Killer T cells, also known scientifically as cytotoxic T lymphocytes (CTLs), are a critical component of our adaptive immune system, playing a vital role in defending the body against intracellular pathogens and cancerous cells. Unlike their B cell counterparts that produce antibodies, Killer T cells are direct effectors, armed with the ability to recognize and eliminate infected or abnormal cells through targeted cytotoxic mechanisms. Their precise and potent action makes them indispensable for maintaining health and preventing the spread of disease.
The Genesis and Evolution of Killer T Cells
The development and maturation of Killer T cells are complex processes orchestrated by the immune system to ensure the generation of highly specific and functional lymphocytes. This journey begins in the bone marrow and culminates in a sophisticated education within the thymus, a central lymphoid organ.
Hematopoiesis: The Ancestral Roots
The lineage of all blood cells, including lymphocytes, traces back to hematopoietic stem cells (HSCs) residing in the bone marrow. These multipotent stem cells possess the remarkable ability to differentiate into various blood cell types, including myeloid and lymphoid progenitors. Early in their development, precursor T cells commit to the T cell lineage. These progenitor cells then migrate from the bone marrow to the thymus to undergo crucial developmental stages. This initial phase of differentiation is fundamental, setting the stage for the specialized functions that Killer T cells will eventually acquire. Without this foundational process of hematopoiesis, the subsequent development and maturation of these crucial immune cells would be impossible.
Thymic Education: A Rigorous Selection Process
The thymus serves as the primary training ground for T cells, where they undergo a rigorous process of selection to ensure they are both self-tolerant and capable of recognizing foreign antigens. This intricate process involves two main stages: positive selection and negative selection.
Positive Selection: Ensuring T Cell Receptor Functionality
Upon entering the thymus, immature T cells, known as thymocytes, begin to express the T cell receptor (TCR). The TCR is a unique protein complex on the surface of T cells that is responsible for recognizing specific antigen fragments presented by other cells. During positive selection, thymocytes whose TCRs can weakly bind to self-MHC (Major Histocompatibility Complex) molecules are selected to survive. MHC molecules are like molecular billboards on the surface of cells, displaying fragments of proteins. If a thymocyte’s TCR cannot interact with self-MHC molecules, it is considered non-functional and is eliminated through apoptosis (programmed cell death). This ensures that surviving T cells are capable of recognizing antigens presented by the body’s own MHC molecules, a prerequisite for effective immune responses.
Negative Selection: Eliminating Self-Reactive Clones
Following positive selection, thymocytes that survived undergo negative selection. In this critical stage, thymocytes with TCRs that bind too strongly to self-MHC-peptide complexes are eliminated. This process is crucial for preventing autoimmune diseases, where the immune system mistakenly attacks the body’s own healthy tissues. The thymus displays a wide array of self-peptides, allowing for the identification and removal of any T cell clone that exhibits a high affinity for self-antigens. This stringent screening process ensures that mature T cells circulating in the periphery are primarily reactive to foreign invaders and not to the body’s own constituents. Only a small fraction of the initial thymocytes survive this dual selection process and mature into functional T cells, including Killer T cells.
Mechanisms of Cytotoxicity: The Killer’s Arsenal
Once matured and released into the bloodstream and lymphatic system, Killer T cells are equipped with a formidable arsenal of molecular tools to identify and eliminate target cells. Their cytotoxic mechanisms are highly specific and efficient, ensuring minimal collateral damage to healthy tissues.
