The fundamental building blocks of matter, atoms, are constantly interacting, forming new substances and transforming existing ones. These interactions are what we call chemical reactions. Understanding the various types of chemical reactions is crucial for comprehending everything from the everyday processes that sustain life to the complex industrial procedures that drive our modern world. From the burning of fuel that powers our vehicles to the photosynthesis that allows plants to create energy, chemical reactions are the engines of change in the universe. This exploration will delve into the primary classifications of chemical reactions, providing a comprehensive overview of how matter rearranges and transforms.
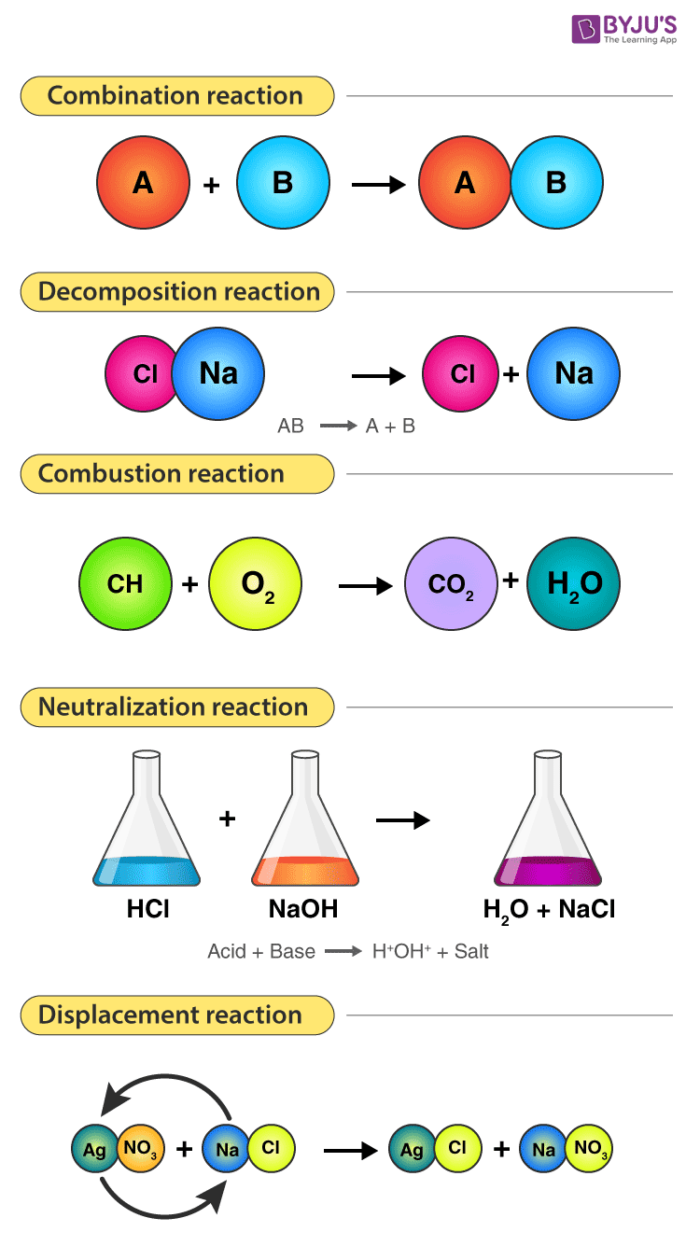
Combination Reactions: The Art of Merging
One of the simplest yet most fundamental types of chemical reactions is the combination reaction, also known as a synthesis reaction. In this process, two or more reactants, which can be elements or compounds, join together to form a single, more complex product. The core principle here is the merging of simpler substances into a more intricate whole. This can be visualized as building with LEGO bricks: you start with individual bricks and combine them to create a larger, more elaborate structure.
Formation of Compounds from Elements
A common manifestation of combination reactions is the formation of compounds from their constituent elements. For instance, when hydrogen gas ($H2$) is ignited in the presence of oxygen gas ($O2$), they combine to form water ($H_2O$). The balanced chemical equation for this reaction is:
$2H2(g) + O2(g) rightarrow 2H_2O(l)$
Here, two molecules of hydrogen and one molecule of oxygen react to produce two molecules of water. Another example is the reaction between sodium metal ($Na$) and chlorine gas ($Cl_2$) to form sodium chloride ($NaCl$), commonly known as table salt:
$2Na(s) + Cl_2(g) rightarrow 2NaCl(s)$
In this case, a highly reactive alkali metal combines with a poisonous gas to form a stable, edible compound. These reactions often release significant amounts of energy, making them exothermic. The driving force behind these reactions is the tendency of atoms to achieve a more stable electron configuration, typically by forming covalent or ionic bonds.
