The term “London Forces” is not a recognized scientific or technical term within the realm of physics or chemistry. It’s highly probable that the query is a misinterpretation or a colloquialism, and the user is likely interested in London Dispersion Forces, also known as van der Waals forces or instantaneous dipole-induced dipole forces. These are a fundamental type of intermolecular force that play a crucial role in the behavior of molecules, particularly in areas related to physical chemistry, material science, and even the interactions of tiny particles, which can have relevance in some niche technological applications.
This article will delve into the nature, origin, and significance of London Dispersion Forces, exploring their impact on various phenomena and their indirect connections to technological advancements.
The Foundation: Intermolecular Forces
Before dissecting London Dispersion Forces, it’s essential to understand the broader context of intermolecular forces (IMFs). These are attractive or repulsive forces that arise between neighboring molecules. Unlike intramolecular forces (like covalent or ionic bonds) that hold atoms together within a molecule, IMFs govern the interactions between separate molecules. These forces are significantly weaker than intramolecular forces but are nevertheless critical in determining the physical properties of substances, such as their melting points, boiling points, viscosity, and solubility.
The primary types of intermolecular forces include:
- Ion-Dipole Forces: Occur between an ion and a polar molecule.
- Hydrogen Bonds: A special type of dipole-dipole interaction involving hydrogen bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine).
- Dipole-Dipole Forces: Exist between two polar molecules, where the positive end of one molecule is attracted to the negative end of another.
