit tremors. Ultimately, BSE leads to severe incapacitation and death.
The pathological hallmark of BSE in cattle, as in other prion diseases, is the accumulation of PrPSc in the brain, particularly in the brainstem and spinal cord. This leads to neuronal vacuolation, astrogliosis (an increase in glial cells that support neurons), and the formation of amyloid plaques composed of PrPSc. The distribution and severity of these pathological changes correlate with the clinical signs observed in the affected animals. The identification of BSE in cattle raised immediate alarms within the scientific and public health communities due to the potential for zoonotic transmission – the spread of disease from animals to humans.
Variant Creutzfeldt-Jakob Disease (vCJD): The Human Impact
The recognition that BSE could be transmitted to humans represented a significant public health crisis. Variant Creutzfeldt-Jakob Disease (vCJD) is the human counterpart to BSE, caused by the same type of prion. The primary route of human exposure to the BSE agent was through the consumption of beef products contaminated with nervous tissue from infected cattle.
Transmission to Humans: The Food Chain Pathway
The link between BSE and vCJD was established through extensive epidemiological and laboratory investigations. It became clear that the consumption of beef, particularly certain cuts and processed products that might contain residual nervous system tissue, from cattle infected with BSE was the principal route of transmission. The prions are most concentrated in the brain, spinal cord, and certain other tissues of infected animals. Even microscopic contamination could potentially lead to infection.
The long incubation period of vCJD, similar to BSE, meant that individuals could be infected for years before symptoms manifested. This made it challenging to identify the extent of the outbreak and to implement effective control measures. The potential for prions to survive standard cooking temperatures further complicated efforts to prevent transmission through food.
Clinical Manifestations and Neuropathology in vCJD
The symptoms of vCJD are devastating and progressive, affecting the central nervous system. Early symptoms are often psychiatric and sensory in nature, including depression, anxiety, social withdrawal, and persistent pain. Over time, neurological signs emerge, such as ataxia (loss of coordination), involuntary movements (dyskinesias), cognitive decline, and eventually dementia. Unlike the classic form of CJD, which can occur in older individuals and has a more rapid progression, vCJD typically affects younger adults and has a longer, though still inevitably fatal, course.
Neuropathologically, the brains of vCJD patients exhibit many similarities to BSE-affected cattle, including spongiform changes and the presence of PrPSc deposits. A characteristic feature of vCJD is the presence of “florid plaques” – dense amyloid deposits surrounded by a halo of vacuolated prion protein – which are a distinctive marker of the disease. The accumulation of PrPSc in specific regions of the brain leads to neuronal dysfunction and death, ultimately resulting in the profound neurological deterioration observed in patients.
Public Health Measures and Future Outlook
The emergence of BSE and vCJD necessitated a robust and multifaceted public health response. These measures have been largely successful in drastically reducing the incidence of new cases, but ongoing vigilance remains crucial.
Prevention and Control Strategies
Following the identification of the link between BSE and vCJD, governments worldwide implemented stringent control measures. These included:
- Banning the feeding of ruminant-derived meat and bone meal to ruminants: This was a critical step in breaking the cycle of prion transmission within the cattle population.
- Removal of specified risk materials (SRMs): This involved the systematic removal and destruction of cattle tissues considered to have the highest risk of containing prions, such as the brain, spinal cord, and tonsils, from the food chain.
- Enhanced surveillance and testing: Regular testing of cattle populations for BSE allowed for early detection and removal of infected animals.
- Traceability and import/export controls: Strict regulations were put in place to track cattle and beef products, preventing the movement of potentially contaminated animals or products across borders.
- Public awareness campaigns: Educating consumers about the risks and the safety measures in place helped to restore confidence in the food supply.
These comprehensive strategies have been remarkably effective. The incidence of BSE in cattle has plummeted, and the number of reported vCJD cases in humans has become extremely rare.
Ongoing Research and Vigilance
Despite the success of control measures, research into prion diseases continues. Scientists are investigating the precise mechanisms of prion replication and pathogenesis, as well as exploring potential therapeutic interventions. While a cure remains elusive, understanding the fundamental biology of prions could pave the way for future treatments.
Furthermore, ongoing surveillance and monitoring are essential to detect any resurgence of BSE or the emergence of new prion threats. The long incubation periods of these diseases mean that a sustained effort is required to ensure long-term public safety. The lessons learned from the BSE and vCJD outbreaks have fundamentally reshaped our understanding of zoonotic diseases and the importance of integrated surveillance systems that monitor both animal and human health. The focus has shifted from simply eradicating disease to building resilient systems that can detect, respond to, and prevent future emerging threats to human and animal well-being.