Formation of Compounds from Simpler Compounds
Combination reactions are not limited to the combination of elements. Simpler compounds can also combine to form more complex ones. For example, sulfur trioxide ($SO3$) can react with water ($H2O$) to form sulfuric acid ($H2SO4$):
$SO3(g) + H2O(l) rightarrow H2SO4(aq)$
Sulfuric acid is a highly corrosive and important industrial chemical used in the production of fertilizers, detergents, and in petroleum refining. Another illustrative example is the reaction between calcium oxide ($CaO$, quicklime) and carbon dioxide ($CO2$) to form calcium carbonate ($CaCO3$, limestone):
$CaO(s) + CO2(g) rightarrow CaCO3(s)$
This reaction is significant in geological processes, such as the formation of stalactites and stalagmites in caves, and in construction, where limestone is a primary building material. The ability of simpler compounds to combine to form more complex ones highlights the versatility of chemical synthesis and the potential for creating a vast array of substances from a limited set of starting materials.
Decomposition Reactions: The Art of Breaking Down
In stark contrast to combination reactions, decomposition reactions involve the breakdown of a single compound into two or more simpler substances. These simpler substances can be elements or smaller compounds. This process is essentially the reverse of synthesis, where a complex entity is disassembled into its constituent parts. Think of it as taking apart a complex LEGO structure to get back the individual bricks. Decomposition reactions often require energy input, such as heat, light, or electricity, to break the chemical bonds within the compound.
Thermal Decomposition
The most common type of decomposition reaction is thermal decomposition, where heat is used to break down a compound. A classic example is the decomposition of calcium carbonate ($CaCO_3$) when heated:
$CaCO3(s) xrightarrow{Delta} CaO(s) + CO2(g)$
Upon heating, calcium carbonate breaks down into calcium oxide and carbon dioxide gas. This process is industrially important for the production of quicklime ($CaO$), which is used in cement and mortar. Another example is the decomposition of potassium nitrate ($KNO3$) upon strong heating, which produces potassium nitrite ($KNO2$) and oxygen gas ($O_2$):
$2KNO3(s) xrightarrow{Delta} 2KNO2(s) + O_2(g)$
This reaction is relevant in pyrotechnics and in some fertilizers. The amount of heat required and the products formed depend on the specific compound and the conditions of the reaction.
Electrolytic Decomposition
Electrolysis is a process that uses electricity to drive a non-spontaneous chemical reaction, often a decomposition. A prominent example is the electrolysis of water ($H2O$), which decomposes into hydrogen gas ($H2$) and oxygen gas ($O_2$):
$2H2O(l) xrightarrow{electrolysis} 2H2(g) + O_2(g)$
This is a crucial method for producing high-purity hydrogen and oxygen on an industrial scale. The decomposition of molten salts can also be achieved through electrolysis. For instance, the electrolysis of molten sodium chloride ($NaCl$) yields molten sodium metal ($Na$) and chlorine gas ($Cl_2$):
$2NaCl(l) xrightarrow{electrolysis} 2Na(l) + Cl_2(g)$
This method is the primary way to produce elemental sodium and chlorine, both of which have significant industrial applications. The energy provided by electricity overcomes the bond energies holding the compound together, facilitating its decomposition.
Photolytic Decomposition
Some compounds can decompose when exposed to light, a process known as photolytic decomposition. A familiar example is the decomposition of silver chloride ($AgCl$), which is light-sensitive and is used in photographic film:
$2AgCl(s) xrightarrow{light} 2Ag(s) + Cl_2(g)$
When light strikes silver chloride, it breaks down into elemental silver and chlorine gas. This property is fundamental to traditional photography. Another example is the decomposition of hydrogen peroxide ($H2O2$) in the presence of light, although this decomposition is also catalyzed by other substances:
$2H2O2(aq) xrightarrow{light} 2H2O(l) + O2(g)$
Hydrogen peroxide is a common household disinfectant and bleaching agent, and its decomposition into water and oxygen is a practical demonstration of photolytic breakdown.
Single Displacement Reactions: The Art of Swapping Partners
Single displacement reactions, also known as single replacement reactions, occur when one element replaces another element in a compound. This type of reaction involves the interaction of an element with a compound, leading to the formation of a new compound and the release of a different element. The fundamental concept here is an exchange, where one participant in a compound is substituted by another.