- London Dispersion Forces: The focus of this discussion, these forces exist between all molecules, regardless of whether they are polar or nonpolar.
Unveiling London Dispersion Forces: Origin and Mechanism
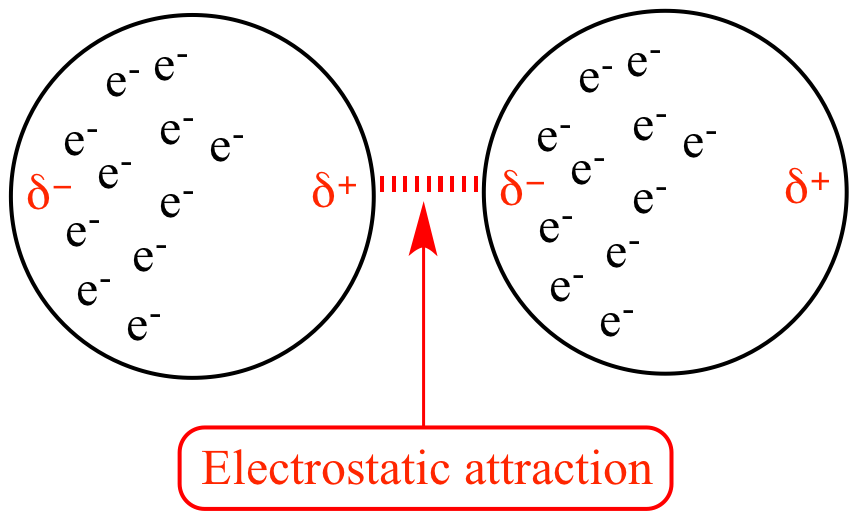
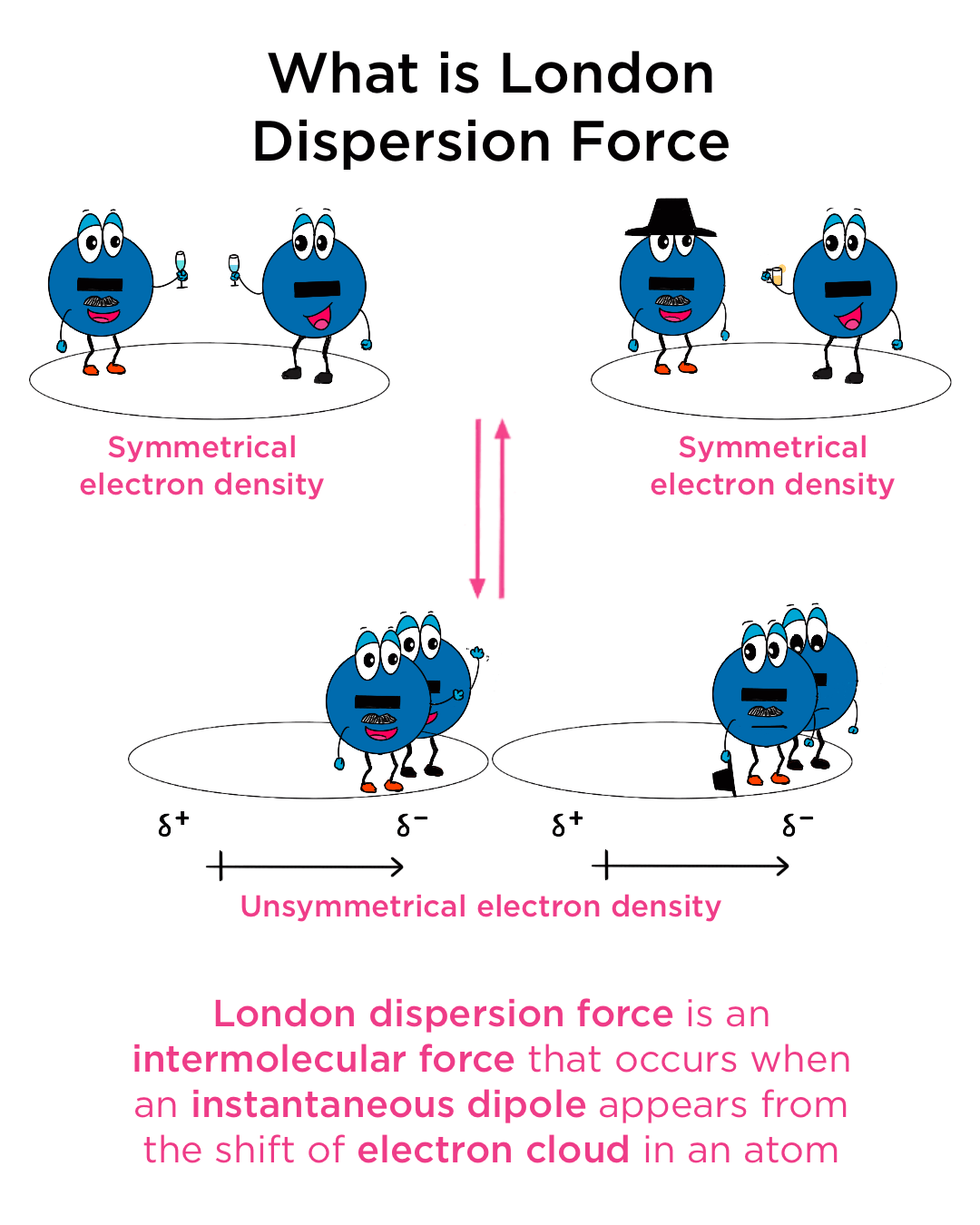
London Dispersion Forces (LDFs) are the weakest of the intermolecular forces, but their ubiquitous nature makes them highly significant. They arise from the temporary fluctuations in electron distribution within molecules.
Electron Cloud Dynamics
At any given moment, the electrons in an atom or molecule are not static; they are in constant motion. This dynamic movement means that the electron cloud is not perfectly symmetrical. At any instant, there might be a slightly greater concentration of electrons on one side of the molecule than on the other. This creates a temporary, instantaneous dipole.
Imagine a nonpolar molecule, like a diatomic oxygen molecule (O₂). While the molecule as a whole is neutral and has no permanent dipole, the electrons orbiting the nuclei are in constant motion. For a fleeting moment, more electrons might be found on the left side of the molecule than on the right. This momentary imbalance results in a slight negative charge on the left side and a slight positive charge on the right.
The Induced Dipole Effect
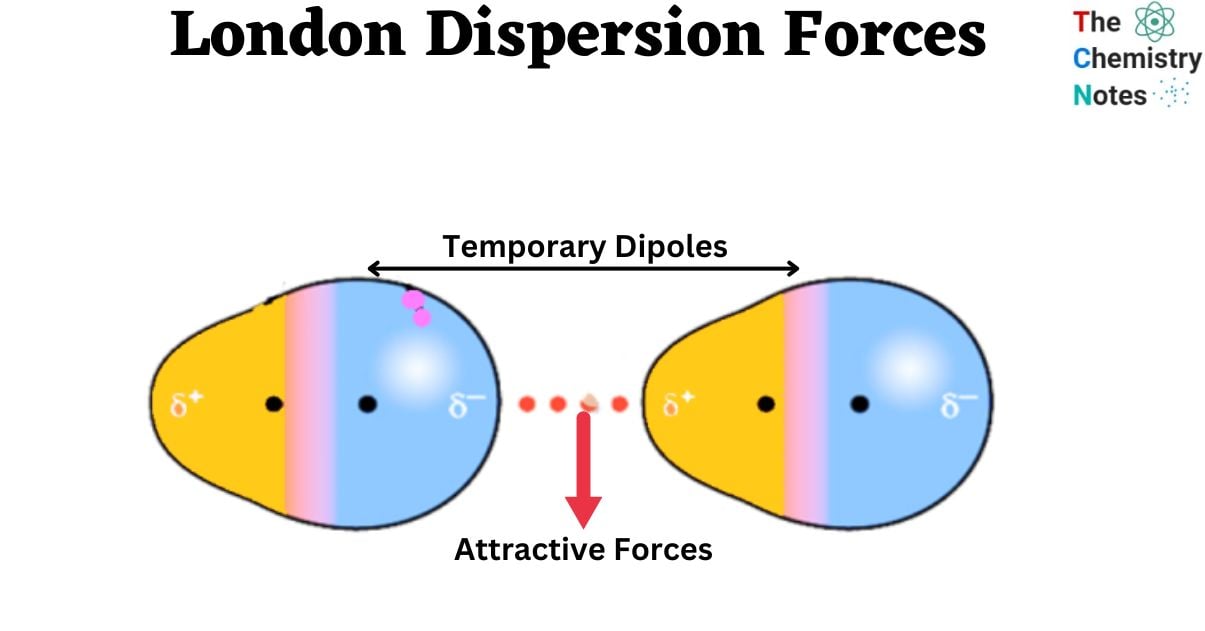
This instantaneous dipole in one molecule can then influence the electron clouds of neighboring molecules. The slight negative charge of the temporary dipole will repel the electrons in an adjacent molecule, pushing them away. Conversely, the slight positive charge will attract the electrons. This process induces a temporary dipole in the neighboring molecule.
