The intricate network of blood vessels that permeates every tissue and organ in the human body is fundamental for life, delivering oxygen and nutrients while removing waste products. Maintaining the integrity and functionality of this vast system, known as the endothelium, is crucial for health. When the endothelium is damaged, whether by injury, disease, or aging, the body initiates complex repair mechanisms. Central to these reparative processes are specialized cells known as Endothelial Progenitor Cells (EPCs). These remarkable cells represent a fascinating frontier in biological research and regenerative medicine, holding immense promise for treating a wide array of vascular diseases.
Endothelial Progenitor Cells are a population of immature cells that possess the capacity to differentiate into mature endothelial cells, which form the inner lining of blood vessels. Unlike mature endothelial cells that primarily perform barrier functions and regulate vascular tone, EPCs are characterized by their proliferative capacity, migratory potential, and ability to contribute to the formation of new blood vessels, a process known as vasculogenesis or angiogenesis. Their discovery revolutionized our understanding of vascular repair and development, shifting the paradigm from viewing the endothelium as a static structure to a dynamic tissue capable of continuous self-renewal and regeneration.
The Discovery and Defining Characteristics of EPCs
The concept of circulating cells contributing to new blood vessel formation was not entirely new, but the definitive identification of EPCs in adult peripheral blood in 1997 by Asahara and colleagues marked a pivotal moment. This groundbreaking discovery provided concrete evidence that adults retain a population of stem-like cells capable of differentiating into endothelial cells and participating in vascular regeneration.
Early Identification and Historical Context
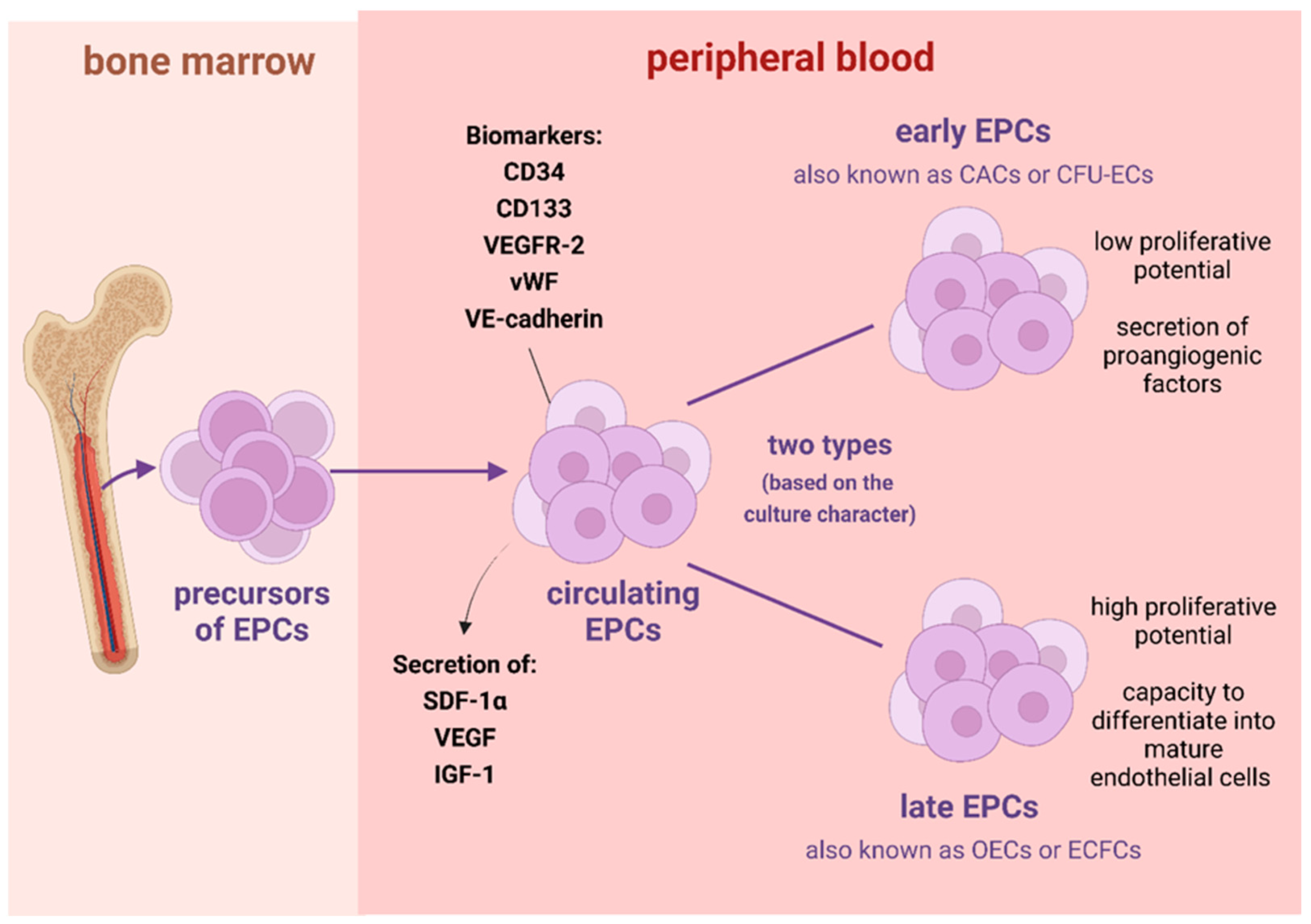
Prior to Asahara’s work, it was largely believed that post-natal angiogenesis relied solely on the sprouting of existing mature endothelial cells. The discovery of EPCs introduced the idea of de novo vessel formation from progenitor cells, a process termed vasculogenesis, which was previously thought to be restricted to embryonic development. The initial identification of EPCs relied on their expression of certain cell surface markers, such as CD34 and CD133 (also found on hematopoietic stem cells), and their ability to uptake acetylated low-density lipoprotein (Ac-LDL) and bind to Ulex europaeus agglutinin I, characteristics shared with mature endothelial cells. This dual expression profile—of stem cell markers and endothelial markers—underlined their transitional nature.
