Polar bonds represent a fundamental concept in chemistry, crucial for understanding the behavior and properties of countless molecules. This article delves into the nature of polar bonds, exploring their formation, characteristics, and significance within the broader context of chemical bonding.
The Foundation: Covalent Bonds
Before dissecting polar bonds, it’s essential to grasp the concept of a covalent bond. Covalent bonds form when two atoms share electrons. This sharing allows each atom to achieve a more stable electron configuration, often resembling that of a noble gas.
Electronegativity: The Driving Force
The driving force behind covalent bond formation, and specifically polar bonds, is electronegativity. Electronegativity is a measure of an atom’s ability to attract shared electrons in a chemical bond. It’s a periodic property, generally increasing across a period from left to right and decreasing down a group. Fluorine, for instance, is the most electronegative element, fiercely pulling electrons towards itself.
Nonpolar Covalent Bonds: Equal Sharing
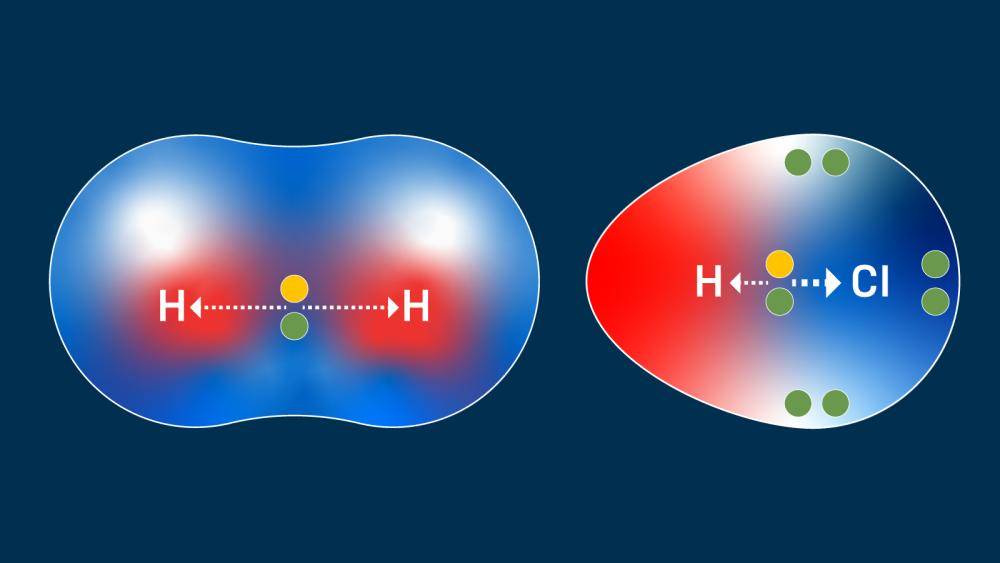
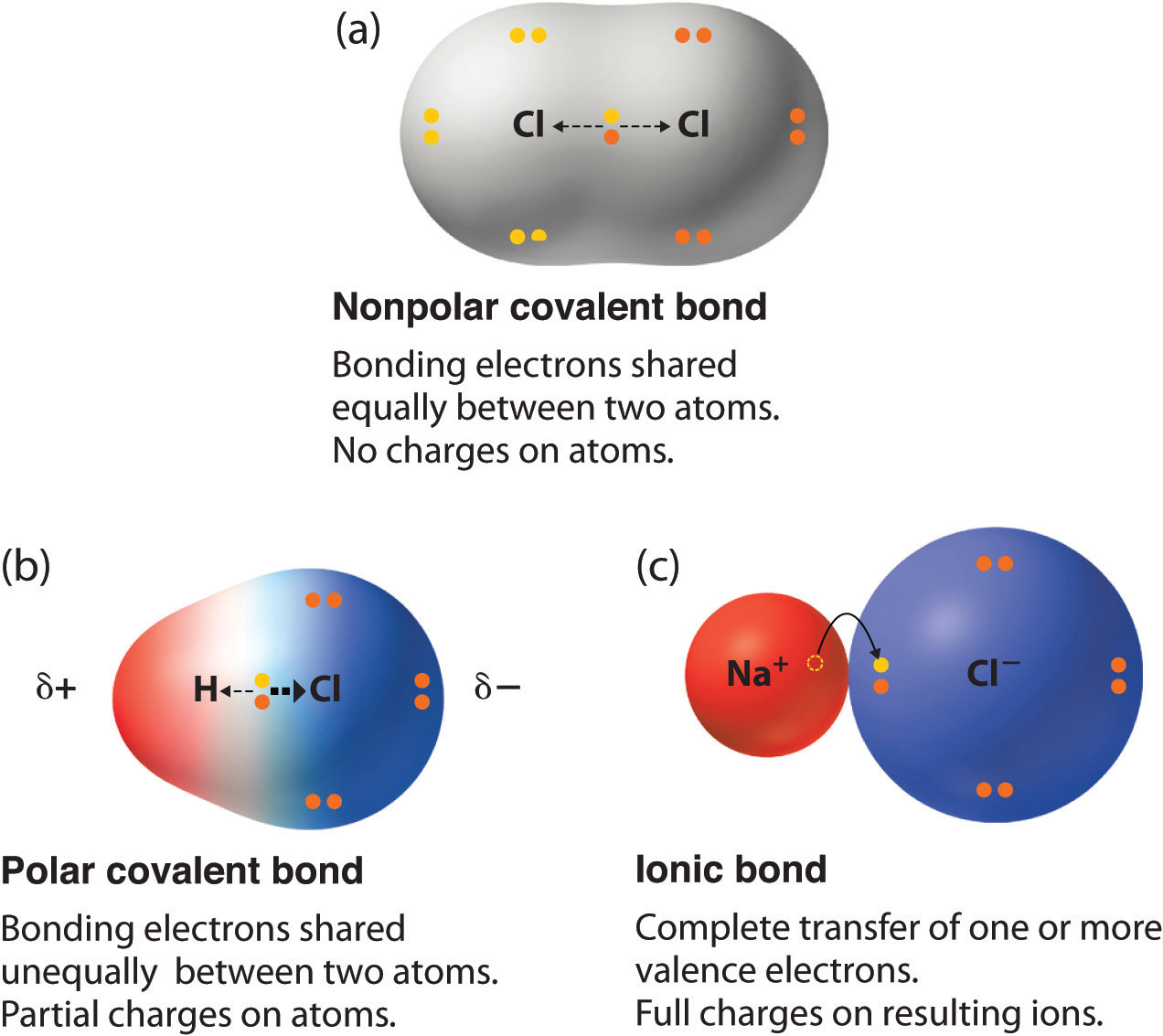
When two atoms with identical or very similar electronegativity values form a covalent bond, the electrons are shared almost equally. This results in a nonpolar covalent bond. In such bonds, there is no significant separation of charge across the bond. Examples include the bonds in diatomic molecules like hydrogen (H₂), oxygen (O₂), and nitrogen (N₂), where two identical atoms are bonded. The electron cloud is distributed symmetrically.
The Genesis of Polar Bonds