Metal Displacement of Another Metal
Metals can displace other metals from their salt solutions if the displacing metal is more reactive. This reactivity is often described by an activity series, which ranks metals based on their tendency to lose electrons. For example, zinc ($Zn$) is more reactive than copper ($Cu$). Therefore, when zinc metal is placed in a solution of copper sulfate ($CuSO_4$), zinc displaces copper:
$Zn(s) + CuSO4(aq) rightarrow ZnSO4(aq) + Cu(s)$
Here, zinc atoms lose electrons to form zinc ions ($Zn^{2+}$), while copper ions ($Cu^{2+}$) gain electrons to become copper atoms. The zinc sulfate solution remains, and solid copper is deposited. Conversely, if copper metal were placed in a zinc sulfate solution, no reaction would occur because copper is less reactive than zinc.
Metal Displacement of Hydrogen

Active metals can also displace hydrogen from acids or water. For instance, when sodium metal ($Na$) reacts with water ($H2O$), it displaces hydrogen, forming sodium hydroxide ($NaOH$) and hydrogen gas ($H2$):
$2Na(s) + 2H2O(l) rightarrow 2NaOH(aq) + H2(g)$
This reaction is quite vigorous and exothermic. Similarly, metals like magnesium ($Mg$) react with acids, such as hydrochloric acid ($HCl$), to produce a salt and hydrogen gas:
$Mg(s) + 2HCl(aq) rightarrow MgCl2(aq) + H2(g)$
The reactivity of the metal determines whether it can displace hydrogen from water or only from acids.
Halogen Displacement
Halogens, like chlorine ($Cl2$), bromine ($Br2$), and iodine ($I_2$), can also displace other halogens from their halide salts. A more reactive halogen will displace a less reactive one. For example, chlorine is more reactive than bromine. Thus, when chlorine gas is bubbled through a solution of potassium bromide ($KBr$), it displaces bromine:
$Cl2(g) + 2KBr(aq) rightarrow 2KCl(aq) + Br2(aq)$
Chlorine atoms gain electrons to form chloride ions, and bromide ions lose electrons to form bromine molecules. This reaction can be observed by the change in color of the solution, as bromine imparts a yellowish-brown hue.
Double Displacement Reactions: The Art of Exchanging Ions
Double displacement reactions, also known as metathesis reactions, involve the exchange of ions between two reacting compounds. In essence, the cations and anions of two different ionic compounds swap partners. These reactions typically occur in aqueous solutions, and a reaction is usually driven by the formation of a precipitate, a gas, or a molecular compound like water.
Precipitation Reactions
When two soluble ionic compounds are mixed, and an insoluble ionic compound is formed, this is called a precipitation reaction. The insoluble compound, the precipitate, settles out of the solution. For example, when a solution of silver nitrate ($AgNO_3$) is mixed with a solution of sodium chloride ($NaCl$), silver chloride ($AgCl$) precipitates out:
$AgNO3(aq) + NaCl(aq) rightarrow AgCl(s) + NaNO3(aq)$
In this reaction, the silver ions ($Ag^+$) from silver nitrate combine with the chloride ions ($Cl^-$) from sodium chloride to form insoluble silver chloride. The sodium ions ($Na^+$) and nitrate ions ($NO_3^-$) remain in solution as sodium nitrate. Predicting whether a precipitate will form relies on solubility rules for ionic compounds.
Acid-Base Neutralization Reactions
Acid-base neutralization reactions are a specific type of double displacement reaction where an acid reacts with a base to form a salt and water. The key process is the reaction between hydrogen ions ($H^+$) from the acid and hydroxide ions ($OH^-$) from the base to form water, a molecular compound. For instance, the reaction between hydrochloric acid ($HCl$) and sodium hydroxide ($NaOH$) is a neutralization reaction:
$HCl(aq) + NaOH(aq) rightarrow NaCl(aq) + H_2O(l)$
Here, $H^+$ ions from $HCl$ combine with $OH^-$ ions from $NaOH$ to form $H_2O$. The remaining ions, $Na^+$ and $Cl^-$, form the salt sodium chloride. These reactions are called neutralization because the acidic and basic properties are canceled out, resulting in a solution that is typically closer to neutral pH.