So, the instantaneous dipole on molecule A creates a complementary induced dipole on molecule B. The positive end of the induced dipole on molecule B is then attracted to the negative end of the instantaneous dipole on molecule A, and vice-versa. This attraction, however weak, constitutes the London Dispersion Force.
Fluctuations and Motion
It’s crucial to understand that these dipoles are temporary. The electron distribution is constantly changing, so the instantaneous dipoles and the induced dipoles are continuously forming, shifting, and disappearing. However, the cumulative effect of these fleeting attractions over a large number of molecules and over time can be substantial.
Factors Influencing the Strength of London Dispersion Forces
While LDFs are universally present, their strength can vary significantly depending on molecular characteristics. Two primary factors influence the magnitude of London Dispersion Forces:
1. Number of Electrons (Molecular Size and Mass)
The more electrons a molecule possesses, the larger and more diffuse its electron cloud. A larger electron cloud is more easily distorted or polarized by the fluctuating electron density. This means that molecules with a greater number of electrons will experience stronger London Dispersion Forces.
- Example: Consider the noble gases. Helium (He) has only two electrons, while Xenon (Xe) has 54 electrons. Consequently, Xenon has significantly stronger LDFs than Helium. This difference is reflected in their boiling points: Helium boils at -268.9 °C, while Xenon boils at -108.1 °C. The higher boiling point of Xenon indicates that more energy is required to overcome the intermolecular forces, which in this case are primarily LDFs.
Larger molecules, generally having more electrons, will exhibit stronger LDFs. For instance, octane (C₈H₁₈) is a liquid at room temperature, while ethane (C₂H₆), a smaller alkane, is a gas. The increased LDFs in octane due to its larger size and greater electron count contribute to its liquid state.
2. Molecular Shape (Surface Area and Proximity)
The shape of a molecule also plays a vital role in determining the strength of London Dispersion Forces. Molecules with a larger surface area allow for greater contact between adjacent molecules, leading to more points of interaction and thus stronger LDFs.
- Example: Compare n-pentane (a linear molecule) and neopentane (a more spherical molecule), both with the formula C₅H₁₂. N-pentane has a higher boiling point (36.1 °C) than neopentane (9.5 °C). This difference arises because the linear shape of n-pentane allows for greater surface area contact and thus stronger LDFs between n-pentane molecules compared to the more compact, spherical neopentane molecules.
This principle is particularly relevant in the context of how molecules pack together in solid or liquid states. A more spread-out, elongated molecule can align itself more effectively with its neighbors, maximizing the attractive forces.
Significance of London Dispersion Forces
Despite being the weakest type of IMF, London Dispersion Forces are incredibly important and have widespread implications across various scientific disciplines and technological applications.
1. Physical Properties of Nonpolar Substances
For nonpolar molecules, LDFs are the only intermolecular forces present. Therefore, they are solely responsible for determining the physical properties of these substances, such as their boiling and melting points, as well as their solubility in nonpolar solvents. Substances like alkanes, diatomic gases (O₂, N₂), and noble gases owe their condensed states (liquids and solids) at low temperatures to LDFs.
2. Contribution to Polar Molecules
Even in polar molecules that exhibit dipole-dipole forces and potentially hydrogen bonding, London Dispersion Forces are still present and contribute to the overall intermolecular attraction. In larger polar molecules, LDFs can sometimes become the dominant force. For example, in long-chain fatty acids, the large nonpolar hydrocarbon tail contributes significantly to LDFs, influencing their physical properties like melting point.