Polar covalent bonds arise when there is a significant difference in electronegativity between the two atoms involved in the bond. This disparity leads to an unequal sharing of electrons.
Unequal Electron Distribution
In a polar covalent bond, the more electronegative atom attracts the shared electrons more strongly. This causes the electron cloud to be denser around the more electronegative atom. Consequently, the more electronegative atom gains a partial negative charge, denoted by the symbol $delta^{-}$ (delta minus). Conversely, the less electronegative atom, which is losing electron density, develops a partial positive charge, denoted by $delta^{+}$ (delta plus).
The Dipole Moment
This separation of charge across a bond creates a bond dipole, which is a measure of the polarity of the bond. The magnitude and direction of this charge separation are quantified by the dipole moment. A dipole moment is a vector quantity, meaning it has both magnitude and direction. The direction points from the positive pole ($delta^{+}$) to the negative pole ($delta^{-}$). A larger electronegativity difference between the bonded atoms results in a larger dipole moment and a more polar bond.
Examples of Polar Bonds
A classic example of a polar bond is the hydrogen-oxygen bond in water (H₂O). Oxygen is significantly more electronegative than hydrogen. Therefore, the electrons in the H-O bonds are pulled closer to the oxygen atom, making the oxygen atom partially negative ($delta^{-}$) and the hydrogen atoms partially positive ($delta^{+}$).
Another common example is the hydrogen-chlorine bond in hydrogen chloride (HCl). Chlorine is more electronegative than hydrogen, leading to a partial negative charge on chlorine and a partial positive charge on hydrogen.