Phenotypic Markers and Heterogeneity
Since their initial identification, the understanding of EPCs has evolved significantly, revealing a complex and heterogeneous population. There isn’t a single, universally accepted set of markers to definitively identify EPCs, largely due to their plasticity and the varying methods used for their isolation and characterization. However, common markers include CD34, CD133, and vascular endothelial growth factor receptor 2 (VEGFR2/KDR). Some researchers further categorize EPCs based on their proliferative capacity and morphology in culture:
- Early EPCs (or Non-Adherent EPCs): These cells appear within a few days of culture, form colonies, and have limited proliferative potential. They are thought to primarily release paracrine factors that support existing endothelial cells and promote angiogenesis.
- Late EPCs (or Endothelial Colony Forming Cells – ECFCs): These emerge later in culture (2-4 weeks), exhibit robust proliferative capacity, form extensive colonies, and possess strong angiogenic potential, directly forming tubular structures in vitro and contributing to vessel formation in vivo. ECFCs are considered true endothelial progenitors with long-term proliferative and clonogenic potential.
This heterogeneity underscores the complexity of EPC biology and suggests that different subsets of EPCs may play distinct roles in various physiological and pathological contexts.
Origin and Niche
While bone marrow is widely accepted as a primary source of EPCs, where they originate from hematopoietic stem cells, other potential sources have been identified. These include peripheral blood, umbilical cord blood, and possibly even resident tissue-specific stem cells in various organs. Once mobilized from their niche, EPCs circulate in the bloodstream, migrating to sites of vascular injury or ischemia where they are recruited by specific chemokines and growth factors. The bone marrow microenvironment, rich in various cytokines and growth factors, plays a critical role in regulating EPC quiescence, proliferation, and mobilization.
Diverse Functions of Endothelial Progenitor Cells in Vascular Homeostasis
EPCs are not merely passive participants in vascular repair; they actively contribute to maintaining vascular health through multiple mechanisms, making them integral players in both physiological processes and disease states.
Role in Angiogenesis and Vasculogenesis
The most celebrated function of EPCs is their direct contribution to the formation of new blood vessels. In situations like tissue ischemia (e.g., after a heart attack or stroke, or in peripheral artery disease), EPCs are mobilized from the bone marrow and recruited to the ischemic site. Here, they can differentiate into mature endothelial cells and incorporate into new blood vessels (vasculogenesis), or they can support the sprouting of existing vessels (angiogenesis) by providing growth factors and cytokines. This dual capacity makes them powerful agents for revascularization, enhancing blood flow to deprived tissues and improving tissue survival and function.
Contribution to Endothelial Repair and Regeneration
Beyond forming entirely new vessels, EPCs are vital for repairing damaged endothelium. The endothelium is constantly subjected to shear stress, inflammation, and oxidative damage, leading to endothelial dysfunction—a precursor to many cardiovascular diseases. EPCs home to these damaged areas, replacing lost or dysfunctional endothelial cells, thereby restoring the integrity of the vascular lining. This reparative function is critical in preventing atherosclerotic plaque formation and progression, as a healthy endothelium acts as a non-thrombogenic surface and regulates vascular permeability and tone.
Immunomodulatory Properties
Emerging research indicates that EPCs possess significant immunomodulatory capabilities. They can interact with various immune cells, influencing inflammatory responses and contributing to immune regulation. For instance, EPCs have been shown to dampen inflammatory reactions, secrete anti-inflammatory cytokines, and promote the resolution of inflammation, which is particularly relevant in chronic vascular diseases where inflammation plays a key role. This immunomodulatory aspect broadens their therapeutic potential beyond mere structural repair, positioning them as cellular agents capable of influencing the microenvironment in damaged tissues.
Therapeutic Potential: EPCs in Regenerative Medicine
The unique properties of EPCs have fueled extensive research into their potential applications in regenerative medicine, particularly for cardiovascular diseases, which remain the leading cause of mortality worldwide.