The Role of the T Cell Receptor (TCR) and MHC Class I
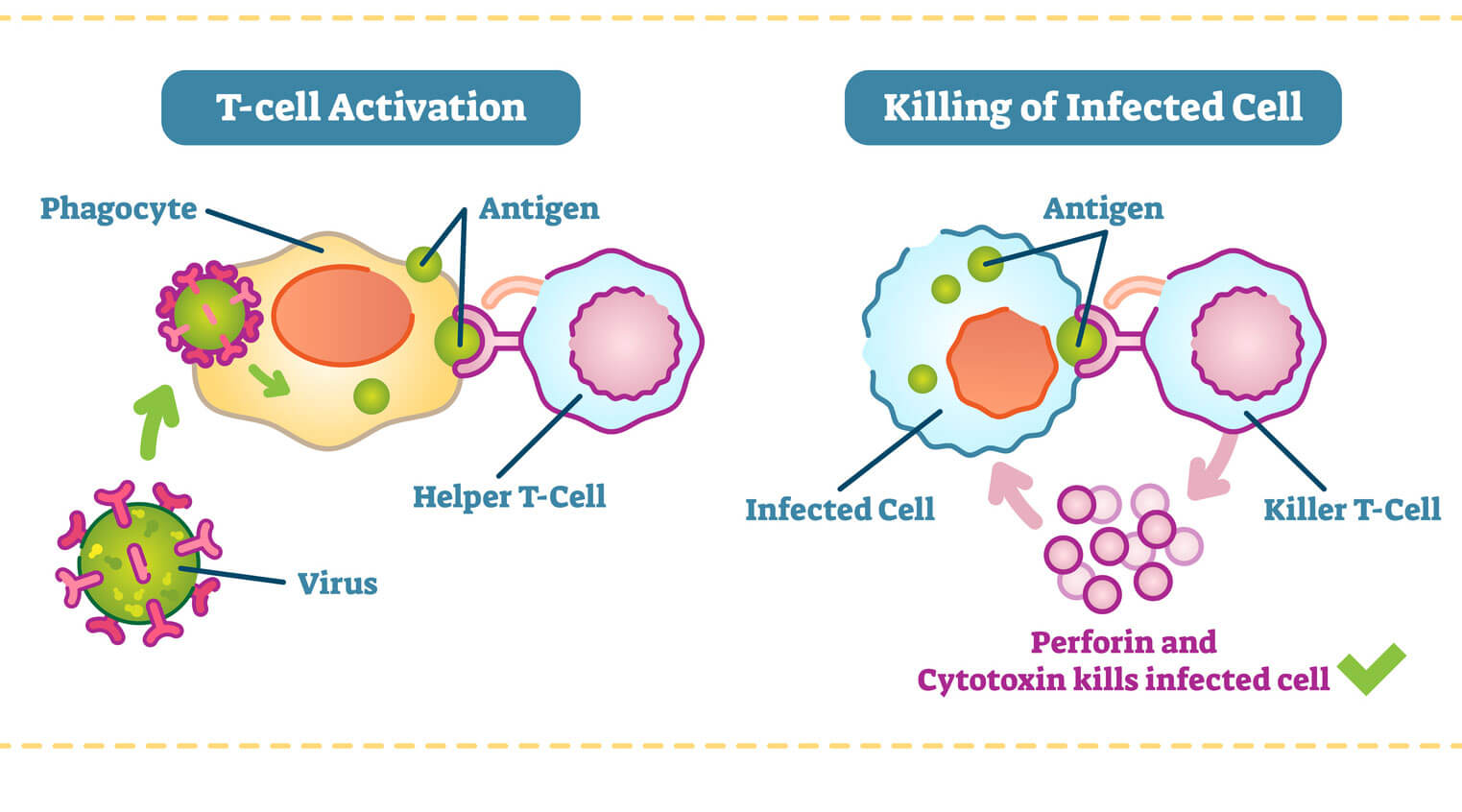
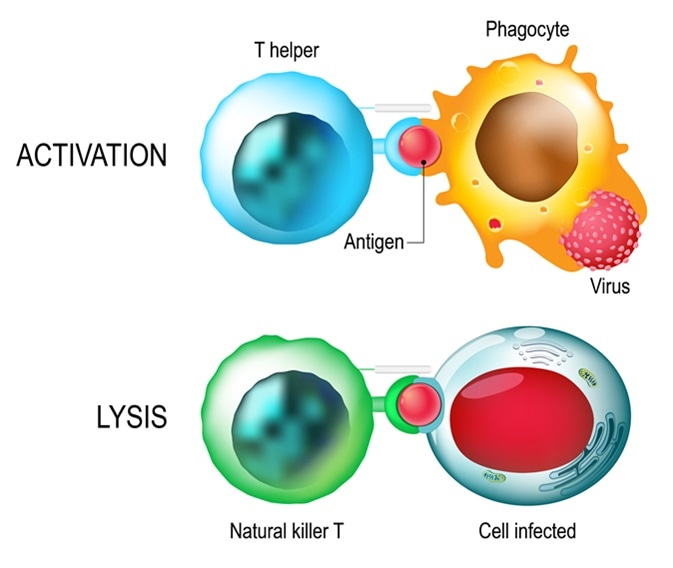
The primary method by which Killer T cells recognize their targets is through the interaction of their T cell receptor (TCR) with specific antigen fragments presented on the surface of target cells. Crucially, this recognition is MHC-restricted. Killer T cells primarily interact with antigens presented by MHC Class I molecules. MHC Class I molecules are found on the surface of almost all nucleated cells in the body. When a cell is infected by a virus or becomes cancerous, it processes viral or abnormal proteins into small peptide fragments. These peptides are then loaded onto MHC Class I molecules and displayed on the cell surface. The Killer T cell’s TCR, along with a co-receptor called CD8, binds to the MHC Class I-peptide complex. This binding event is highly specific, ensuring that Killer T cells only engage with cells presenting antigens they are programmed to recognize.