3. Surface Chemistry and Adhesion
LDFs are crucial in many surface phenomena. Adhesion, the tendency of dissimilar particles or surfaces to cling to one another, can be significantly influenced by LDFs. This is relevant in:
- Adhesives and Coatings: Many glues and coatings rely on weak intermolecular forces, including LDFs, to bond surfaces.
- Friction: The friction between surfaces can be attributed, in part, to the van der Waals forces (which include LDFs) between the molecules at the points of contact.
4. Biological Systems
While hydrogen bonding and electrostatic interactions are often highlighted in biological contexts, LDFs play a subtle but important role in molecular recognition and the stability of biomolecules. For instance, the hydrophobic effect, a driving force for protein folding and the formation of cell membranes, is largely driven by the tendency of nonpolar molecules to minimize their contact with water. This minimization is achieved through increased van der Waals interactions (including LDFs) among the nonpolar molecules themselves, effectively pushing them together.
5. Material Science and Nanotechnology
In advanced materials and nanotechnology, precise control over molecular interactions is paramount. LDFs are considered when designing:
- Self-Assembling Materials: The controlled assembly of molecules into ordered structures often relies on predictable intermolecular forces, including LDFs.
- Nanoparticles and Colloids: The stability and aggregation behavior of nanoparticles in a solution are influenced by the balance of attractive and repulsive forces, with LDFs contributing to the attraction.
- Graphene and 2D Materials: The stacking of layers in materials like graphite and graphene is held together by strong LDFs. Understanding these forces is critical for manipulating and utilizing these advanced materials.
Indirect Connections to Drone Technology and Related Fields
While “London Forces” are a fundamental concept in chemistry and physics, their direct application in the specified drone-related categories (Drones, Flight Technology, Cameras & Imaging, Drone Accessories, Aerial Filmmaking, Tech & Innovation) is minimal. However, an understanding of these forces can indirectly inform advancements in these areas through the materials used and the underlying physical principles.
1. Material Science for Drone Components
- Lightweight and Strong Materials: Drones are constructed from advanced composite materials, plastics, and metals. The synthesis and properties of these materials are governed by the intermolecular forces within their molecular structures. For example, polymers used in drone bodies and propellers are held together by a combination of covalent bonds and weaker intermolecular forces, including LDFs. Optimizing material design for strength-to-weight ratio involves understanding these forces.
- Battery Technology: The electrolytes and electrode materials in drone batteries are complex chemical systems. The interactions between ions, solvent molecules, and electrode surfaces are influenced by LDFs, affecting battery performance, lifespan, and safety.
2. Miniature Sensors and Electronics
- Nanomaterials in Sensors: As drone technology incorporates increasingly sophisticated sensors (e.g., for environmental monitoring, navigation), the use of nanomaterials becomes more prevalent. The behavior of nanoparticles, their surface interactions, and their ability to bind to specific targets are all influenced by LDFs.
- Adhesion in Electronics: The bonding of delicate electronic components to circuit boards or housings in small, vibration-prone devices like drones relies on adhesives. The effectiveness of these adhesives is a testament to controlled intermolecular forces, including LDFs, providing mechanical stability.
3. Camera Stabilization and Gimbal Technology
- Lubrication and Friction: The smooth operation of gimbal systems, which stabilize drone cameras, depends on precisely engineered bearings and lubricants. The properties of these lubricants and the friction between moving parts are influenced by intermolecular forces. Minimizing friction often involves using materials that exhibit weaker LDFs or have specific molecular alignments.
- Material Properties for Optics: The lenses and housings of drone cameras are made from materials whose optical properties are intrinsically linked to their molecular structure and intermolecular interactions.
4. Material Properties for Propellers and Aerodynamics
- Aerodynamic Surfaces: The shape and material of drone propellers are critical for efficient flight. The interaction of air with the propeller surface involves fluid dynamics, where the properties of the air molecules themselves (influenced by LDFs) and the surface chemistry of the propeller material play a role.
- Resilience and Durability: Propellers must withstand significant stress and impacts. The toughness and resilience of the composite materials used are a consequence of the interplay of strong intramolecular bonds and weaker intermolecular forces.
In conclusion, while the direct nomenclature “London Forces” might not appear in typical drone industry jargon, the underlying principles of London Dispersion Forces are fundamental to the very existence and performance of the materials and technologies that enable modern drones. They are a pervasive, albeit often understated, force shaping the physical world around us, from the smallest molecules to the complex components of advanced aerial vehicles.