Molecular Polarity: Beyond Individual Bonds
While polar bonds are essential for understanding molecular polarity, they are not the sole determinant. The overall polarity of a molecule depends on two factors:
The Polarity of Individual Bonds
As discussed, the presence and degree of polarity in each bond within a molecule are fundamental.
The Molecular Geometry
The three-dimensional arrangement of atoms in a molecule, known as its molecular geometry, plays a critical role. Even if a molecule contains polar bonds, it can be nonpolar overall if the molecular geometry causes the bond dipoles to cancel each other out. This often occurs in symmetrical molecules.
Symmetrical Molecules
In symmetrical molecules, the individual bond dipoles are oriented in such a way that their vector sum is zero. For instance, carbon dioxide (CO₂) has two polar C=O bonds. However, the molecule is linear, with the two oxygen atoms positioned on opposite sides of the carbon atom. The dipole moment of one C=O bond points in one direction, and the dipole moment of the other C=O bond points in the exact opposite direction with equal magnitude. These dipoles cancel each other out, making CO₂ a nonpolar molecule despite having polar bonds.
Similarly, carbon tetrachloride (CCl₄) has four polar C-Cl bonds. The molecule adopts a tetrahedral geometry, which is highly symmetrical. The four bond dipoles, pointing towards the vertices of the tetrahedron, effectively cancel each other out, rendering CCl₄ nonpolar.
Asymmetrical Molecules
In contrast, asymmetrical molecules often exhibit a net dipole moment, making them polar. Water (H₂O) is a prime example. While the O-H bonds are polar, the molecule has a bent geometry. The two O-H bond dipoles do not cancel out; instead, they add up to create a significant net dipole moment, with the oxygen atom being the negative pole and the hydrogen atoms forming the positive pole. This polarity is responsible for many of water’s unique properties.
Ammonia (NH₃) is another example. The N-H bonds are polar, and the molecule has a trigonal pyramidal shape. The bond dipoles add up to create a net dipole moment, with the nitrogen atom being the negative pole.
Properties Influenced by Polar Bonds
The presence of polar bonds and the resulting molecular polarity have profound implications for the physical and chemical properties of substances.
Intermolecular Forces
Polar molecules interact with each other through dipole-dipole interactions. In these interactions, the partially positive end of one polar molecule is attracted to the partially negative end of another polar molecule. These forces are stronger than the London dispersion forces that affect all molecules and are weaker than ionic bonds or covalent bonds.
Hydrogen bonding is a particularly strong type of dipole-dipole interaction that occurs when a hydrogen atom is bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine) and is attracted to a lone pair of electrons on another highly electronegative atom in a different molecule. Hydrogen bonds are crucial in biological systems, such as holding together the two strands of DNA.
Solubility
Polarity significantly influences solubility. “Like dissolves like” is a general rule of thumb. Polar substances tend to dissolve in polar solvents (like water), while nonpolar substances dissolve in nonpolar solvents (like hexane). This is because the intermolecular forces between solute and solvent molecules are similar in polar-polar or nonpolar-nonpolar mixtures, leading to favorable interactions. Polar solutes cannot easily overcome the strong intermolecular forces within a nonpolar solvent, and vice versa.
Boiling and Melting Points
Substances with polar molecules generally have higher boiling and melting points compared to nonpolar substances of similar molecular weight. This is due to the stronger intermolecular forces (dipole-dipole interactions and hydrogen bonding) that must be overcome to change the state of matter. More energy is required to separate polar molecules than nonpolar molecules.
Reactivity
Polarity can also influence the chemical reactivity of a molecule. The partial charges on polar bonds can make certain atoms more susceptible to attack by nucleophiles (electron-rich species) or electrophiles (electron-deficient species). For example, in an SN2 reaction, a nucleophile might attack the partially positive carbon atom in a polar C-X bond (where X is a halogen).

Conclusion: The Ubiquity of Polar Bonds
Polar bonds are not merely an academic concept; they are the bedrock upon which much of our understanding of chemistry, biology, and material science is built. From the solvent properties of water to the structure of biological macromolecules and the behavior of everyday plastics, the influence of polar bonds is pervasive. Recognizing and understanding the principles of polar bonding allows us to predict and explain the diverse and fascinating properties of the chemical world around us. They are a testament to the subtle yet powerful ways in which electrons, the fundamental currency of chemical interaction, dictate the macroscopic world.