Cardiovascular Diseases (Ischemia, Atherosclerosis)
In conditions like myocardial infarction (heart attack) and critical limb ischemia, where blood supply to tissues is severely compromised, EPC-based therapies aim to enhance natural revascularization. Clinical trials exploring the infusion of autologous (patient’s own) EPCs have shown promising results in improving cardiac function, reducing anginal symptoms, and promoting blood flow in ischemic limbs. By facilitating the formation of collateral vessels and repairing damaged endothelium, EPCs offer a compelling strategy to mitigate tissue damage and improve patient outcomes. Similarly, in atherosclerosis, where plaque buildup narrows arteries, EPCs may help stabilize plaques and improve endothelial function, potentially slowing disease progression.
Peripheral Artery Disease (PAD)
PAD affects millions globally, leading to reduced blood flow to the limbs, often resulting in pain, ulcers, and in severe cases, amputation. EPC therapy offers a non-surgical option for patients with severe PAD who are not candidates for traditional revascularization. Direct injection of EPCs into ischemic muscles has demonstrated the potential to improve microcirculation, reduce pain, and promote wound healing in preclinical models and early clinical trials.
Wound Healing and Tissue Engineering
The angiogenic and reparative capacities of EPCs are also highly relevant in chronic wound healing, particularly in patients with diabetes, where impaired angiogenesis and endothelial dysfunction contribute to persistent ulcers. EPCs can accelerate wound closure by promoting vascularization within the wound bed. In tissue engineering, EPCs are being incorporated into scaffolds and biomaterials to create vascularized constructs, which is a significant challenge in engineering larger tissues and organs for transplantation or repair. Creating functional vascular networks within engineered tissues is essential for nutrient supply and waste removal, and EPCs are key to achieving this.
Challenges and Future Directions in Clinical Application
Despite their immense potential, the clinical application of EPCs faces several challenges. These include standardizing isolation and expansion protocols, ensuring cell viability and homing efficiency, and determining optimal dosing and delivery methods. The quality and quantity of EPCs can vary significantly among individuals, especially in patients with comorbidities like diabetes or advanced age, which often leads to dysfunctional or fewer EPCs. Future research is focused on enhancing EPC function through genetic modification, optimizing ex vivo expansion techniques, combining EPCs with growth factors or biomaterials, and developing strategies to improve endogenous EPC mobilization. Furthermore, understanding the precise mechanisms of action and developing more specific targeting strategies will be crucial for translating EPC therapy into routine clinical practice.
Factors Influencing EPC Mobilization and Function
The efficacy of endogenous EPCs in vascular repair is significantly influenced by various physiological and pathological factors, as well as therapeutic interventions.
Physiological Stimuli (Exercise, Hypoxia)
Certain physiological stimuli are known to enhance EPC mobilization and function. Regular physical exercise, for instance, has been consistently shown to increase circulating EPC levels and improve their functional capacity, contributing to cardiovascular health. Moderate hypoxia (low oxygen levels) can also trigger EPC release from the bone marrow, as the body attempts to compensate for oxygen deficit by promoting angiogenesis. These natural mechanisms highlight the body’s intrinsic capacity for vascular repair and suggest lifestyle interventions as complementary strategies to enhance EPC activity.
Pathological Conditions (Diabetes, Renal Disease)
Conversely, many pathological conditions impair EPC number and function. Chronic diseases such as diabetes mellitus, chronic kidney disease, hypertension, and hypercholesterolemia are associated with reduced EPC counts, impaired migratory capacity, and decreased angiogenic potential. This impairment contributes to the accelerated progression of vascular complications observed in these patient populations, as the body’s natural reparative mechanisms are compromised. Understanding these deleterious effects is crucial for developing targeted therapies that can restore EPC function in diseased individuals.
Pharmacological Modulators
A range of pharmacological agents has been identified that can influence EPCs. Statins, commonly prescribed for cholesterol reduction, have been shown to increase EPC mobilization and improve their function, contributing to their pleiotropic cardiovascular benefits. Angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs), used for hypertension and heart failure, also positively affect EPCs. Similarly, certain growth factors like vascular endothelial growth factor (VEGF) and granulocyte colony-stimulating factor (G-CSF) are potent mobilizers of EPCs and are being explored for therapeutic use. Elucidating the precise mechanisms by which these drugs interact with EPCs can lead to the development of novel pharmacological strategies to enhance vascular repair.
In conclusion, Endothelial Progenitor Cells represent a dynamic and versatile cell population critical for maintaining vascular homeostasis, repairing damaged vessels, and fostering new blood vessel formation. Their discovery has opened new avenues in understanding vascular biology and offers compelling therapeutic strategies for a multitude of vascular diseases. While challenges remain in harnessing their full clinical potential, ongoing research continues to unveil their complex biology and refine approaches for their application, promising a future where EPC-based therapies could revolutionize regenerative medicine and significantly improve patient outcomes for those suffering from cardiovascular and other vascular-related conditions. The journey from basic science to clinical translation for EPCs is a testament to the power of continuous technological and innovative advancements in biotechnology.