Perforin and Granzymes: Inducing Apoptosis
Upon successful recognition of a target cell, Killer T cells initiate a cascade of events designed to trigger programmed cell death in the target. Two key components of this cytotoxic machinery are perforin and granzymes.
Perforin: Creating Pores in the Target Membrane
Perforin is a pore-forming protein released by Killer T cells. Upon binding to the target cell, the Killer T cell forms an immunological synapse, a specialized junction between the two cells. Within this synapse, the Killer T cell releases cytotoxic granules containing perforin and granzymes. Perforin molecules then assemble into a ring-like structure within the target cell membrane, forming pores. These pores disrupt the integrity of the target cell membrane, making it permeable to various molecules.
Granzymes: Triggering Programmed Cell Death
Granzymes are a family of serine proteases also stored in the cytotoxic granules. Once perforin has created pores in the target cell membrane, granzymes can enter the target cell. Inside the target cell, granzymes initiate apoptosis by activating key executioner caspases, a group of enzymes that dismantle the cell in a controlled manner. This process involves breaking down cellular DNA, proteins, and organelles, ultimately leading to the formation of apoptotic bodies that are then cleared by phagocytic cells, such as macrophages. This controlled demolition prevents the release of cellular contents, which could otherwise trigger inflammation or spread infection.
Fas Ligand and Fas Receptor: An Alternative Pathway
In addition to the perforin-granzyme pathway, Killer T cells can also induce apoptosis through the Fas ligand (FasL) and Fas receptor (FasR) pathway. The Killer T cell expresses FasL on its surface, while the target cell expresses FasR. When a Killer T cell binds to a target cell, FasL on the Killer T cell can engage with FasR on the target cell. This interaction triggers intracellular signaling pathways within the target cell that also lead to the activation of caspases and the initiation of apoptosis. This pathway provides an alternative mechanism for inducing cell death, particularly effective against certain types of infected or cancerous cells.
The Significance and Therapeutic Potential of Killer T Cells
The remarkable capabilities of Killer T cells extend beyond their fundamental role in innate immunity. Their specificity and cytotoxic power have made them a focal point for innovative therapeutic strategies in the fight against diseases like cancer.
Defense Against Viral Infections
One of the primary roles of Killer T cells is to combat viral infections. Viruses replicate within host cells, making them intracellular pathogens. Once a cell is infected, it displays viral antigens on its MHC Class I molecules, flagging it for destruction by Killer T cells. By eliminating infected cells, Killer T cells prevent the virus from replicating and spreading to new cells, thereby controlling the infection. For instance, in infections like influenza or hepatitis, Killer T cells are crucial for clearing infected hepatocytes and respiratory epithelial cells. Their ability to swiftly target and destroy infected cells is vital for preventing the establishment of chronic infections and severe disease.
Immune Surveillance and Cancer Elimination
Killer T cells are also essential for immune surveillance, a continuous process by which the immune system patrols the body for signs of disease. This includes the detection and elimination of cancerous cells. Cancer cells often acquire mutations that lead to the expression of abnormal proteins, which can be presented on MHC Class I molecules. Killer T cells are capable of recognizing these tumor-associated antigens and eliminating the nascent cancer cells before they can proliferate and form tumors. This constant surveillance is a critical defense mechanism that prevents many potentially life-threatening cancers from developing.
CAR T-Cell Therapy: Harnessing the Power of Killer T Cells
The groundbreaking development of Chimeric Antigen Receptor (CAR) T-cell therapy represents a paradigm shift in cancer treatment, largely leveraging the inherent cytotoxic power of Killer T cells. In CAR T-cell therapy, a patient’s own T cells, including Killer T cells, are collected and genetically engineered in a laboratory. This genetic modification involves introducing a synthetic receptor, the CAR, onto the surface of the T cells. The CAR is designed to specifically recognize and bind to a particular antigen found on the surface of cancer cells. Once engineered, these CAR T cells are expanded in number and then infused back into the patient. Upon re-entry into the body, the CAR T cells actively seek out and destroy cancer cells that express the targeted antigen. This therapy has shown remarkable success in treating certain types of blood cancers, such as B-cell leukemias and lymphomas, offering a potent and personalized approach to cancer immunotherapy. The ability to redirect the specificity of Killer T cells to target cancer cells has opened new avenues for treating previously intractable malignancies.