Gas-Forming Reactions
In some double displacement reactions, one of the products is a gas that escapes from the solution, driving the reaction forward. For example, the reaction between hydrochloric acid ($HCl$) and sodium carbonate ($Na2CO3$) produces carbon dioxide gas ($CO_2$):
$2HCl(aq) + Na2CO3(aq) rightarrow 2NaCl(aq) + H2O(l) + CO2(g)$
The formation of carbon dioxide gas is evident as effervescence. Similarly, reactions involving sulfides can produce hydrogen sulfide gas ($H_2S$):
$FeS(s) + 2HCl(aq) rightarrow FeCl2(aq) + H2S(g)$
The release of a gas is a strong driving force for these reactions to occur.
Combustion Reactions: The Art of Rapid Reaction with Oxygen
Combustion is a rapid chemical process that involves the reaction of a substance with an oxidant, usually oxygen, to produce heat and light. It is a highly exothermic process, meaning it releases a significant amount of energy. While most commonly associated with burning, combustion encompasses a range of rapid oxidation reactions.
Combustion of Hydrocarbons
The most familiar type of combustion involves hydrocarbons, which are compounds composed of hydrogen and carbon. When a hydrocarbon burns completely in the presence of sufficient oxygen, the products are carbon dioxide ($CO2$) and water ($H2O$). For example, the combustion of methane ($CH_4$), the primary component of natural gas, is:
$CH4(g) + 2O2(g) rightarrow CO2(g) + 2H2O(g) + text{heat}$
This reaction is the basis for many heating and power generation systems. The burning of fuels like gasoline, propane, and wood are all examples of hydrocarbon combustion.
Incomplete Combustion
If the supply of oxygen is limited during the combustion of a hydrocarbon, incomplete combustion occurs. In addition to carbon dioxide and water, incomplete combustion produces carbon monoxide ($CO$), a toxic gas, and soot (elemental carbon, $C$). For example, incomplete combustion of methane can yield:
$2CH4(g) + 3O2(g) rightarrow 2CO(g) + 4H_2O(g) + text{heat}$
or
$CH4(g) + O2(g) rightarrow C(s) + 2H_2O(g) + text{heat}$
Incomplete combustion is a concern due to the production of carbon monoxide, which is odorless and colorless, and can be fatal. This is why proper ventilation is crucial when using combustion appliances.
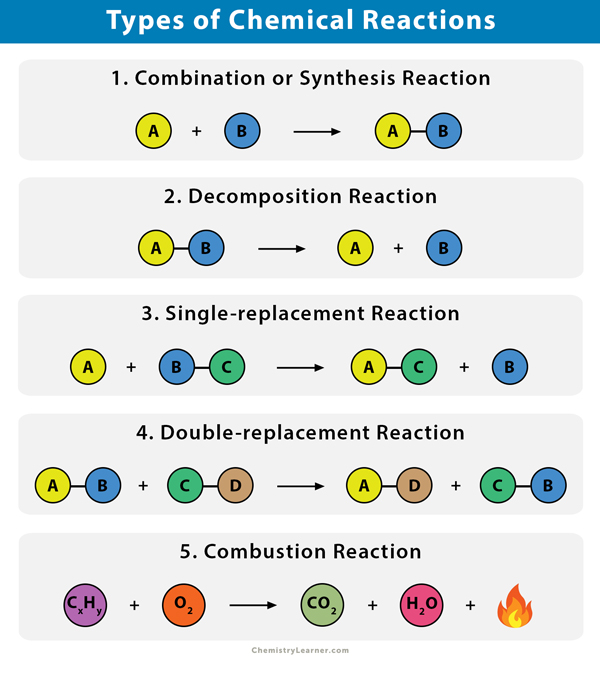
Combustion of Other Substances
Combustion is not limited to hydrocarbons. Other substances can also undergo combustion. For instance, the combustion of sulfur in oxygen produces sulfur dioxide:
$S(s) + O2(g) rightarrow SO2(g) + text{heat}$
Metals can also combust, although often requiring higher temperatures or specific conditions. The combustion of magnesium, for example, produces magnesium oxide and releases a bright light:
$2Mg(s) + O_2(g) rightarrow 2MgO(s) + text{heat}$
Understanding combustion is vital for safety, energy production, and environmental studies.
In conclusion, the classification of chemical reactions into categories such as combination, decomposition, single displacement, double displacement, and combustion provides a framework for understanding the myriad of transformations that matter undergoes. Each type of reaction represents a distinct pattern of atomic and molecular rearrangement, governed by fundamental chemical principles. By recognizing these types, we gain a deeper appreciation for the dynamic and ever-changing nature of the chemical world around us.
